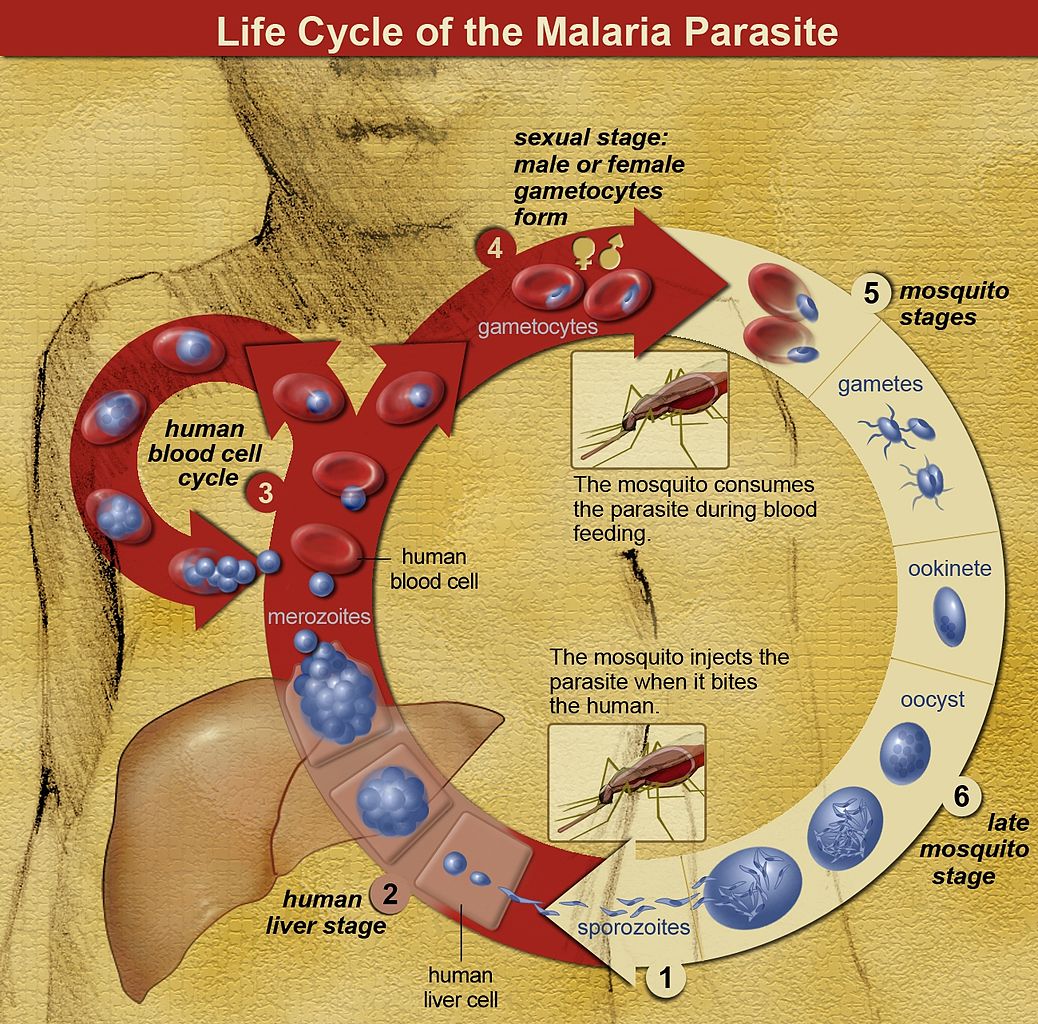
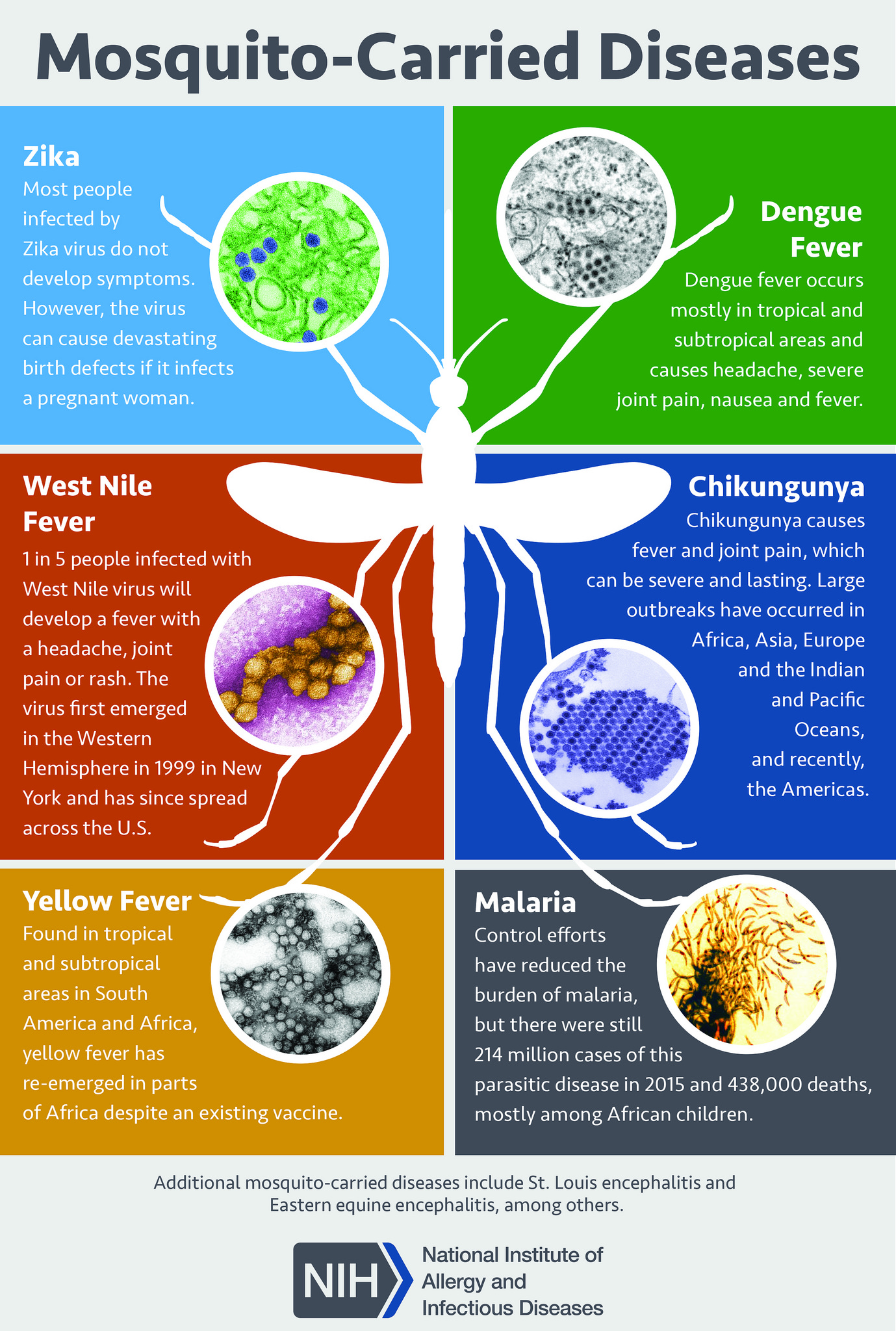
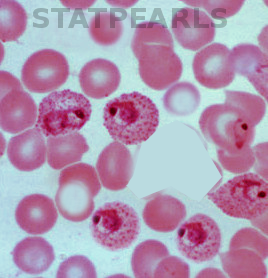
[1]
Howes RE, Battle KE, Mendis KN, Smith DL, Cibulskis RE, Baird JK, Hay SI. Global Epidemiology of Plasmodium vivax. The American journal of tropical medicine and hygiene. 2016 Dec 28:95(6 Suppl):15-34. doi: 10.4269/ajtmh.16-0141. Epub 2016 Jul 11
[PubMed PMID: 27402513]
[2]
Gunalan K, Lo E, Hostetler JB, Yewhalaw D, Mu J, Neafsey DE, Yan G, Miller LH. Role of Plasmodium vivax Duffy-binding protein 1 in invasion of Duffy-null Africans. Proceedings of the National Academy of Sciences of the United States of America. 2016 May 31:113(22):6271-6. doi: 10.1073/pnas.1606113113. Epub 2016 May 17
[PubMed PMID: 27190089]
[3]
Gunalan K, Niangaly A, Thera MA, Doumbo OK, Miller LH. Plasmodium vivax Infections of Duffy-Negative Erythrocytes: Historically Undetected or a Recent Adaptation? Trends in parasitology. 2018 May:34(5):420-429. doi: 10.1016/j.pt.2018.02.006. Epub 2018 Mar 9
[PubMed PMID: 29530446]
[4]
Dayananda KK, Achur RN, Gowda DC. Epidemiology, drug resistance, and pathophysiology of Plasmodium vivax malaria. Journal of vector borne diseases. 2018 Jan-Mar:55(1):1-8. doi: 10.4103/0972-9062.234620. Epub
[PubMed PMID: 29916441]
[5]
Newman RD, Parise ME, Barber AM, Steketee RW. Malaria-related deaths among U.S. travelers, 1963-2001. Annals of internal medicine. 2004 Oct 5:141(7):547-55
[PubMed PMID: 15466772]
[6]
Hemmer CJ, Holst FG, Kern P, Chiwakata CB, Dietrich M, Reisinger EC. Stronger host response per parasitized erythrocyte in Plasmodium vivax or ovale than in Plasmodium falciparum malaria. Tropical medicine & international health : TM & IH. 2006 Jun:11(6):817-23
[PubMed PMID: 16772003]
[7]
Baird JK. Evidence and implications of mortality associated with acute Plasmodium vivax malaria. Clinical microbiology reviews. 2013 Jan:26(1):36-57. doi: 10.1128/CMR.00074-12. Epub
[PubMed PMID: 23297258]
[8]
Price RN, Tjitra E, Guerra CA, Yeung S, White NJ, Anstey NM. Vivax malaria: neglected and not benign. The American journal of tropical medicine and hygiene. 2007 Dec:77(6 Suppl):79-87
[PubMed PMID: 18165478]
[9]
Kochar DK, Saxena V, Singh N, Kochar SK, Kumar SV, Das A. Plasmodium vivax malaria. Emerging infectious diseases. 2005 Jan:11(1):132-4
[PubMed PMID: 15705338]
[10]
Val F, Machado K, Barbosa L, Salinas JL, Siqueira AM, Costa Alecrim MG, Portillo HD, Bassat Q, Monteiro WM, Guimarães Lacerda MV. Respiratory Complications of Plasmodium vivax Malaria: Systematic Review and Meta-Analysis. The American journal of tropical medicine and hygiene. 2017 Sep:97(3):733-743. doi: 10.4269/ajtmh.17-0131. Epub 2017 Jul 19
[PubMed PMID: 28722625]
Level 1 (high-level) evidence
[11]
Boyce MR, O'Meara WP. Use of malaria RDTs in various health contexts across sub-Saharan Africa: a systematic review. BMC public health. 2017 May 18:17(1):470. doi: 10.1186/s12889-017-4398-1. Epub 2017 May 18
[PubMed PMID: 28521798]
Level 1 (high-level) evidence
[12]
Abba K, Kirkham AJ, Olliaro PL, Deeks JJ, Donegan S, Garner P, Takwoingi Y. Rapid diagnostic tests for diagnosing uncomplicated non-falciparum or Plasmodium vivax malaria in endemic countries. The Cochrane database of systematic reviews. 2014 Dec 18:2014(12):CD011431. doi: 10.1002/14651858.CD011431. Epub 2014 Dec 18
[PubMed PMID: 25519857]
Level 1 (high-level) evidence
[13]
Haditsch M. Quality and reliability of current malaria diagnostic methods. Travel medicine and infectious disease. 2004 Aug-Nov:2(3-4):149-60
[PubMed PMID: 17291976]
Level 2 (mid-level) evidence
[14]
Maltha J, Gillet P, Jacobs J. Malaria rapid diagnostic tests in endemic settings. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2013 May:19(5):399-407. doi: 10.1111/1469-0691.12151. Epub 2013 Feb 25
[PubMed PMID: 23438048]
[15]
Pattanasin S, Proux S, Chompasuk D, Luwiradaj K, Jacquier P, Looareesuwan S, Nosten F. Evaluation of a new Plasmodium lactate dehydrogenase assay (OptiMAL-IT) for the detection of malaria. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2003 Nov-Dec:97(6):672-4
[PubMed PMID: 16117960]
[16]
Tjitra E, Suprianto S, Dyer M, Currie BJ, Anstey NM. Field evaluation of the ICT malaria P.f/P.v immunochromatographic test for detection of Plasmodium falciparum and Plasmodium vivax in patients with a presumptive clinical diagnosis of malaria in eastern Indonesia. Journal of clinical microbiology. 1999 Aug:37(8):2412-7
[PubMed PMID: 10405377]
[17]
Fransisca L, Kusnanto JH, Satoto TB, Sebayang B, Supriyanto, Andriyan E, Bangs MJ. Comparison of rapid diagnostic test Plasmotec Malaria-3, microscopy, and quantitative real-time PCR for diagnoses of Plasmodium falciparum and Plasmodium vivax infections in Mimika Regency, Papua, Indonesia. Malaria journal. 2015 Mar 5:14():103. doi: 10.1186/s12936-015-0615-5. Epub 2015 Mar 5
[PubMed PMID: 25890368]
[18]
Dalrymple U, Arambepola R, Gething PW, Cameron E. How long do rapid diagnostic tests remain positive after anti-malarial treatment? Malaria journal. 2018 Jun 8:17(1):228. doi: 10.1186/s12936-018-2371-9. Epub 2018 Jun 8
[PubMed PMID: 29884184]
[19]
Recht J, Ashley EA, White NJ. Use of primaquine and glucose-6-phosphate dehydrogenase deficiency testing: Divergent policies and practices in malaria endemic countries. PLoS neglected tropical diseases. 2018 Apr:12(4):e0006230. doi: 10.1371/journal.pntd.0006230. Epub 2018 Apr 19
[PubMed PMID: 29672516]
[20]
Baird JK, Hoffman SL. Primaquine therapy for malaria. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2004 Nov 1:39(9):1336-45
[PubMed PMID: 15494911]
[21]
Kovacs SD, Rijken MJ, Stergachis A. Treating severe malaria in pregnancy: a review of the evidence. Drug safety. 2015 Feb:38(2):165-81. doi: 10.1007/s40264-014-0261-9. Epub
[PubMed PMID: 25556421]
[22]
Dondorp A, Nosten F, Stepniewska K, Day N, White N, South East Asian Quinine Artesunate Malaria Trial (SEAQUAMAT) group. Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet (London, England). 2005 Aug 27-Sep 2:366(9487):717-25
[PubMed PMID: 16125588]
Level 1 (high-level) evidence
[23]
Jones KL, Donegan S, Lalloo DG. Artesunate versus quinine for treating severe malaria. The Cochrane database of systematic reviews. 2007 Oct 17:(4):CD005967
[PubMed PMID: 17943870]
Level 1 (high-level) evidence
[24]
Hess KM, Goad JA, Arguin PM. Intravenous artesunate for the treatment of severe malaria. The Annals of pharmacotherapy. 2010 Jul-Aug:44(7-8):1250-8. doi: 10.1345/aph.1M732. Epub 2010 Jun 15
[PubMed PMID: 20551300]
[26]
Goodyer LI, Croft AM, Frances SP, Hill N, Moore SJ, Onyango SP, Debboun M. Expert review of the evidence base for arthropod bite avoidance. Journal of travel medicine. 2010 May-Jun:17(3):182-92. doi: 10.1111/j.1708-8305.2010.00402.x. Epub
[PubMed PMID: 20536888]
[27]
Pryce J, Richardson M, Lengeler C. Insecticide-treated nets for preventing malaria. The Cochrane database of systematic reviews. 2018 Nov 6:11(11):CD000363. doi: 10.1002/14651858.CD000363.pub3. Epub 2018 Nov 6
[PubMed PMID: 30398672]
Level 1 (high-level) evidence