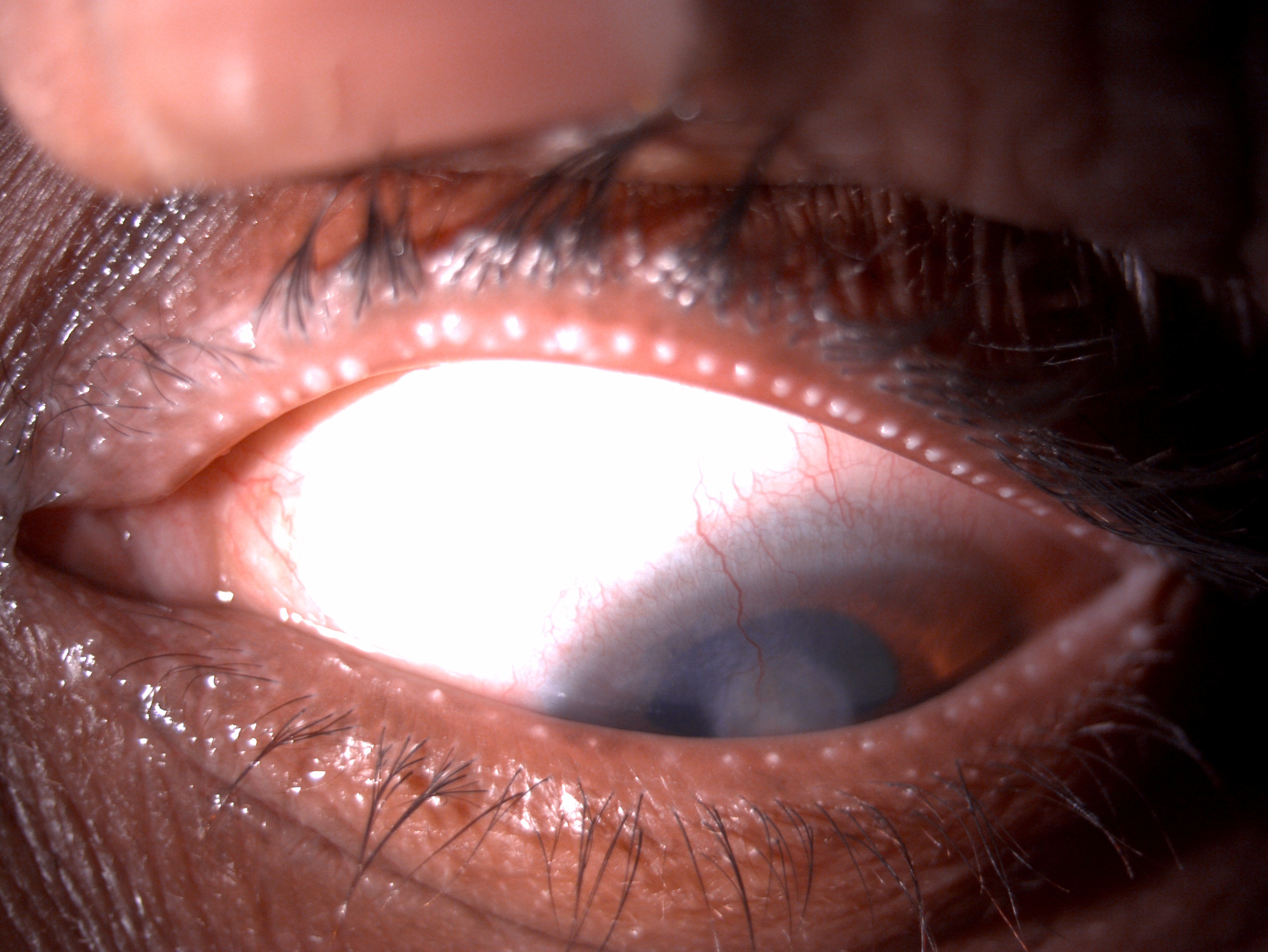
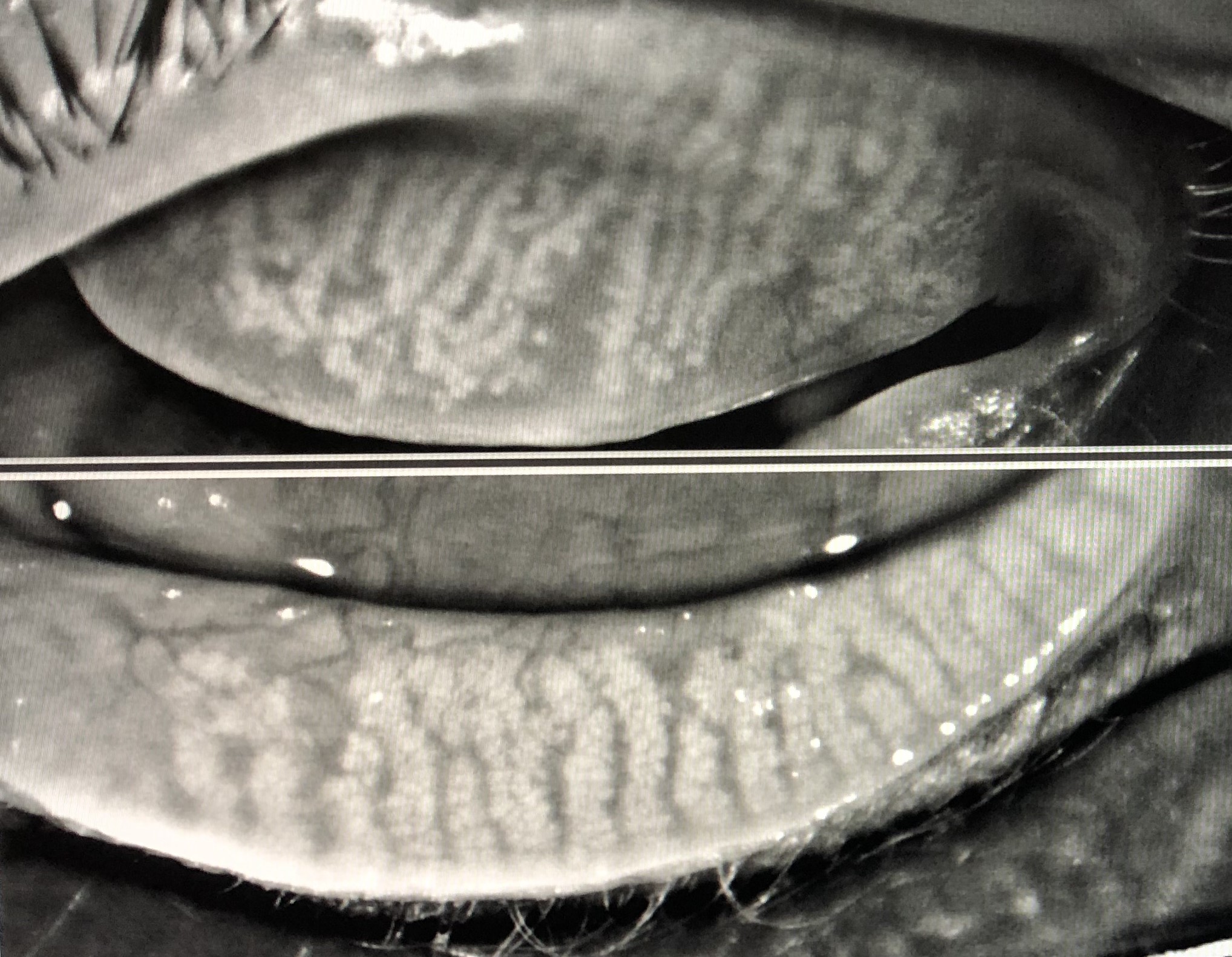
[1]
Nelson JD, Shimazaki J, Benitez-del-Castillo JM, Craig JP, McCulley JP, Den S, Foulks GN. The international workshop on meibomian gland dysfunction: report of the definition and classification subcommittee. Investigative ophthalmology & visual science. 2011 Mar:52(4):1930-7. doi: 10.1167/iovs.10-6997b. Epub 2011 Mar 30
[PubMed PMID: 21450914]
[2]
Milner MS, Beckman KA, Luchs JI, Allen QB, Awdeh RM, Berdahl J, Boland TS, Buznego C, Gira JP, Goldberg DF, Goldman D, Goyal RK, Jackson MA, Katz J, Kim T, Majmudar PA, Malhotra RP, McDonald MB, Rajpal RK, Raviv T, Rowen S, Shamie N, Solomon JD, Stonecipher K, Tauber S, Trattler W, Walter KA, Waring GO 4th, Weinstock RJ, Wiley WF, Yeu E. Dysfunctional tear syndrome: dry eye disease and associated tear film disorders - new strategies for diagnosis and treatment. Current opinion in ophthalmology. 2017 Jan:27 Suppl 1(Suppl 1):3-47. doi: 10.1097/01.icu.0000512373.81749.b7. Epub
[PubMed PMID: 28099212]
Level 3 (low-level) evidence
[3]
Knop E, Knop N, Millar T, Obata H, Sullivan DA. The international workshop on meibomian gland dysfunction: report of the subcommittee on anatomy, physiology, and pathophysiology of the meibomian gland. Investigative ophthalmology & visual science. 2011 Mar:52(4):1938-78. doi: 10.1167/iovs.10-6997c. Epub 2011 Mar 30
[PubMed PMID: 21450915]
[4]
Tomlinson A, Bron AJ, Korb DR, Amano S, Paugh JR, Pearce EI, Yee R, Yokoi N, Arita R, Dogru M. The international workshop on meibomian gland dysfunction: report of the diagnosis subcommittee. Investigative ophthalmology & visual science. 2011 Mar:52(4):2006-49. doi: 10.1167/iovs.10-6997f. Epub 2011 Mar 30
[PubMed PMID: 21450918]
[5]
Chhadva P, Goldhardt R, Galor A. Meibomian Gland Disease: The Role of Gland Dysfunction in Dry Eye Disease. Ophthalmology. 2017 Nov:124(11S):S20-S26. doi: 10.1016/j.ophtha.2017.05.031. Epub
[PubMed PMID: 29055358]
[6]
Geerling G, Baudouin C, Aragona P, Rolando M, Boboridis KG, Benítez-Del-Castillo JM, Akova YA, Merayo-Lloves J, Labetoulle M, Steinhoff M, Messmer EM. Emerging strategies for the diagnosis and treatment of meibomian gland dysfunction: Proceedings of the OCEAN group meeting. The ocular surface. 2017 Apr:15(2):179-192. doi: 10.1016/j.jtos.2017.01.006. Epub 2017 Jan 27
[PubMed PMID: 28132878]
[7]
Putnam CM. Diagnosis and management of blepharitis: an optometrist's perspective. Clinical optometry. 2016:8():71-78. doi: 10.2147/OPTO.S84795. Epub 2016 Aug 8
[PubMed PMID: 30214351]
Level 3 (low-level) evidence
[8]
Schaumberg DA, Nichols JJ, Papas EB, Tong L, Uchino M, Nichols KK. The international workshop on meibomian gland dysfunction: report of the subcommittee on the epidemiology of, and associated risk factors for, MGD. Investigative ophthalmology & visual science. 2011 Mar:52(4):1994-2005. doi: 10.1167/iovs.10-6997e. Epub 2011 Mar 30
[PubMed PMID: 21450917]
[9]
Gurlevik U, Kemeriz F, Yasar E. The effect of isotretinoin on meibomian glands in eyes: a pilot study. International ophthalmology. 2022 Jul:42(7):2071-2078. doi: 10.1007/s10792-021-02205-1. Epub 2022 Jan 3
[PubMed PMID: 34978652]
Level 3 (low-level) evidence
[10]
Huang J, Guo MX, Xiang DM, Yan LF, Yu Y, Han L, Wang JX, Lu XH. The association of demodex infestation with pediatric chalazia. BMC ophthalmology. 2022 Mar 16:22(1):124. doi: 10.1186/s12886-022-02261-w. Epub 2022 Mar 16
[PubMed PMID: 35291979]
[11]
Lee WJ, Kim M, Lee SH, Chun YS, Kim KW. The varied influence of ocular Demodex infestation on dry eye disease and meibomian gland dysfunction across different age groups. Scientific reports. 2023 Sep 28:13(1):16324. doi: 10.1038/s41598-023-43674-x. Epub 2023 Sep 28
[PubMed PMID: 37770583]
[12]
Ding G, Tan Y, Zhang C, Zhang Y, Wang X. Analysis of Demodex infection rate and risk factors in patients with meibomian gland dysfunction. International ophthalmology. 2023 Mar:43(3):877-884. doi: 10.1007/s10792-022-02490-4. Epub 2022 Sep 15
[PubMed PMID: 36109404]
[13]
Hassanzadeh S, Varmaghani M, Zarei-Ghanavati S, Heravian Shandiz J, Azimi Khorasani A. Global Prevalence of Meibomian Gland Dysfunction: A Systematic Review and Meta-Analysis. Ocular immunology and inflammation. 2021 Jan 2:29(1):66-75. doi: 10.1080/09273948.2020.1755441. Epub 2020 Jun 26
[PubMed PMID: 32589483]
Level 1 (high-level) evidence
[14]
Suzuki T. Inflamed Obstructive Meibomian Gland Dysfunction Causes Ocular Surface Inflammation. Investigative ophthalmology & visual science. 2018 Nov 1:59(14):DES94-DES101. doi: 10.1167/iovs.17-23345. Epub
[PubMed PMID: 30481812]
[15]
Messmer EM. The pathophysiology, diagnosis, and treatment of dry eye disease. Deutsches Arzteblatt international. 2015 Jan 30:112(5):71-81; quiz 82. doi: 10.3238/arztebl.2015.0071. Epub
[PubMed PMID: 25686388]
[16]
Gutgesell VJ, Stern GA, Hood CI. Histopathology of meibomian gland dysfunction. American journal of ophthalmology. 1982 Sep:94(3):383-7
[PubMed PMID: 7124880]
[18]
Efron N. Grading scales for contact lens complications. Ophthalmic & physiological optics : the journal of the British College of Ophthalmic Opticians (Optometrists). 1998 Mar:18(2):182-6
[PubMed PMID: 9692040]
[19]
Paugh JR, Kwan J, Christensen M, Nguyen AL, Senchyna M, Meadows D. Development of a Meibomian Gland Dysfunction-Specific Symptom Questionnaire. Eye & contact lens. 2018 Jan:44(1):6-14. doi: 10.1097/ICL.0000000000000294. Epub
[PubMed PMID: 27466720]
[20]
Okumura Y, Inomata T, Iwata N, Sung J, Fujimoto K, Fujio K, Midorikawa-Inomata A, Miura M, Akasaki Y, Murakami A. A Review of Dry Eye Questionnaires: Measuring Patient-Reported Outcomes and Health-Related Quality of Life. Diagnostics (Basel, Switzerland). 2020 Aug 5:10(8):. doi: 10.3390/diagnostics10080559. Epub 2020 Aug 5
[PubMed PMID: 32764273]
Level 2 (mid-level) evidence
[21]
Ha M, Kim JS, Hong SY, Chang DJ, Whang WJ, Na KS, Kim EC, Kim HS, Hwang HS. Relationship between eyelid margin irregularity and meibomian gland dropout. The ocular surface. 2021 Jan:19():31-37. doi: 10.1016/j.jtos.2020.11.007. Epub 2020 Nov 24
[PubMed PMID: 33246034]
[22]
Robin M, Liang H, Rabut G, Augstburger E, Baudouin C, Labbé A. The Role of Meibography in the Diagnosis of Meibomian Gland Dysfunction in Ocular Surface Diseases. Translational vision science & technology. 2019 Nov:8(6):6. doi: 10.1167/tvst.8.6.6. Epub 2019 Nov 12
[PubMed PMID: 31737430]
[23]
Wise RJ, Sobel RK, Allen RC. Meibography: A review of techniques and technologies. Saudi journal of ophthalmology : official journal of the Saudi Ophthalmological Society. 2012 Oct:26(4):349-56. doi: 10.1016/j.sjopt.2012.08.007. Epub
[PubMed PMID: 23961019]
[24]
Lee JM, Jeon YJ, Kim KY, Hwang KY, Kwon YA, Koh K. Ocular surface analysis: A comparison between the LipiView(®) II and IDRA(®). European journal of ophthalmology. 2021 Sep:31(5):2300-2306. doi: 10.1177/1120672120969035. Epub 2020 Dec 2
[PubMed PMID: 33267698]
[25]
Arita R, Fukuoka S, Kawashima M. Proposed Algorithm for Management of Meibomian Gland Dysfunction Based on Noninvasive Meibography. Journal of clinical medicine. 2020 Dec 27:10(1):. doi: 10.3390/jcm10010065. Epub 2020 Dec 27
[PubMed PMID: 33375436]
[26]
Sabeti S, Kheirkhah A, Yin J, Dana R. Management of meibomian gland dysfunction: a review. Survey of ophthalmology. 2020 Mar-Apr:65(2):205-217. doi: 10.1016/j.survophthal.2019.08.007. Epub 2019 Sep 5
[PubMed PMID: 31494111]
Level 3 (low-level) evidence
[27]
Yokoi N, Bron AJ, Tiffany JM, Maruyama K, Komuro A, Kinoshita S. Relationship between tear volume and tear meniscus curvature. Archives of ophthalmology (Chicago, Ill. : 1960). 2004 Sep:122(9):1265-9
[PubMed PMID: 15364704]
[28]
Wong S, Srinivasan S, Murphy PJ, Jones L. Comparison of meibomian gland dropout using two infrared imaging devices. Contact lens & anterior eye : the journal of the British Contact Lens Association. 2019 Jun:42(3):311-317. doi: 10.1016/j.clae.2018.10.014. Epub 2018 Nov 7
[PubMed PMID: 30413376]
[29]
Daniel E, Maguire MG, Pistilli M, Bunya VY, Massaro-Giordano GM, Smith E, Kadakia PA, Asbell PA, Dry Eye Assessment and Management (DREAM) Study Research Group. Grading and baseline characteristics of meibomian glands in meibography images and their clinical associations in the Dry Eye Assessment and Management (DREAM) study. The ocular surface. 2019 Jul:17(3):491-501. doi: 10.1016/j.jtos.2019.04.003. Epub 2019 Apr 22
[PubMed PMID: 31022469]
[30]
Pucker AD, Jones-Jordan LA, Marx S, Powell DR, Kwan JT, Srinivasan S, Sickenberger W, Jones L, Contact Lens Assessment of Symptomatic Subjects (CLASS) Study Group. Clinical factors associated with contact lens dropout. Contact lens & anterior eye : the journal of the British Contact Lens Association. 2019 Jun:42(3):318-324. doi: 10.1016/j.clae.2018.12.002. Epub 2018 Dec 8
[PubMed PMID: 30538060]
[31]
Rolando M, Merayo-Lloves J. Management Strategies for Evaporative Dry Eye Disease and Future Perspective. Current eye research. 2022 Jun:47(6):813-823. doi: 10.1080/02713683.2022.2039205. Epub 2022 May 6
[PubMed PMID: 35521685]
Level 3 (low-level) evidence
[32]
Swiderska K, Read ML, Blackie CA, Maldonado-Codina C, Morgan PB. Latest developments in meibography: A review. The ocular surface. 2022 Jul:25():119-128. doi: 10.1016/j.jtos.2022.06.002. Epub 2022 Jun 18
[PubMed PMID: 35724917]
[33]
Nichols KK, Foulks GN, Bron AJ, Glasgow BJ, Dogru M, Tsubota K, Lemp MA, Sullivan DA. The international workshop on meibomian gland dysfunction: executive summary. Investigative ophthalmology & visual science. 2011 Mar 30:52(4):1922-9. doi: 10.1167/iovs.10-6997a. Epub 2011 Mar 30
[PubMed PMID: 21450913]
[34]
Kocamiş Ö, Temel E, Aşikgarip N, Örnek K. Electronic Device Screen Time and Meibomian Gland Morphology in Children. Journal of ophthalmic & vision research. 2021 Oct-Dec:16(4):531-537. doi: 10.18502/jovr.v16i4.9741. Epub 2021 Oct 25
[PubMed PMID: 34840674]
[35]
Macsai MS. The role of omega-3 dietary supplementation in blepharitis and meibomian gland dysfunction (an AOS thesis). Transactions of the American Ophthalmological Society. 2008:106():336-56
[PubMed PMID: 19277245]
[36]
Oleñik A, Mahillo-Fernández I, Alejandre-Alba N, Fernández-Sanz G, Pérez MA, Luxan S, Quintana S, Martínez de Carneros Llorente A, García-Sandoval B, Jiménez-Alfaro I. Benefits of omega-3 fatty acid dietary supplementation on health-related quality of life in patients with meibomian gland dysfunction. Clinical ophthalmology (Auckland, N.Z.). 2014:8():831-6. doi: 10.2147/OPTH.S62470. Epub 2014 Apr 30
[PubMed PMID: 24812490]
Level 2 (mid-level) evidence
[37]
Garrigue JS, Amrane M, Faure MO, Holopainen JM, Tong L. Relevance of Lipid-Based Products in the Management of Dry Eye Disease. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2017 Nov:33(9):647-661. doi: 10.1089/jop.2017.0052. Epub 2017 Sep 28
[PubMed PMID: 28956698]
[38]
Kim HY, Lee JE, Oh HN, Song JW, Han SY, Lee JS. Clinical efficacy of combined topical 0.05% cyclosporine A and 0.1% sodium hyaluronate in the dry eyes with meibomian gland dysfunction. International journal of ophthalmology. 2018:11(4):593-600. doi: 10.18240/ijo.2018.04.09. Epub 2018 Apr 18
[PubMed PMID: 29675376]
[39]
Qiao J, Yan X. Emerging treatment options for meibomian gland dysfunction. Clinical ophthalmology (Auckland, N.Z.). 2013:7():1797-803. doi: 10.2147/OPTH.S33182. Epub 2013 Sep 9
[PubMed PMID: 24043929]
Level 2 (mid-level) evidence
[40]
Pflugfelder SC, Cao A, Galor A, Nichols KK, Cohen NA, Dalton M. Nicotinic acetylcholine receptor stimulation: A new approach for stimulating tear secretion in dry eye disease. The ocular surface. 2022 Jul:25():58-64. doi: 10.1016/j.jtos.2022.05.001. Epub 2022 May 10
[PubMed PMID: 35550851]
[41]
Maskin SL. Intraductal meibomian gland probing relieves symptoms of obstructive meibomian gland dysfunction. Cornea. 2010 Oct:29(10):1145-52. doi: 10.1097/ICO.0b013e3181d836f3. Epub
[PubMed PMID: 20622668]
[42]
Magno M, Moschowits E, Arita R, Vehof J, Utheim TP. Intraductal meibomian gland probing and its efficacy in the treatment of meibomian gland dysfunction. Survey of ophthalmology. 2021 Jul-Aug:66(4):612-622. doi: 10.1016/j.survophthal.2020.11.005. Epub 2021 Jan 29
[PubMed PMID: 33352147]
Level 3 (low-level) evidence
[43]
Syed ZA, Sutula FC. Dynamic Intraductal Meibomian Probing: A Modified Approach to the Treatment of Obstructive Meibomian Gland Dysfunction. Ophthalmic plastic and reconstructive surgery. 2017 Jul/Aug:33(4):307-309. doi: 10.1097/IOP.0000000000000876. Epub
[PubMed PMID: 28221295]
[44]
Lane SS, DuBiner HB, Epstein RJ, Ernest PH, Greiner JV, Hardten DR, Holland EJ, Lemp MA, McDonald JE 2nd, Silbert DI, Blackie CA, Stevens CA, Bedi R. A new system, the LipiFlow, for the treatment of meibomian gland dysfunction. Cornea. 2012 Apr:31(4):396-404. doi: 10.1097/ICO.0b013e318239aaea. Epub
[PubMed PMID: 22222996]
[45]
Greiner JV. Long-Term (3 Year) Effects of a Single Thermal Pulsation System Treatment on Meibomian Gland Function and Dry Eye Symptoms. Eye & contact lens. 2016 Mar:42(2):99-107. doi: 10.1097/ICL.0000000000000166. Epub
[PubMed PMID: 26222095]
[46]
Finis D, König C, Hayajneh J, Borrelli M, Schrader S, Geerling G. Six-month effects of a thermodynamic treatment for MGD and implications of meibomian gland atrophy. Cornea. 2014 Dec:33(12):1265-70. doi: 10.1097/ICO.0000000000000273. Epub
[PubMed PMID: 25321941]
[47]
Wesley G, Bickle K, Downing J, Fisher B, Greene B, Heinrich C, Kading D, Kannarr S, Miller J, Modi S, Ludwick D, Tauber J, Srinivasan S, Manoj V. Comparison of Two Thermal Pulsation Systems in the Treatment of Meibomian Gland Dysfunction: A Randomized, Multicenter Study. Optometry and vision science : official publication of the American Academy of Optometry. 2022 Apr 1:99(4):323-332. doi: 10.1097/OPX.0000000000001892. Epub
[PubMed PMID: 35383732]
Level 1 (high-level) evidence
[48]
Li S, Yang K, Wang J, Li S, Zhu L, Feng J, Tian L, Jie Y. Effect of a Novel Thermostatic Device on Meibomian Gland Dysfunction: A Randomized Controlled Trial in Chinese Patients. Ophthalmology and therapy. 2022 Feb:11(1):261-270. doi: 10.1007/s40123-021-00431-5. Epub 2021 Nov 25
[PubMed PMID: 34822140]
Level 1 (high-level) evidence
[49]
Goldberg DJ. Current trends in intense pulsed light. The Journal of clinical and aesthetic dermatology. 2012 Jun:5(6):45-53
[PubMed PMID: 22768357]
[50]
Bäumler W, Vural E, Landthaler M, Muzzi F, Shafirstein G. The effects of intense pulsed light (IPL) on blood vessels investigated by mathematical modeling. Lasers in surgery and medicine. 2007 Feb:39(2):132-9
[PubMed PMID: 17066482]
[51]
Brinton M, Kossler AL, Patel ZM, Loudin J, Franke M, Ta CN, Palanker D. Enhanced Tearing by Electrical Stimulation of the Anterior Ethmoid Nerve. Investigative ophthalmology & visual science. 2017 Apr 1:58(4):2341-2348. doi: 10.1167/iovs.16-21362. Epub
[PubMed PMID: 28431436]
[52]
Bhandari V, Reddy JK. Blepharitis: always remember demodex. Middle East African journal of ophthalmology. 2014 Oct-Dec:21(4):317-20. doi: 10.4103/0974-9233.142268. Epub
[PubMed PMID: 25371637]
[53]
Kassan SS, Moutsopoulos HM. Clinical manifestations and early diagnosis of Sjögren syndrome. Archives of internal medicine. 2004 Jun 28:164(12):1275-84
[PubMed PMID: 15226160]
[54]
Vazirani J, Donthineni PR, Goel S, Sane SS, Mahuvakar S, Narang P, Shanbhag SS, Basu S. Chronic cicatrizing conjunctivitis: A review of the differential diagnosis and an algorithmic approach to management. Indian journal of ophthalmology. 2020 Nov:68(11):2349-2355. doi: 10.4103/ijo.IJO_604_20. Epub
[PubMed PMID: 33120615]
[55]
Cope JR, Konne NM, Jacobs DS, Dhaliwal DK, Rhee MK, Yin J, Steinemann TL. Corneal Infections Associated with Sleeping in Contact Lenses - Six Cases, United States, 2016-2018. MMWR. Morbidity and mortality weekly report. 2018 Aug 17:67(32):877-881. doi: 10.15585/mmwr.mm6732a2. Epub 2018 Aug 17
[PubMed PMID: 30114003]
Level 3 (low-level) evidence
[56]
Zhang XB, Ding YH, He W. The association between demodex infestation and ocular surface manifestations in meibomian gland dysfunction. International journal of ophthalmology. 2018:11(4):589-592. doi: 10.18240/ijo.2018.04.08. Epub 2018 Apr 18
[PubMed PMID: 29675375]
[57]
Arita R, Suehiro J, Haraguchi T, Maeda S, Maeda K, Tokoro H, Amano S. Topical diquafosol for patients with obstructive meibomian gland dysfunction. The British journal of ophthalmology. 2013 Jun:97(6):725-9. doi: 10.1136/bjophthalmol-2012-302668. Epub 2013 Apr 12
[PubMed PMID: 23584719]
[58]
Maskin SL, Testa WR. Growth of meibomian gland tissue after intraductal meibomian gland probing in patients with obstructive meibomian gland dysfunction. The British journal of ophthalmology. 2018 Jan:102(1):59-68. doi: 10.1136/bjophthalmol-2016-310097. Epub 2017 Jun 7
[PubMed PMID: 28592418]
[59]
Jiang X, Deng A, Yang J, Bai H, Yang Z, Wu J, Lv H, Li X, Wen T. Pathogens in the Meibomian gland and conjunctival sac: microbiome of normal subjects and patients with Meibomian gland dysfunction. Infection and drug resistance. 2018:11():1729-1740. doi: 10.2147/IDR.S162135. Epub 2018 Oct 11
[PubMed PMID: 30349330]
[60]
Arita R, Fukuoka S. Non-pharmaceutical treatment options for meibomian gland dysfunction. Clinical & experimental optometry. 2020 Nov:103(6):742-755. doi: 10.1111/cxo.13035. Epub 2020 Jan 13
[PubMed PMID: 31943385]
[61]
Yin Y, Gong L. Reversibility of Gland Dropout and Significance of Eyelid Hygiene Treatment in Meibomian Gland Dysfunction. Cornea. 2017 Mar:36(3):332-337. doi: 10.1097/ICO.0000000000001042. Epub
[PubMed PMID: 27755193]
[62]
Mizoguchi S, Iwanishi H, Arita R, Shirai K, Sumioka T, Kokado M, Jester JV, Saika S. Ocular surface inflammation impairs structure and function of meibomian gland. Experimental eye research. 2017 Oct:163():78-84. doi: 10.1016/j.exer.2017.06.011. Epub
[PubMed PMID: 28950941]