[1]
Mazzone SB, Undem BJ. Vagal Afferent Innervation of the Airways in Health and Disease. Physiological reviews. 2016 Jul:96(3):975-1024. doi: 10.1152/physrev.00039.2015. Epub
[PubMed PMID: 27279650]
[2]
Williams EK, Chang RB, Strochlic DE, Umans BD, Lowell BB, Liberles SD. Sensory Neurons that Detect Stretch and Nutrients in the Digestive System. Cell. 2016 Jun 30:166(1):209-21. doi: 10.1016/j.cell.2016.05.011. Epub 2016 May 26
[PubMed PMID: 27238020]
[3]
de Moraes ER, Kushmerick C, Naves LA. Morphological and functional diversity of first-order somatosensory neurons. Biophysical reviews. 2017 Oct:9(5):847-856. doi: 10.1007/s12551-017-0321-3. Epub 2017 Sep 9
[PubMed PMID: 28889335]
[4]
Connor M, Naves LA, McCleskey EW. Contrasting phenotypes of putative proprioceptive and nociceptive trigeminal neurons innervating jaw muscle in rat. Molecular pain. 2005 Oct 24:1():31
[PubMed PMID: 16242047]
[5]
Jessen KR, Mirsky R, Lloyd AC. Schwann Cells: Development and Role in Nerve Repair. Cold Spring Harbor perspectives in biology. 2015 May 8:7(7):a020487. doi: 10.1101/cshperspect.a020487. Epub 2015 May 8
[PubMed PMID: 25957303]
Level 3 (low-level) evidence
[6]
Salzer JL. Schwann cell myelination. Cold Spring Harbor perspectives in biology. 2015 Jun 8:7(8):a020529. doi: 10.1101/cshperspect.a020529. Epub 2015 Jun 8
[PubMed PMID: 26054742]
Level 3 (low-level) evidence
[8]
Dubin AE, Patapoutian A. Nociceptors: the sensors of the pain pathway. The Journal of clinical investigation. 2010 Nov:120(11):3760-72. doi: 10.1172/JCI42843. Epub 2010 Nov 1
[PubMed PMID: 21041958]
[9]
López-Barneo J, Ortega-Sáenz P, González-Rodríguez P, Fernández-Agüera MC, Macías D, Pardal R, Gao L. Oxygen-sensing by arterial chemoreceptors: Mechanisms and medical translation. Molecular aspects of medicine. 2016 Feb-Mar:47-48():90-108. doi: 10.1016/j.mam.2015.12.002. Epub 2015 Dec 18
[PubMed PMID: 26709054]
[10]
Nattie E, Li A. Central chemoreceptors: locations and functions. Comprehensive Physiology. 2012 Jan:2(1):221-54. doi: 10.1002/cphy.c100083. Epub
[PubMed PMID: 23728974]
[11]
Butler SJ, Bronner ME. From classical to current: analyzing peripheral nervous system and spinal cord lineage and fate. Developmental biology. 2015 Feb 15:398(2):135-46. doi: 10.1016/j.ydbio.2014.09.033. Epub 2014 Oct 24
[PubMed PMID: 25446276]
[12]
Newbern JM. Molecular control of the neural crest and peripheral nervous system development. Current topics in developmental biology. 2015:111():201-31. doi: 10.1016/bs.ctdb.2014.11.007. Epub 2015 Jan 22
[PubMed PMID: 25662262]
[13]
Zochodne DW. Local blood flow in peripheral nerves and their ganglia: Resurrecting key ideas around its measurement and significance. Muscle & nerve. 2018 Jun:57(6):884-895. doi: 10.1002/mus.26031. Epub 2018 Jan 11
[PubMed PMID: 29211922]
[14]
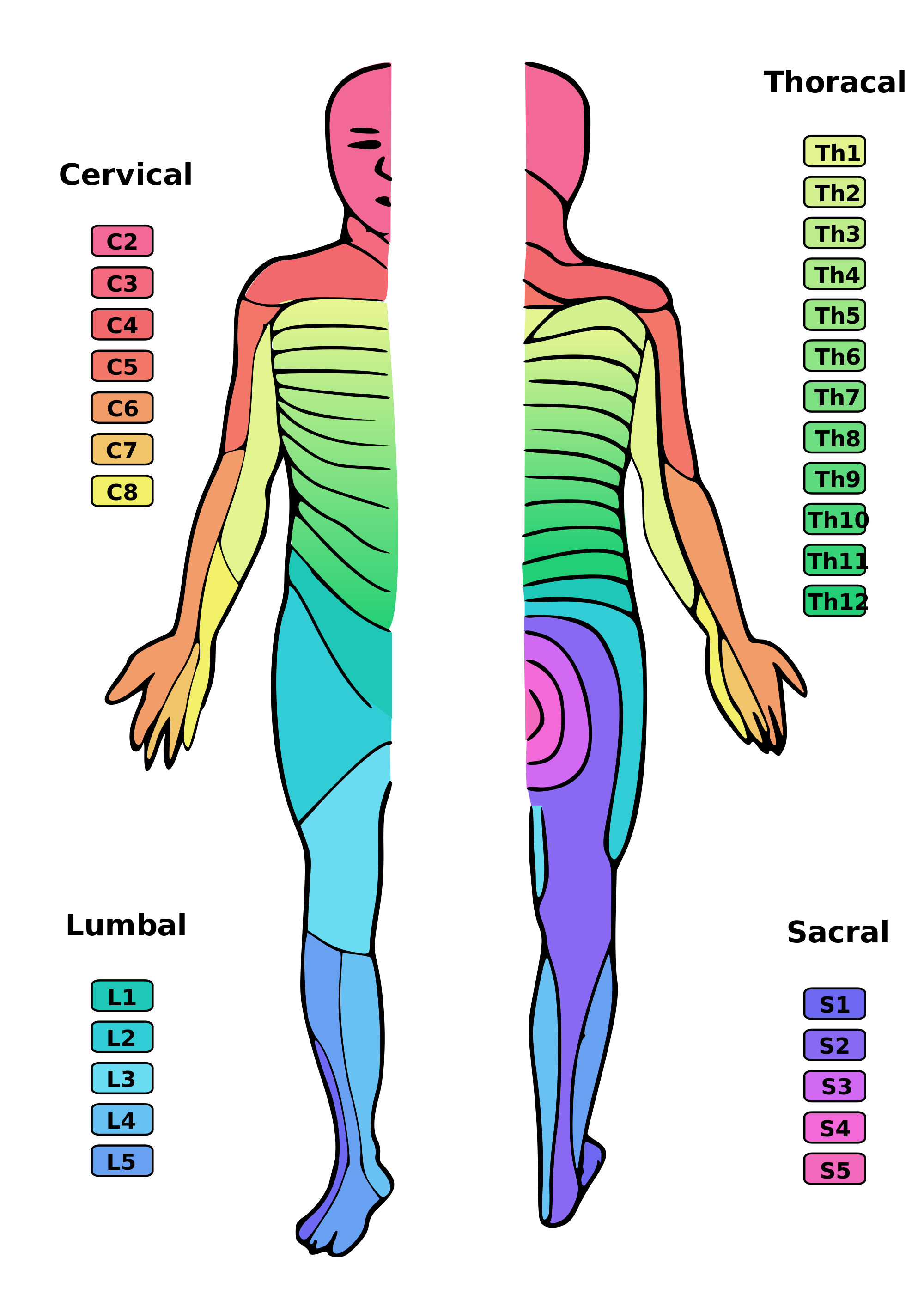
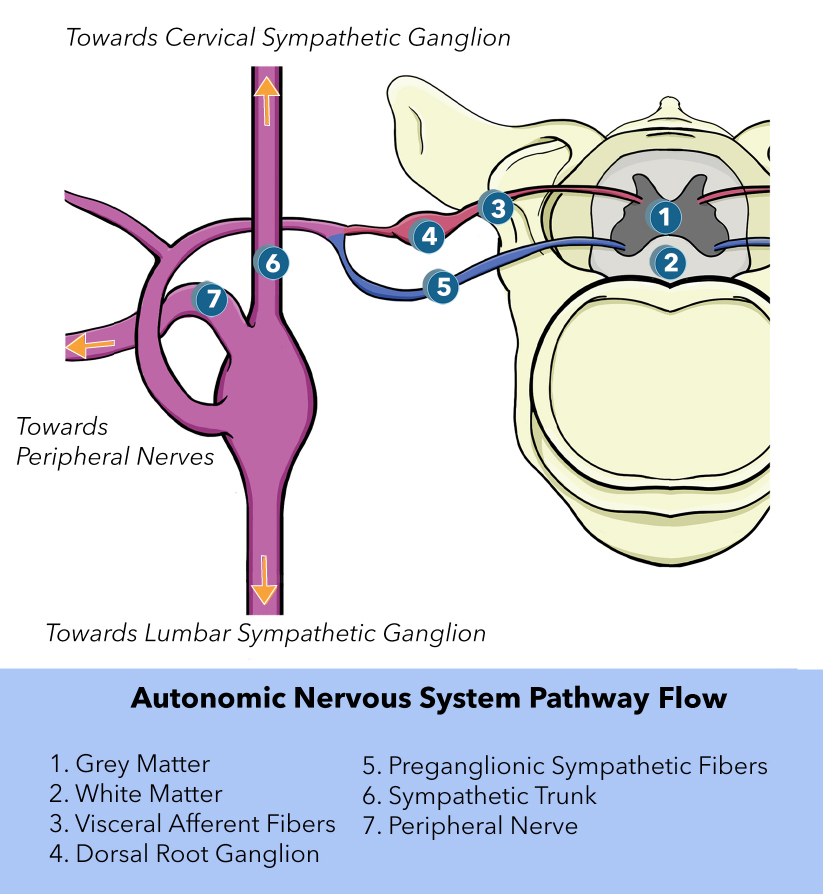
Jänig W. Neurobiology of visceral afferent neurons: neuroanatomy, functions, organ regulations and sensations. Biological psychology. 1996 Jan 5:42(1-2):29-51
[PubMed PMID: 8770369]
[16]
Thomas K, Minutello K, M Das J. Neuroanatomy, Cranial Nerve 9 (Glossopharyngeal). StatPearls. 2023 Jan:():
[PubMed PMID: 30969699]
[17]
Breit S, Kupferberg A, Rogler G, Hasler G. Vagus Nerve as Modulator of the Brain-Gut Axis in Psychiatric and Inflammatory Disorders. Frontiers in psychiatry. 2018:9():44. doi: 10.3389/fpsyt.2018.00044. Epub 2018 Mar 13
[PubMed PMID: 29593576]
[19]
Jeon SK, Paik DJ, Hwang YI. Variations in sural nerve formation pattern and distribution on the dorsum of the foot. Clinical anatomy (New York, N.Y.). 2017 May:30(4):525-532. doi: 10.1002/ca.22873. Epub 2017 Apr 8
[PubMed PMID: 28281304]
[20]
McConnell OJ, Longley R, Gunasekera M. Isometachromin, a new cytotoxic sesquiterpenoid from a deep water sponge of the family Spongiidae. Experientia. 1992 Sep 15:48(9):891-2
[PubMed PMID: 1397187]
[21]
Timmers HJ, Wieling W, Karemaker JM, Lenders JW. Denervation of carotid baro- and chemoreceptors in humans. The Journal of physiology. 2003 Nov 15:553(Pt 1):3-11
[PubMed PMID: 14528027]
[22]
Rathmann W, Enck P, Frieling T, Gries FA. Visceral afferent neuropathy in diabetic gastroparesis. Diabetes care. 1991 Nov:14(11):1086-9
[PubMed PMID: 1797493]
[23]
Foreman RD, Garrett KM, Blair RW. Mechanisms of cardiac pain. Comprehensive Physiology. 2015 Apr:5(2):929-60. doi: 10.1002/cphy.c140032. Epub
[PubMed PMID: 25880519]
[24]
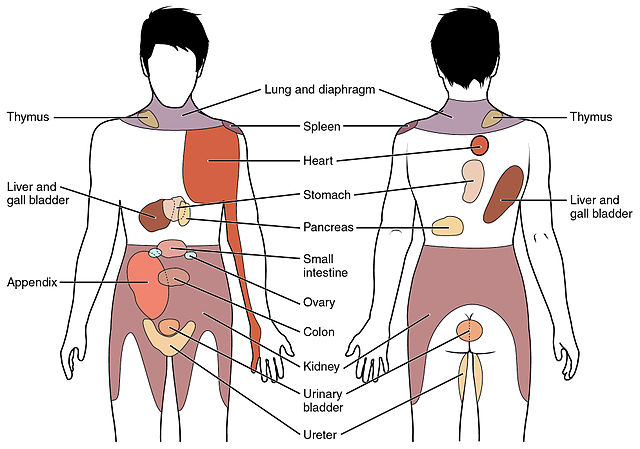
Murray GM. Guest Editorial: referred pain. Journal of applied oral science : revista FOB. 2009 Nov-Dec:17(6):i
[PubMed PMID: 20027423]
Level 3 (low-level) evidence