Introduction
Basivertebral nerve (BVN) ablation is a minimally invasive spinal procedure targeting the BVN, which is responsible for carrying nociceptive information from damaged vertebral endplates, an entity recently postulated as a source of chronic axial low back pain (LBP).[1][2][3][4]
In the past, other structures were considered as sole contributors to the etiology of chronic axial LBP, such as intervertebral discs (IVD), zygapophyseal facet joints, ligaments, sacroiliac joints, muscles, etc.[5][6][7] However, the recent understanding that vertebral endplates are particularly susceptible to inflammatory changes, fissuring, post-traumatic degeneration, and intraosseous edema due to their highly vascularized and innervated terminals from the basivertebral nerve and venous plexus suggests that these structures are particularly likely to contribute to LBP symptomatology, in addition to other structures.[8][9][10][11]
Finding the source of chronic axial LBP is clinically challenging, and often 80% of diagnoses are described as non-specific LBP, and in only 20% of cases, an anatomical source can be attributed.[12] Perhaps it is due to this variability and uncertainty that many interventions for the treatment of chronic axial LBP directly targeting anatomical structures, such as IVD, muscles, facet joints, and ligaments, have limited success rates and variable outcomes in the general population.
Several studies have reported a high incidence of vertebral endplate damage in up to 43% of subjects suffering from chronic axial LBP symptoms. These tend to manifest differently than when the etiology is from other structures.[7][8]
Often, vertebral endplate pain patients tend to present with significant functional impairment and debilitating pain while seated, standing, or during spinal flexion (in contrast to extension), with the pain reported as a burning, deep and achy, located in the midline region of the lumbar spine without radicular symptoms, and without motor weakness, numbness or tingling.[4][7][10] In fact, vertebral pain from damaged endplates tends to present clinically different than non-specific etiologies with reported greater frequency and longer duration of painful episodes and worse outcomes with conservative treatment and surgery.[11][10][13][14]
Treatment options for chronic axial LBP from damaged vertebral endplates start with conservative care, similar to other treatment algorithms, such as oral analgesics, opioids, and therapeutic exercises. However, conservative methods tend to be ineffective, and the identification and diagnosis with history and physical exam of this subset of patients with pathoanatomical vertebral endplate damage on diagnostic imaging are crucial to optimize outcomes and offer an effective treatment option, such as the BVN ablation.[8][15]
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
Chronic axial LBP remains a global healthcare problem affecting 30 million people in the United States and costing the healthcare system 90 billion dollars yearly.[16][17] With such a high financial burden, clinicians and researchers need to understand pathoanatomical considerations and the phenotypical profile in chronic axial LBP associated with vertebral endplate damage to target this source and optimize treatment options and success rates.
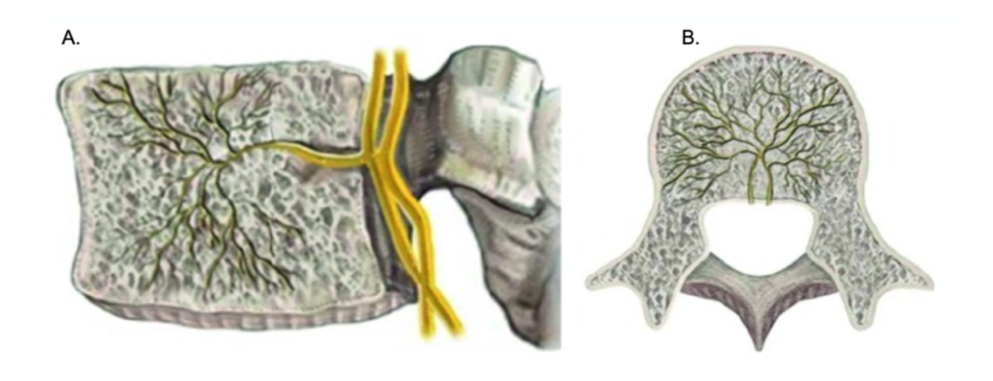
The sinuvertebral nerve arises from the ventral ramus of the spinal nerves bilaterally and takes a recurrent course to enter the spinal canal traveling towards the posterior aspect of the vertebra and entering the vertebral body through the basivertebral foramen. This foramen is located midline in the posterior vertebral body and is the entry point for the neurovascular bundle consistent with the BVN and the basivertebral vascular plexus. Although there is anatomical variability within its course, the nerve travels anteriorly into the vertebral body about 30% to 50%, where it forms a trunk cluster of fibers that migrate cranially and caudally towards the vertebral endplates responsible for supplying nociceptive input from damaged vertebral endplates. This branching point is the anatomical site (Figure 1) targeted for the ablative procedure.[2][3][18]
Vertebral endplates are the superior and inferior edge of the vertebral body, and these structures are highly susceptible to injury, post-traumatic degeneration, fissuring, intraosseous edema, and inflammation, which are visible phenotypic markers of vertebral pain noted on magnetic resonance images (MRI) classified as vertebral endplate Modic changes.[5][19][20][21]
There are three types of Modic changes (MCs), type 1, type 2, and type 3 and these differ based on MRI findings (Figure 2). Type 1 MCs represent vertebral endplate disruption, fissuring, degeneration, active inflammatory vertebral endplate changes and manifest as hypodense or decreased signal intensity of fibrovascular intraosseous bone marrow edema on T1-weighted MRI sequence and as hyperintense or increase signal intensity on T2-weighted MRI sequence. Type 2 MCs represent fatty bone marrow infiltration/replacement of subacute or chronic nature and show an increased signal intensity in both T1 and T2 MRI sequence images. Type 3 MCs are described as decreased intensity in both T1 and T2 MRI sequences.[14][20][22][23]
Although MCs are a radiological finding on MRI, the association of these findings in addition to a history and exam compatible with axial LBP symptoms has been reported by numerous clinical and basic science studies suggesting a positive association for a specific vertebral etiology (vetebrogenic pain). Nociceptive input from these damaged vertebral endplates carried by the BVN is directly related to inflammatory cytokines, substance P, and calcitonin gene-related peptide (CGRP), histologically confirmed with protein gene product (PGP) 9.5 positive staining under microscopy supporting this hypothesis. In particular, type 1 MCs have been reported to have a stronger direct association with more debilitating, severe LBP of longer duration, greater frequency, and worse functional impairments than those subjects with chronic axial LBP but without MCs, hence the great need to focus on a treatment intervention directly targeting this pain generator, such as ablation of the BVN.[4][6][9][20][22][23]
Indications
Clinical studies published to date on BVN ablation utilized a similar patient population, including patients with chronic axial LBP greater than 6 months of duration that has been refractory to non-operative treatment for at least 6 months, in addition to the confirmed presence of MCs type 1 or type 2 on MRI in at least one vertebral endplate, at one or more levels from the L3 to S1 region. Some studies excluded subjects with a history of spinal surgery, spinal stenosis, and use of opioids, while other studies included these, allowing for a more generalizable patient population more similar to daily clinical practice.[24][25][26]
Additional requirements when selecting patients for BVN ablation include documented evidence of significant impairments in function, debilitating pain unresponsive to conservative care, confirmed skeletal maturity on diagnostic imaging, and a history and physical exam supporting other potential primary sources of pain have been ruled out, and vertebral pain is likely. The Food and Drug Administration (FDA) cleared the procedure in 2016 for patients that meet the above criteria.[27][28][29][30]
Contraindications
Contraindications to BVN ablation are generally similar to other interventional spine procedures, such as systemic infections, spinal infections, pregnancy, incomplete skeletal maturity, implantable pulse generators (pacemakers, defibrillators), patients with severe cardiopulmonary compromise, patients where the targeted ablation zone is < 10 mm away from a sensitive structure not intended to be ablated, including the vertebral foramen (spinal canal), and in situations where unintended tissue damage may result based on the clinical assessment of the physician such as spine surgery at the treatment level where existing hardware is within the zone of the BVN ablation.[31][27][24][25]
Additional considerations reported in the literature as an exclusion among published studies included morbid obesity (due to potential tool length limitations in accessing target with elevated levels of adipose tissue in the lumbar region), patients where symptomatic spinal stenosis or radicular pain is the primary pain source, and situations where injury may result from the procedure based on physician assessment such as osteoporosis (particularly in patients with a prior vertebral compression fracture and/or who are actively taking hormonal therapy), in patients with metastatic disease or local malignancy, and in patients with risk for bleeding such as those diagnosed with thrombocytopenia or coagulopathy disorders.[26][28][29][30][32][33][34][35]
Equipment
Safe and accurate targeting of the BVN is critical to the success of the ablation procedure. The procedure is performed by a pain physician, spine surgeon, or interventional radiologist with experience in image-guided spinal procedures, preferably with C-arm fluoroscopy or computerized tomography (CT) guidance. A combination of anterior-posterior (AP), lateral, and oblique C-arm fluoroscopic views are utilized to localize the pedicle and to guide the positioning of instruments. A single C-arm is most often used. However, two C-arms may be used, one for lateral and one for AP and oblique images.
In addition to a CT or fluoroscopy imaging, equipment for BVN ablation includes: an introducer diamond or bevel tipped trocar to access the vertebral body through a transpedicular approach; a curved cannula assembly with accompanying straight stylet to create a channel in the vertebral body to the BVN terminus (target site), and a bipolar radiofrequency ablation probe and radiofrequency generator to create the ablative lesion in the nerve terminus (see technique section below for details). Additional supplies include standardized surgical sterile supplies, such as sterile gowns, gloves, sterile hats, shoe covers, masks, appropriate drapes, sponges, laps, and other supplies to minimize risks of infection.
Personnel
An experienced clinician must perform the procedure. In addition to the clinician performing the procedure, intraoperative personnel includes a fluoroscopy technologist (radiology technician), scrub technician, circulating nurse, and an anesthesiologist or nurse anesthetist for appropriate sedation to keep the patient comfortable safe, and monitored.
Preparation
In addition to a detailed medical history and physical examination to support the etiology of vertebral pain as the source of the patient's chronic axial LBP, the physician must document radiological evidence of type 1 or type 2 Modic changes on MRI and complete treatment history to support the rationale behind the procedure, including longstanding debilitating LBP with functional impairments greater than 6 months unresponsive to at least 6 months of conservative care.
The BVN ablation procedure utilizes a similar transpedicular approach to the vertebral augmentation procedure. Although there is no consensus on perioperative management before BVN ablation, it is reasonable to follow a similar standard of care to other percutaneous spinal interventions. In preparation for the procedure, the following should be obtained: complete metabolic profile (CMP), complete blood count (CBC), and a coagulative profile to assess bleeding risk and rule out underlying infection, severe thrombocytopenia, anemia, or metabolic disorders.
It is also highly recommended to follow guidelines on blood transfusion, discontinuation of anticoagulation, which are variable based on the anticoagulant in question, and perioperative intravenous (IV) antibiotics, such as 2 grams of cefazolin (or 600 mg clindamycin if penicillin allergy) 60 minutes pre-procedure, as a prophylactic measure standard of care to interventional spinal procedures. Based on the patient's comorbidities and physician's judgment, the procedure may be performed under general and monitored anesthesia care (MAC) sedation.
Technique or Treatment
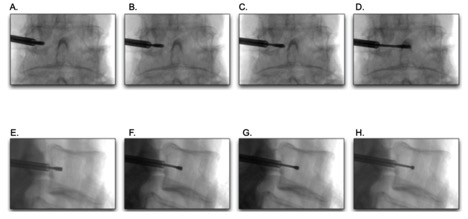
BVN ablation is performed in an outpatient setting by properly trained physicians and surgeons. This procedure has technical similarities with vertebra augmentation as it uses a transpedicular approach, although an extrapedicular approach has been described, and with lumbar radiofrequency ablation, as it uses an ablative lesion to the BVN to interrupt nociceptive signaling from damaged vertebral endplates. The first step in this procedure is to position the patient in a prone position under general anesthesia or MAC with continuous cardiac monitoring, pulse oximetry, and blood pressure. Second, utilizing a standardized sterile technique, the patient is prepped, and the target level and location of entry are marked and confirmed with C-arm fluoroscopy or CT guidance. Obtaining perfect image guidance is essential for this procedure, so the first step should be to rotate the C-arm to obtain a true anterior-posterior (AP) view of the pedicles at the level of entry to optimize a transpedicular approach.
The skin entry point and angle are marked with a sterile marker, and the skin is anesthetized with 1% lidocaine anesthetic, and a small incision is made with a scalpel. Next, a 22-gauge spinal needle may be used at the target entry position to anesthetize the pathway towards the periosteum. An introducer trocar is placed along the same trajectory and advanced through the pedicle, starting at the superior lateral aspect until it passes the posterior vertebral body wall. It is important to maintain a trajectory superior to the inferior cortex and lateral to the medial cortex of the pedicle to prevent trocar entry into the spinal canal or near neural elements. As the trocar is slowly advanced using a mallet, multiple AP and lateral C-arm views are obtained to ensure a safe trajectory.
Once the vertebra body is breached, the trocar is removed from the introducer cannula, and a curved cannula assembly (curved cannula and curved stylet) is that facilitates the creation of a curved channel toward the target site at the BVN terminus found midline, approximately 30% to 50% across the vertebral body width from posterior to anterior (Figure 3). Once the target site is confirmed with AP and lateral C-arm views, the curved stylet is removed, and a bipolar radiofrequency probe is inserted. The probe is connected to a radiofrequency generator, and radiofrequency energy is used to create approximately a 1 cm spherical lesion (85 degrees C for 15 minutes) at the BVN terminus. Upon completion, the radiofrequency probe and the introducer cannula are removed, and the subcutaneous tissue and skin are closed in a standard sterile fashion with a pressure dressing and skin glue or steri-strips. Sutures or staples are usually not necessary.
Patients are transferred to a post-anesthesia care unit (PACU) for monitoring. Vital signs and neurological function are reassessed after the procedure. Patients should be discharged the same day. Post-discharge, patients should be instructed to monitor the surgical site and be educated on signs of infection, activity restrictions (no lifting greater than 15 lbs, twisting, bending of the spine), and to not submerge themselves in water for at least 48 hours after the procedure. Follow-up post-procedure visits and full return to activity are based on each clinician's discretion.
Complications
The procedure is considered generally safe, with a very low rate of adverse events (AEs) reported among all clinical studies, totaling 473 procedures. Temporary exacerbation in LBP symptoms and incisional pain were the most commonly reported self-limiting AEs post-procedure. Additional minor AEs reported include transient radiculitis, which resolved after oral medication, and rare instances of non-permanent lumbar/sacral radiculopathy, nerve root injury, and motor/sensory deficits.
Serious AEs reported in the 473 clinical procedures included one case of a vertebral compression fracture in a sham crossover-procedure patient taking hormonal therapy, and in commercially treated patients, there have been two serious adverse events that were device-procedure related, one case of retroperitoneal hemorrhage, and one case of vertebral compression fracture. There have been no reports of thermal injuries, spinal cord injury, avascular necrosis, and post-procedure infections were documented. Among the AEs reported in the clinical studies that were device-procedure related, the median time to resolution was 66.5 days postoperatively.[32][33][34][35]
Clinical Significance
The cumulative data among multiple studies on BVN ablation demonstrated consistent, reproducible, clinically meaningful, and statistically significant improvement in reducing pain and improving function in patients with diagnostic evidence of vertebral pain. Among 11 clinical studies with direct patient data, 3 reviews, 2 clinical guidelines, and 1 systematic review published to date, there is a consensus that the procedure is effective at short-term and perhaps long-term in patients that meet the above-described indications.[24][25][26]
Documented improvements in pain reduction measured by the visual analog scale, functional disability measured by the Oswestry disability index, opioid utilization reduction, and improved quality of life measured by the short-form 36 and EQ-5D-5L have been reported. In fact, studies have pointed out that there is evidence that BVN ablation is more beneficial than the current standard of care for the treatment of low back pain from damaged vertebral endplates.[28][29][30]
The current literature with level Ia-IV studies consistently exhibits positive outcomes with a minimal clinically important difference in reducing pain and improving function, and based upon the available present data, society guidelines strongly recommend BVN ablation in a subset of patients with chronic axial LBP, particularly those with pain greater than 6 months of duration, unresponsive to conservative treatment for at least 6 months, and with diagnostic evidence of vertebral endplate damage (MCs) suggesting a vertebrogenic etiology.[32][33][34][35]
Enhancing Healthcare Team Outcomes
Interprofessional team multidisciplinary collaboration is essential for optimal outcomes in low back pain treatment, and a risk/benefit analysis should always be completed before proceeding with this intervention, as any other spinal procedure, the standard of care should always be followed with conservative methods first, and a treatment plan embracing shared decision making between the patient and the health care team. Clinicians are responsible for proper diagnosis, perioperative assessment, surgical intervention, and postoperative follow-up.
In addition, in this interprofessional model, clinicians act as guides to patients along the healthcare continuum of the low back pain treatment algorithm. Nursing facilitates coordination of activities among therapists and other consultants that help patients achieve their maximum clinical improvement after the intervention and alongside the device representative and the physician educating patients on the expected pre and post-intervention course. This interprofessional, multidisciplinary model will lead to the optimization of patient outcomes. [Level 5]
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
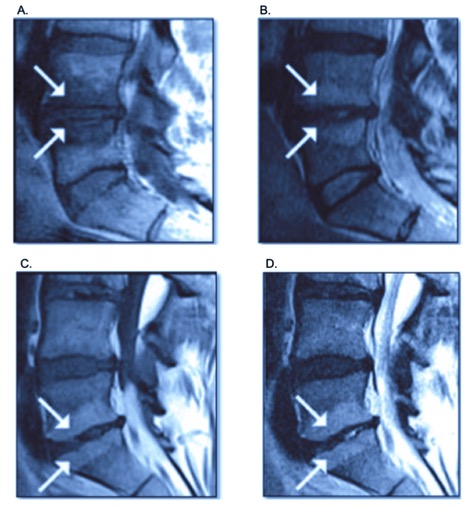
Figure 2. Magnetic resonance image (MRI) of the lumbar spine demonstrating type 1 (A, B) and type 2 (C,D) vertebral endplate damage with Modic changes. The white arrows point to the signaling changes on different MRI sequences. Modic changes type 1 with hypodense or decreased signal intensity of fibrovascular intraosseous bone marrow edema on T1-weighted MRI sequence and as hyperintense or increase signal intensity on T2-weighted MRI sequence, while Modic type 2 shows increased signal intensity in both T1 and T2 MRI sequence images. Contributed courtesy of Relievant Medsystems Inc.
(Click Image to Enlarge)
References
Mok FP, Samartzis D, Karppinen J, Fong DY, Luk KD, Cheung KM. Modic changes of the lumbar spine: prevalence, risk factors, and association with disc degeneration and low back pain in a large-scale population-based cohort. The spine journal : official journal of the North American Spine Society. 2016 Jan 1:16(1):32-41. doi: 10.1016/j.spinee.2015.09.060. Epub 2015 Oct 8 [PubMed PMID: 26456851]
Kim HS, Wu PH, Jang IT. Lumbar Degenerative Disease Part 1: Anatomy and Pathophysiology of Intervertebral Discogenic Pain and Radiofrequency Ablation of Basivertebral and Sinuvertebral Nerve Treatment for Chronic Discogenic Back Pain: A Prospective Case Series and Review of Literature. International journal of molecular sciences. 2020 Feb 21:21(4):. doi: 10.3390/ijms21041483. Epub 2020 Feb 21 [PubMed PMID: 32098249]
Level 2 (mid-level) evidenceTzika M, Paraskevas GK, Piagkou M, Papatolios AK, Natsis K. Basivertebral foramina of true vertebrae: morphometry, topography and clinical considerations. Surgical and radiologic anatomy : SRA. 2021 Jun:43(6):889-907. doi: 10.1007/s00276-021-02690-0. Epub 2021 Feb 17 [PubMed PMID: 33598754]
Kjaer P, Korsholm L, Bendix T, Sorensen JS, Leboeuf-Yde C. Modic changes and their associations with clinical findings. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2006 Sep:15(9):1312-9 [PubMed PMID: 16896838]
Level 2 (mid-level) evidenceWeishaupt D, Zanetti M, Hodler J, Min K, Fuchs B, Pfirrmann CW, Boos N. Painful Lumbar Disk Derangement: Relevance of Endplate Abnormalities at MR Imaging. Radiology. 2001 Feb:218(2):420-7 [PubMed PMID: 11161156]
Fras C, Kravetz P, Mody DR, Heggeness MH. Substance P-containing nerves within the human vertebral body. an immunohistochemical study of the basivertebral nerve. The spine journal : official journal of the North American Spine Society. 2003 Jan-Feb:3(1):63-7 [PubMed PMID: 14589248]
Kuisma M, Karppinen J, Niinimäki J, Ojala R, Haapea M, Heliövaara M, Korpelainen R, Taimela S, Natri A, Tervonen O. Modic changes in endplates of lumbar vertebral bodies: prevalence and association with low back and sciatic pain among middle-aged male workers. Spine. 2007 May 1:32(10):1116-22 [PubMed PMID: 17471095]
Jensen RK, Leboeuf-Yde C. Is the presence of modic changes associated with the outcomes of different treatments? A systematic critical review. BMC musculoskeletal disorders. 2011 Aug 10:12():183. doi: 10.1186/1471-2474-12-183. Epub 2011 Aug 10 [PubMed PMID: 21831312]
Level 1 (high-level) evidenceBailey JF, Liebenberg E, Degmetich S, Lotz JC. Innervation patterns of PGP 9.5-positive nerve fibers within the human lumbar vertebra. Journal of anatomy. 2011 Mar:218(3):263-70. doi: 10.1111/j.1469-7580.2010.01332.x. Epub 2011 Jan 12 [PubMed PMID: 21223256]
DePalma MJ, Ketchum JM, Saullo T. What is the source of chronic low back pain and does age play a role? Pain medicine (Malden, Mass.). 2011 Feb:12(2):224-33. doi: 10.1111/j.1526-4637.2010.01045.x. Epub 2011 Jan 25 [PubMed PMID: 21266006]
Level 2 (mid-level) evidenceLotz JC, Fields AJ, Liebenberg EC. The role of the vertebral end plate in low back pain. Global spine journal. 2013 Jun:3(3):153-64. doi: 10.1055/s-0033-1347298. Epub 2013 May 23 [PubMed PMID: 24436866]
Deyo RA, Mirza SK, Martin BI. Back pain prevalence and visit rates: estimates from U.S. national surveys, 2002. Spine. 2006 Nov 1:31(23):2724-7 [PubMed PMID: 17077742]
Level 3 (low-level) evidenceJensen TS, Karppinen J, Sorensen JS, Niinimäki J, Leboeuf-Yde C. Vertebral endplate signal changes (Modic change): a systematic literature review of prevalence and association with non-specific low back pain. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2008 Nov:17(11):1407-22. doi: 10.1007/s00586-008-0770-2. Epub 2008 Sep 12 [PubMed PMID: 18787845]
Level 2 (mid-level) evidenceJärvinen J, Karppinen J, Niinimäki J, Haapea M, Grönblad M, Luoma K, Rinne E. Association between changes in lumbar Modic changes and low back symptoms over a two-year period. BMC musculoskeletal disorders. 2015 Apr 22:16():98. doi: 10.1186/s12891-015-0540-3. Epub 2015 Apr 22 [PubMed PMID: 25897658]
Jensen RK, Leboeuf-Yde C, Wedderkopp N, Sorensen JS, Manniche C. Rest versus exercise as treatment for patients with low back pain and Modic changes. A randomized controlled clinical trial. BMC medicine. 2012 Feb 29:10():22. doi: 10.1186/1741-7015-10-22. Epub 2012 Feb 29 [PubMed PMID: 22376791]
Level 1 (high-level) evidenceDagenais S, Caro J, Haldeman S. A systematic review of low back pain cost of illness studies in the United States and internationally. The spine journal : official journal of the North American Spine Society. 2008 Jan-Feb:8(1):8-20. doi: 10.1016/j.spinee.2007.10.005. Epub [PubMed PMID: 18164449]
Level 1 (high-level) evidenceHoy D, Bain C, Williams G, March L, Brooks P, Blyth F, Woolf A, Vos T, Buchbinder R. A systematic review of the global prevalence of low back pain. Arthritis and rheumatism. 2012 Jun:64(6):2028-37. doi: 10.1002/art.34347. Epub 2012 Jan 9 [PubMed PMID: 22231424]
Level 1 (high-level) evidenceShayota B, Wong TL, Fru D, David G, Iwanaga J, Loukas M, Tubbs RS. A comprehensive review of the sinuvertebral nerve with clinical applications. Anatomy & cell biology. 2019 Jun:52(2):128-133. doi: 10.5115/acb.2019.52.2.128. Epub 2019 Jun 30 [PubMed PMID: 31338228]
Modic MT, Steinberg PM, Ross JS, Masaryk TJ, Carter JR. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology. 1988 Jan:166(1 Pt 1):193-9 [PubMed PMID: 3336678]
Level 2 (mid-level) evidenceAlbert HB, Kjaer P, Jensen TS, Sorensen JS, Bendix T, Manniche C. Modic changes, possible causes and relation to low back pain. Medical hypotheses. 2008:70(2):361-8 [PubMed PMID: 17624684]
Applebaum A, Nessim A, Cho W. Modic Change: An Emerging Complication in the Aging Population. Clinical spine surgery. 2022 Feb 1:35(1):12-17. doi: 10.1097/BSD.0000000000001168. Epub [PubMed PMID: 33769981]
Herlin C, Kjaer P, Espeland A, Skouen JS, Leboeuf-Yde C, Karppinen J, Niinimäki J, Sørensen JS, Storheim K, Jensen TS. Modic changes-Their associations with low back pain and activity limitation: A systematic literature review and meta-analysis. PloS one. 2018:13(8):e0200677. doi: 10.1371/journal.pone.0200677. Epub 2018 Aug 1 [PubMed PMID: 30067777]
Level 1 (high-level) evidenceMera Y, Teraguchi M, Hashizume H, Oka H, Muraki S, Akune T, Kawaguchi H, Nakamura K, Tamai H, Tanaka S, Yoshida M, Yoshimura N, Yamada H. Association between types of Modic changes in the lumbar region and low back pain in a large cohort: the Wakayama spine study. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2021 Apr:30(4):1011-1017. doi: 10.1007/s00586-020-06618-x. Epub 2020 Oct 9 [PubMed PMID: 33037486]
Fischgrund JS, Rhyne A, Franke J, Sasso R, Kitchel S, Bae H, Yeung C, Truumees E, Schaufele M, Yuan P, Vajkoczy P, Depalma M, Anderson DG, Thibodeau L, Meyer B. Intraosseous Basivertebral Nerve Ablation for the Treatment of Chronic Low Back Pain: 2-Year Results From a Prospective Randomized Double-Blind Sham-Controlled Multicenter Study. International journal of spine surgery. 2019 Apr:13(2):110-119. doi: 10.14444/6015. Epub 2019 Apr 30 [PubMed PMID: 31131209]
Level 2 (mid-level) evidenceKhalil JG, Smuck M, Koreckij T, Keel J, Beall D, Goodman B, Kalapos P, Nguyen D, Garfin S, INTRACEPT Trial Investigators. A prospective, randomized, multicenter study of intraosseous basivertebral nerve ablation for the treatment of chronic low back pain. The spine journal : official journal of the North American Spine Society. 2019 Oct:19(10):1620-1632. doi: 10.1016/j.spinee.2019.05.598. Epub 2019 Jun 20 [PubMed PMID: 31229663]
Level 2 (mid-level) evidenceMarkman JD, Rhyne AL, Sasso RC, Patel AA, Hsu WK, Fischgrund JS, Edidin AA, Vajkoczy P. Association Between Opioid Use and Patient-Reported Outcomes in a Randomized Trial Evaluating Basivertebral Nerve Ablation for the Relief of Chronic Low Back Pain. Neurosurgery. 2020 Mar 1:86(3):343-347. doi: 10.1093/neuros/nyz093. Epub [PubMed PMID: 31034561]
Level 1 (high-level) evidenceTruumees E, Macadaeg K, Pena E, Arbuckle J 2nd, Gentile J 2nd, Funk R, Singh D, Vinayek S. A prospective, open-label, single-arm, multi-center study of intraosseous basivertebral nerve ablation for the treatment of chronic low back pain. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2019 Jul:28(7):1594-1602. doi: 10.1007/s00586-019-05995-2. Epub 2019 May 21 [PubMed PMID: 31115683]
Fischgrund JS, Rhyne A, Macadaeg K, Moore G, Kamrava E, Yeung C, Truumees E, Schaufele M, Yuan P, DePalma M, Anderson DG, Buxton D, Reynolds J, Sikorsky M. Long-term outcomes following intraosseous basivertebral nerve ablation for the treatment of chronic low back pain: 5-year treatment arm results from a prospective randomized double-blind sham-controlled multi-center study. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society. 2020 Aug:29(8):1925-1934. doi: 10.1007/s00586-020-06448-x. Epub 2020 May 25 [PubMed PMID: 32451777]
Level 1 (high-level) evidenceDe Vivo AE, D'Agostino G, D'Anna G, Al Qatami H, Gil I, Ventura F, Manfrè L. Intra-osseous basivertebral nerve radiofrequency ablation (BVA) for the treatment of vertebrogenic chronic low back pain. Neuroradiology. 2021 May:63(5):809-815. doi: 10.1007/s00234-020-02577-8. Epub 2020 Oct 13 [PubMed PMID: 33051706]
Smuck M, Khalil J, Barrette K, Hirsch JA, Kreiner S, Koreckij T, Garfin S, Mekhail N, INTRACEPT Trial Investigators. Prospective, randomized, multicenter study of intraosseous basivertebral nerve ablation for the treatment of chronic low back pain: 12-month results. Regional anesthesia and pain medicine. 2021 Aug:46(8):683-693. doi: 10.1136/rapm-2020-102259. Epub 2021 May 24 [PubMed PMID: 34031220]
Level 2 (mid-level) evidenceKim HS, Adsul N, Yudoyono F, Paudel B, Kim KJ, Choi SH, Kim JH, Chung SK, Choi JH, Jang JS, Jang IT, Oh SH. Transforaminal Epiduroscopic Basivertebral Nerve Laser Ablation for Chronic Low Back Pain Associated with Modic Changes: A Preliminary Open-Label Study. Pain research & management. 2018:2018():6857983. doi: 10.1155/2018/6857983. Epub 2018 Aug 14 [PubMed PMID: 30186540]
Nguyen KML, Nguyen DTD. Minimally Invasive Treatment for Degenerative Lumbar Spine. Techniques in vascular and interventional radiology. 2020 Dec:23(4):100700. doi: 10.1016/j.tvir.2020.100700. Epub 2020 Oct 5 [PubMed PMID: 33308584]
Urits I, Noor N, Johal AS, Leider J, Brinkman J, Fackler N, Vij N, An D, Cornett EM, Kaye AD, Viswanath O. Basivertebral Nerve Ablation for the Treatment of Vertebrogenic Pain. Pain and therapy. 2021 Jun:10(1):39-53. doi: 10.1007/s40122-020-00211-2. Epub 2020 Oct 31 [PubMed PMID: 33128702]
Conger A, Schuster NM, Cheng DS, Sperry BP, Joshi AB, Haring RS, Duszynski B, McCormick ZL. The Effectiveness of Intraosseous Basivertebral Nerve Radiofrequency Neurotomy for the Treatment of Chronic Low Back Pain in Patients with Modic Changes: A Systematic Review. Pain medicine (Malden, Mass.). 2021 May 21:22(5):1039-1054. doi: 10.1093/pm/pnab040. Epub [PubMed PMID: 33544851]
Level 1 (high-level) evidenceLorio M, Clerk-Lamalice O, Beall DP, Julien T. International Society for the Advancement of Spine Surgery Guideline-Intraosseous Ablation of the Basivertebral Nerve for the Relief of Chronic Low Back Pain. International journal of spine surgery. 2020 Feb:14(1):18-25. doi: 10.14444/7002. Epub 2020 Feb 29 [PubMed PMID: 32128298]