Continuing Education Activity
With over 100 different species, the genus Mycoplasma is a unique bacterium that lacks a cell wall and causes a wide range of symptoms and infections. The subspecies mycoplasma pneumoniae is the most widely studied and although it is typically associated with "atypical pneumonia" it can also lead to infections of other anatomical sites such as skin, central nervous system, blood, heart, and joints. Mycoplasma genitalium, another Mycoplasma species is raising increasing concern as a cause of sexually transmitted infections. Ureaplasma species has been identified as a cause of urologic, gynecologic and obstetric morbidity with associated complications in men, women, and neonates. This activity describes the pathophysiology, evaluation, and management of common mycoplasma infections and highlights the role of the interprofessional team in caring for affected patients.
Objectives:
- Identify the epidemiology of mycoplasma infections.
- Summarize the organ systems in which various species of mycoplasma have caused infection.
- Describe the treatment and management options available for mycoplasma infections.
- Explain interprofessional team strategies for improving care coordination and communication to improve outcomes in patients with mycoplasma infections.
Introduction
Mycoplasma is a term used to refer to any of the members of the class Mollicutes which include Mycoplasma and Ureaplasma.[1] With over 100 different species, the genus Mycoplasma is a unique bacterium that lacks a cell wall and causes a wide range of symptoms and infections. This organism, first discovered in 1898, was known initially as a parasitic infection to animals and has become most commonly known in modern medicine for the subspecies, Mycoplasma pneumoniae. Mycoplasma pneumoniae commonly causes "atypical pneumonia" a name derived because of its lack of response to antibiotics. Mycoplasma pneumoniae is the most widely studied Mycoplasma species due to its increasing prevalence. Despite its name, M. pneumoniae has also correlate with infections in other anatomical sites such as skin, central nervous system, blood, heart, and joints. Mycoplasma genitalium, another Mycoplasma species is raising increasing concern as a cause of sexually transmitted infections. Ureaplasma species has been identified as a cause of urologic, gynecologic and obstetric morbidity with associated complications in men, women, and neonates. This article discusses the most common mycoplasmal infections and its impact on modern practice.
Etiology
Although there are rare instances of Mycoplasma transmission from animals to humans, Mycoplasma is predominantly transmitted by human to human contact. It primarily affects mucosal areas of the respiratory tract and urogenital tract. The organism Mycoplasma pneumoniae attaches to ciliated epithelium in the upper and lower respiratory tract via attachment by a specialized organelle.[2] Droplets containing the organism spread the infection from host to host. The mechanics of transmission to non-respiratory tissues is not well understood, but the direct invasion of tissue appears to be a significant contributor. Immunologic features of the mucosa and autoimmune triggers contribute as well.[3]
Numerous commensal mycoplasmas also inhabit the body, primarily the oropharynx, with most common being Mycoplasma salivarium and Mycoplasma orale. These species are not pathogenic except for immunosuppressed individuals in which the organism may become opportunistic.[4] Ureaplasma species, another sub-class of Mollicutes, are known to cause urethral, gynecologic and obstetric infections. Ureaplasma species, Mycoplasma genitalium, and M. hominis are genitourinary mucosal organisms and infection is spread through direct sexual contact.
Epidemiology
Documentation of Mycoplasma infections exists worldwide. Overall, there is no evidence to suggest a difference in susceptibilities in sexes regardless of subspecies. Mycoplasma pneumoniae is associated with up to 15-20% of cases of community-acquired pneumonia.[5] Spread is facilitated in "close quarters" like hospitals, military bases, and schools. Respiratory infections from M. pneumoniae, primarily occur in children ages 5 to 14 years old and young adults. Infants younger than 6 months are rarely infected; likely the result of residual maternal immunity. Carriers may not develop obvious symptoms, so the true prevalence of mycoplasmal infection is likely higher than reported.
Active M. pneumoniae infection is most distinguishable for its gradual onset of symptoms and incubation period ranging from 2 to 3 weeks. In the United States, the increased frequency of M. pneumoniae diagnoses in the summer to early fall months is likely because there is a lower incidence of other respiratory infections during these months. Epidemics often occur in 3 to 5-year intervals.
Ureaplasma species, Mycoplasma genitalium, and Mycoplasma hominis may be isolated from up to 80% of sexually active females, but these organisms do not universally correlate with active infection. Mycoplasma genitalium may cause up to 20% of the cases of nongonococcal urethritis, cervicitis, and post-pregnancy PID noted.[6] Prevalence of the carrier state is higher in lower socioeconomic groups and individuals with multiple partners.
Pathophysiology
Mycoplasma is an obligate parasite and primarily an extracellular pathogen that has developed a specialized organelle for attachment to host cells. The attachment of Mycoplasma to a host cell prevents ciliary clearance of the organism. Hydrogen peroxide and superoxide radicals produced by the Mycoplasma cause oxidative damage to the host cells. Cellular damage causes activation of the innate immune system triggering chemotaxis and cytotoxic effects of cytokine release. Mycoplasma can invade tissues directly and replicate intracellularly in other organs.[3] Autoimmune reactions also contribute to the extrapulmonary effects of mycoplasmal infection.
Histopathology
Overall, there are few distinguishing histologic characteristics of Mycoplasma. "In fatal cases, death is associated with diffuse pneumonia, adult respiratory distress syndrome, vascular thrombosis, disseminated intravascular coagulation, and multiorgan failure. Biopsy of the lung at autopsy shows bronchiolitis and focal sites of bronchiolitis obliterans with alveolar edema and intra-alveolar hemorrhages." Electron microscopy of lung tissue demonstrates Mycoplasma organisms localized on the surface of the bronchiolar epithelium. Lung biopsy also shows ulcerated mucosal surfaces, destroyed ciliated epithelium, extensive monocellular infiltration (macrophages, lymphocytes), and neutrophil infiltration.[3] Recent studies investigating the potential role of Mycoplasma pneumoniae in chronic lung disease such as asthma show cellular changes consistent with the chronic inflammatory changes described above.[3]
Toxicokinetics
Community-acquired distress syndrome (CARDS) toxin is an exotoxin identified as a significant virulence factor of M. pneumoniae. M. pneumoniae encodes transcription of the CARDS toxin-encoding gene upon exposure to host cells, and the resultant exotoxin plays an essential role in Mycoplasma host cell interactions. Exposure of animal models to the CARDS exotoxin reproduces the clinical features of mycoplasmal infection including increased cytokine production, eosinophilia, and hyperreactivity of airway resembling asthma.[3] Antigens to the CARDS exotoxin have been demonstrated in bronchial washings of humans recovering from mycoplasmal infection further implicating its role in human disease.
History and Physical
Mycoplasma pneumoniae has multiple different manifestations dependent on site of infections, but it most commonly correlates with respiratory tract infection. Clinical presentation can vary from mild to severe illness often noted in children ages 5 to 14 years old. Symptoms are often gradual in onset and nonspecific. Patients may present with fever, headache, sore throat, and cough. On examination, patients present with a sore throat that rarely has exudates or lymphadenopathy. Upon auscultation, the patient may have variable findings dependent on the extent of infection. Rales and rhonchi (if the involvement is merely tracheobronchitis) or dullness with crackles (if pneumonia has developed) may be present on auscultation. Often, patients have a long convalescence with a prolonged cough for up to 4 to 6 weeks. Mycoplasma pneumoniae may with mucocutaneous eruptions related to erythema multiforme and Steven Johnson syndrome. Urticaria and anaphylactoid purpura are rare findings.
Urogenital infections caused by Ureaplasma species, Mycoplasma genitalium, and M. hominis may present with nongonococcal urethritis, dysuria, urgency, and urethral discharge in men. Many infected women are asymptomatic but may experience pelvic pain if pelvic inflammatory disease develops.
Evaluation
The diagnosis of Mycoplasma infection is usually made clinically. Given the prolonged turnaround time, cost, and limited availability, clinical laboratory findings are rarely the primary diagnostic tools. One may use culture, serology, or PCR to confirm and support the diagnosis.
Culture is used to type organisms and have high specificity; however, the technique is time-consuming and rarely used to guide treatment. M. genitalium may take as much as 8 weeks to grow in culture. Serum antibody tests for Mycoplasma pneumoniae are most frequently used in the hospital setting because the turnaround time is short; IgM increases within 1 week of symptom onset and IgG increases 2 weeks later. One disadvantage to antibody tests is the slow decline which may take up to 1 year to resolve and lead to false-positive results. PCR testing of nasopharyngeal aspirates, urethral and vaginal swabs have the advantage of specificity; however, its disadvantages are price and delayed turnaround time.
Radiological imaging may be useful if atypical pneumonia is suspected, but radiological findings may be variable. The four most common findings seen on chest x-rays are peribronchial and perivascular interstitial infiltrates (49%), airspace consolidation (38%), reticulonodular opacification (8%), and nodular or mass- like opacification (5%).[7] In about 20% of cases, involvement is bilateral.
Treatment / Management
Because it lacks a cell wall, Mycoplasma is resistant to many common antibiotics; therefore, choices for antimicrobial therapy are limited. Macrolides, tetracyclines, and newer fluoroquinolones have been found to have the highest efficacy against mycoplasma infections. Potential toxicities in children limit treatment options to macrolides. Azithromycin and clarithromycin are most frequently used to treat atypical pneumonia because they are better tolerated than erythromycin. Treatment with Macrolides is typically up to a 5-day course.
Tetracyclines are preferable if there central nervous system involvement. Doxycycline is the first choice to treat M. hominis and U. urealyticum species. Fluoroquinolones are bactericidal and therefore useful for immunocompromised patients and unusual cases of systemic infection. Length of treatment will vary pending the patient's condition and illness.[8] Treatment of sexual partners is recommended in cases of genitourinary tract infection.
Differential Diagnosis
Due to its variable presentation, Mycoplasma often has a broad differential diagnosis.
Mycoplasma pneumoniae has most frequently presented as community-acquired pneumonia. Other diagnoses commonly confused with M. pneumoniae are:
- Typical bacterial pneumonia
- Viral pneumonia
- Tuberculosis
- Pulmonary embolism
- Empyema
- Lung abscess
Ureaplasma species, M. genitalium, and M. hominis species correlate more with genitourinary infections. Other differentials include:
- N. gonorrhea
- C. trachomatis
Prognosis
Considering that most cases of mycoplasma infections involve a single organ infection with few systemic symptoms, the overall prognosis is favorable with full recovery following treatment; however, re-infection may occur shortly after the completion of therapy. In rare instances, there have been fulminant or fatal cases of Mycoplasma pneumoniae often related to complications or severe systemic involvement leading to multi-organ failure. Fulminant cases of M. pneumoniae may account for 0.5 to 0.2% of all cases.[7]
Complications
Although rare, reports of fatalities because of M. pneumoniae infection exist. Up to about 25% of individuals who develop respiratory infections from M. pneumoniae experience extrapulmonary complications.[9] Autoimmune reactions and direct tissue invasion are suspected to be responsible for these complications. The most common complications are the invasion of the central nervous system. Neurologic symptoms may occur up to 1 to 2 weeks following the onset of respiratory symptoms. Up to 20% of patients have no sign of respiratory illness.[10] Patients may present with encephalitis (most common in children), aseptic meningitis, polyradiculitis, cranial nerve palsies, acute disseminated encephalomyelitis, optic neuritis, mental confusion, acute psychosis, Guillain-Barre Syndrome, brachial plexus neuropathy, ataxia, and choreoathetosis.
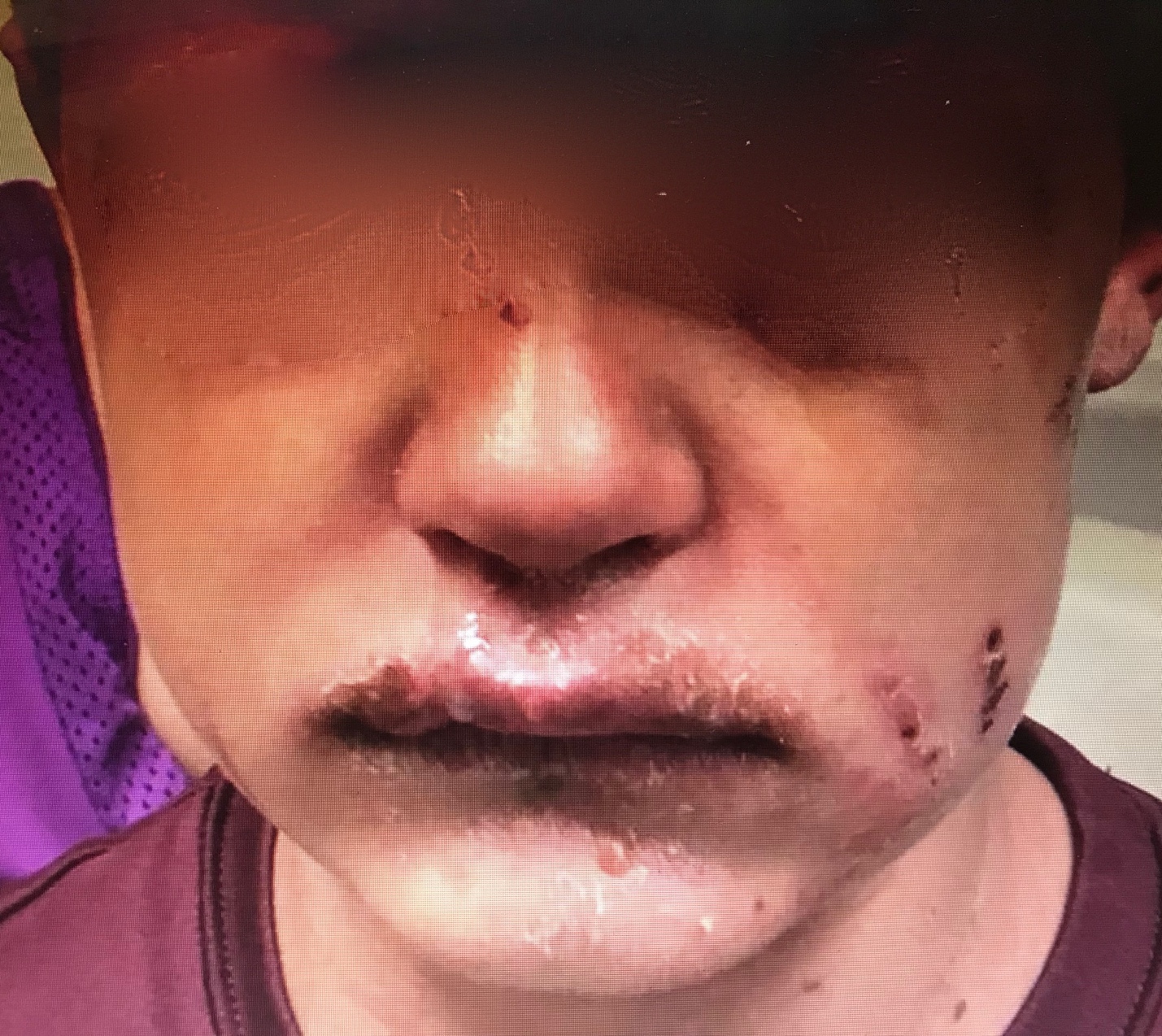
Dermatological complications (Steven-Johnson syndrome, ulcerative stomatitis, bullous exanthems, and erythema multiforme) occur in up to 25% of patients.[11]
Synovial fluid involvement causes septic arthritis, arthralgias, osteomyelitis, and poly arthropathies. Approximately 14% of patients infected with M. pneumoniae are affected.[12]
Cardiac complications (pericarditis, cardiac tamponade, myocarditis, myopericarditis, and endocarditis) are rare but documented in 1 to 8.5% of patients with serological evidence of infection.[13] Cardiac complications most often affect adults.
Hemolytic anemia is a complication seen in children infected by Mycoplasma pneumoniae more often than in adults. Cross-reacting cold agglutinins are believed to be the responsible mechanism. There are also reports of thrombotic thrombocytopenia purpura and disseminated intravascular coagulation. If one considers subclinical forms of these hematological disorders, they may affect 50% of patients.
Other complications of M. pneumoniae infection involve the GI tract (nausea, vomiting, hepatitis, pancreatitis), the kidneys (acute glomerulonephritis, renal failure), ears (otitis externa, otitis media, myringitis), and eyes (conjunctivitis, iritis, optic disk swelling, retinitis).
Ureaplasma and M. genitalium have links to pregnancy complications and neonatal risk. Recent studies correlate Ureaplasma parvum infection with an increased risk for preterm birth, low weight infants, and bronchopulmonary disease in preterm neonates. M. hominis correlates with mid-trimester abortions and early miscarriages. As a cause for sexually transmitted infections, M. genitalium correlates with pelvic inflammatory disease and chorioamnionitis.[14]
Deterrence and Patient Education
Exposure commonly occurs in close quarter areas such as military camps, schools, and hospitals. Hand hygiene is vital to prevent the spread of infection. Because mycoplasmal infection is spread by via aerosolization, droplet precautions are essential to prevent spread among hospitalized patients.
Regarding individuals affected by Ureaplasma species, M. genitalium, and M. hominis are recommended to use condoms to prevent the spread of the STI. Sexual partners should also receive treatment to prevent reinfection.
Pearls and Other Issues
- Mycoplasma is a diverse bacterium that is not only limited to respiratory infections but may cause an array of different symptoms clinicians must consider in their differential.
- Mycoplasma species have newly arisen as a sexually transmitted infection that if left untreated may lead to complications in pregnant women.
- The primary treatment for mycoplasmal infections include macrolides, tetracyclines, and fluoroquinolones. Macrolides provide the only choice for children because of potential toxic profiles of tetracyclines and fluoroquinolones.
- Although rare, serious and potentially fatal complications may arise from Mycoplasma if disseminated to other organs including the central nervous system and heart.
Enhancing Healthcare Team Outcomes
Interpersonal communication is essential in the hospital setting to prevent the spread of the infection to other patients. All members of the healthcare team bear a responsibility to avoid the spread of infection to non-infected patients. Nurses closely monitor the patient's clinical condition and help to establish and maintain droplet precautions during the patient's stay. All members of the healthcare team should promote adherence to droplet precautions by wearing masks and hand hygiene before and after each visit. Pharmacists and physicians will work to optimize antimicrobial therapy.