Indications
FDA-Approved Indications
Glaucoma and Ocular Hypertension
Ophthalmic topical bimatoprost is a synthetic prostamide structurally related to prostaglandin F2 alpha (F2α). This drug received approval from the United States Food and Drug Administration (FDA) in 2001 as a treatment to lower intraocular pressure (IOP). Evidence indicates that topical bimatoprost reduces IOP in eyes with primary open-angle glaucoma (POAG) and ocular hypertension.[1] Growth of the eyelashes is a well-documented side effect of bimatoprost during the treatment of glaucoma.[2][3]
Hypotrichosis
In 2008, the FDA-approved ophthalmic bimatoprost to treat hypotrichosis, such as for patients who are post-chemotherapy.[4] Bimatoprost ophthalmic solution is often purchased as an over-the-counter (OTC) ophthalmic applicator product for cosmetic purposes as it can cause longer, thicker, and darker eyelashes.[5] Generic bimatoprost became available in 2015.
The ophthalmic bimatoprost is available in topical 0.01% and 0.03% concentrations. When used to lower IOP, a once-daily dosing of bimatoprost may be used alone or concomitantly with another drug, such as timolol.[6] In a 12-month comparison study in lowering IOP, bimatoprost 0.01% was comparable to bimatoprost 0.03%. However, the 0.01% formulation had less frequent and severe adverse ocular events, which became the primary concentration available in the United States.[7] Preservative-free and generic formulations of bimatoprost are currently available worldwide.
Eyelash Growth
Once-daily bimatoprost ophthalmic solution of 0.03% applied to the upper eyelashes has been studied extensively to enhance eyelash growth with favorable safety profiles and tolerability.[2][8]
Mechanism of Action
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Mechanism of Action
Chemistry
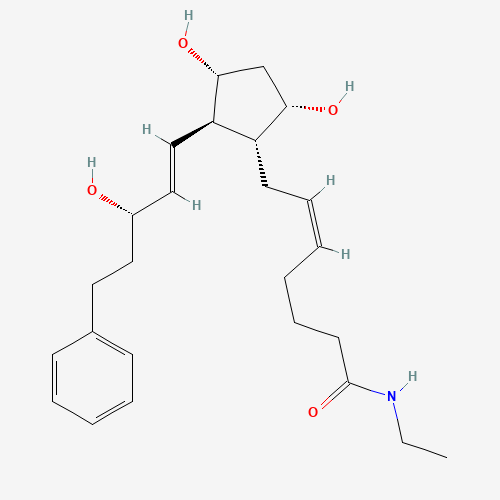
The molecular formula for bimatoprost is C25H37NO4 (see Image. Bimatoprost Structure). The IUPAC chemical name is (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(E,3S)-3-hydroxy-5-phenylpent-1-enyl]cyclopentyl]-N-ethylhept-5-enamide. This drug is a synthetic prostamide analog that is structurally similar to prostaglandin (PG) F2α. Prostamides are fatty acid amides that differ from prostaglandins because they lack a negatively charged carboxylic acid and tend to be neutral.[9] The pH ranges from 6.8 to 7.8. Bimatoprost has a molecular weight of 415.58 g/mol. The solution used in ophthalmology drops is sterile, colorless, isotonic, and clear, with an osmolarity of about 290 mOsm/kg.[10] The multidose formulation has shown stability for at least 28 days when stored at 2 °C to 25 °C, with a shelf-life of about 2 years.[11]
Pharmacodynamics
Prostaglandins, which were initially thought to be derived from the prostate gland (hence the name), were first discovered in semen in the 1930s.[9] Prostaglandins elicit effects via interaction with corresponding G-protein coupled receptors found throughout the body.[12] Studies based on ocular extracts showed the presence of prostaglandins capable of contracting the iris. The effects of topically applied PG2α were first reported in human volunteers in 1985.[13] PG receptors exist in ciliary and corneal epithelium, iris stroma, ciliary muscle, choroid, trabecula meshwork, and retina.[14] Of the 9 different types of prostaglandin receptors, bimatoprost has been shown to have an agonist activity and high affinity for PG receptors EP1, EP3, and FP.[15]
Pharmacokinetics
Bimatoprost is a synthetic structural analog of prostaglandin F2α and has been hypothesized to exert its effects as an agonist through prostaglandin FP receptors.[16][17] Because bimatoprost is an amide rather than an ester, it is often labeled as a prostamide.[18] When applied topically, penetration occurs through the cornea and sclera. The preferential target for this drug has been reported to be the ciliary body and cornea, showing 10-fold higher concentrations in the ciliary body compared to aqueous humor.[19][20] Systemic absorption is minimal, reaching peak plasma concentrations within 10 minutes and no longer detectable after 2 hours, with no signs of tissue accumulation.[21] The elimination half-life of the drug administered intravenously is about 45 minutes, mainly through urine and about 25% via feces.[22]
Mechanism of Action in the Reduction of Intraocular Pressure
It has been well demonstrated and validated that bimatoprost and several topical prostaglandin analogs sustainably and effectively reduce interaocular pressure (IOP) and are used as a standard first-line treatment of POAG.[23] This is achieved primarily through enhancing aqueous outflow through the uveoscleral pathway.[24] Specifically, bimatoprost increases the level of remodeling enzymes like tissue inhibitors of metalloproteinases (TIMP) and matrix metalloproteinases (MMP) in the ciliary muscles and sclera, in addition to causing a widening of the spaces in connective tissue to enhance aqueous outflow.[25] These MMPs then induce the decomposition of collagen within the extracellular matrix, resulting in improved pressure-insensitive uveoscleral outflow.[26] Ciliary muscle relaxation and contractile activity induced by bimatoprost have also been shown to play a role in decreasing IOP.[27][28]
Several studies have also demonstrated that bimatoprost might have a bimodal mechanism and encourage trabecular and uveoscleral outflow.[24] Studies based on prostaglandins have shown that this class of drugs can also cause a relaxation of the trabecular meshwork (TM), thus enhancing conventional pressure-sensitive outflow mechanisms.[29] Bimatoprost has not demonstrated any effect on aqueous humor production to reduce IOP.[30]
Clinical Efficacy
It is shown that lowering IOP in patients with ocular hypertension and glaucoma may reduce the risk of disease progression.[7] All forms of current treatment, including medical topical drops, laser treatments, and surgery, are all geared at lowering IOP to slow down irreversible glaucomatous progression.[31] Numerous studies have confirmed that IOP reduction ranges from 28% to 31% with prostaglandins.[32]
Compared to the other current forms of prostaglandin ocular drops, which include latanoprost, travoprost, and tafluprost, numerous studies have confirmed that bimatoprost has greater or similar efficacy in reducing IOP than others.[33] Several randomized trials have shown that the switch from other prostaglandins to bimatoprost showed better IOP-reducing efficacy than travoprost, tafluprost, and latanoprost with little switching back rates and a small statistically significant reduction in mean IOP after the switch.[34][35]
The effects in reducing IOP begin about 4 hours after the topical instillation of bimatoprost, which reaches maximum effects after 8 to 12 hours and lasts for 24 hours; it must be administered only once daily, preferably at night.[36]
Bimatoprost (Lumigan) first became available in 2001 as a multidose 0.03% (0.3 mg/mL) ophthalmic solution with benzalkonium chloride (BAK). This prostaglandin reduces IOP and is considered one of the most efficient antiglaucomatous topical and first-line therapies compared to other monotherapy drop alternatives.[37] Although bimatoprost 0.03% is very effective in reducing IOP, compliance rates tend to be less when compared to other prostaglandins due to the adverse effects of conjunctival hyperemia, eyelash growth, periocular redness, and pigmentation, which tend to be higher with bimatoprost 0.03%.[38][39] Preservative-free formulations of 0.03% bimatoprost then became available to reduce the adverse effects of BAK on the ocular surface.[40]
In 2010, Allergan began selling Lumigan 0.01% (0.1 mg/mL) ophthalmic solution to the public. Numerous studies comparing the 2 different single and fixed formulations of bimatoprost, 0.03% and 0.01%, have reported similar efficacy in reducing IOP but less adverse effects for bimatoprost 0.01% in patients with glaucoma and ocular hypertension.[41][42] Preservative-free formulations of bimatoprost 0.01% and generics are currently available, which have shown similar efficacy in reducing IOP with a better tolerability profile when compared to other bimatoprost formulations.[43]
The majority of the studies regarding bimatoprost have shown clinical efficiency and first-line therapy in reducing IOP in patients with primary open-angle glaucoma (POAG) and ocular hypertension (OHT).[44][45] Numerous trials have included patients with different forms of glaucoma, like pseudoexfoliative, pigmentary, chronic angle-closure, etc.[42][46][47]
Safety and Tolerability
Treatment with bimatoprost once daily has a favorable safety profile and efficacy and is tolerated in most patients with glaucoma and ocular hypertension when compared with other prostaglandins.[48] The adverse effects of conjunctival hyperemia, periocular pigmentation, and eyelash growth are slightly higher with the bimatoprost 0.03% formulation.[49] However, the 0.01% preservative-free formulation shows improvement in ocular side effects similar to other prostaglandins, corresponding to improved patient comfort and enhanced therapeutic compliance.[50]
Prevalence and Pharmacoeconomics
It has been estimated that more than 110 million people worldwide (older than 40 years old) will be affected by glaucoma in 2040.[51] Annual costs for treating glaucoma in 2000 were reported to be more than $1 billion in the US and $200 million in Canada, with about 50% undiagnosed and not treated because of the asymptomatic nature of the disease.[52] The annual health-related costs for glaucoma worldwide reported in 1999 ranged from $300 to $950 per patient, with 25% to 35% of the expenses for drug therapy.[52]
The only identified modifiable risk factor to slow down or prevent glaucomatous progression and vision loss is the reduction in IOP, in which first-line therapy is by medical management.[53] Analyses based on cost-effective treatments by US healthcare providers for POAG and OHT showed lower costs per treatment with bimatoprost compared to other therapeutic regimens.[54] Studies conducted in Austria, the UK, Finland, France, Spain, Asia, Italy, and numerous other parts of the world have confirmed the efficacy and cost-related benefits related to the use of bimatoprost in patients with OHT and POAG.[52] European studies reported that adding bimatoprost to first-line therapy with timolol was the most common form of treatment in managing patients with glaucoma and OHT.[55]
Enhancement of Hair and Eyelash Growth
The ability of bimatoprost to promote the growth and thickening of eyelashes was first noted during glaucoma clinical trials to investigate the potential to reduce IOP. Although the previously mentioned prostaglandin receptors are involved in regenerating the dermal papilla outer root sheath and throughout hair follicles, it remains unknown whether bimatoprost affects this pathway.[16][56] Murine models have shown that bimatoprost stimulates eyelash hair growth and increases the length.[57] The drug promotes anagen phases of hair growth by arresting catagen phase entry. Bimatoprost increases the generated hair's thickness and bulb diameter; it does not generate new hair follicles.[58] Bimatoprost also tends to darken the treated lashes.[59]
A series of studies demonstrated that with a 2-week course of bimatoprost, a more significant proportion of follicles were in the hair cycle's anagen phase and a decreased portion in the telogen phase, suggesting an increased transition from telogen to anagen. The study hypothesized that this increased proportion of follicles in the anagen phase allows for the observable enhanced growth of eyelashes with bimatoprost.[3] The FDA approved bimatoprost 0.03% solution for treating hypotrichosis of eyelashes in 2008 after the reported side effects of this drug in patients using it for glaucoma and OHT.[60]
Bimatoprost has also been considered for hypotrichosis of the eyebrow. A large randomized study of 357 subjects with hypotrichosis of the eyebrow reported a statistically significant increase of at least one grade in Global Wyebrow Assessment in patients treated with bimatoprost 0.03% compared with vehicle.[61]
Other uses for bimatoprost include treatment for dermatological diseases, including alopecia, hair loss or repigmentation, vitiligo, and localized leukoderma.[58][62][63] Bimatoprost has shown a higher percentage of hair regrowth than other treatment regimes in patients with autoimmune-induced patchy alopecia areata.[64][65] Clinical trials have also demonstrated efficacy, safety, and benefits in women and men with androgenetic alopecia.[66]
Administration
Applying Bimatoprost Ophthalmic Solution for Glaucoma and Ocular Hypertension
Bimatoprost ophthalmic solution 0.01% or 0.03% may be used as eye drops applied directly to the ocular surface. The suggested dose of bimatoprost is 1 drop every evening. The patient should wait 5 minutes between administering bimatoprost and other ophthalmic drugs and artificial tears. With bimatoprost, the reduction of IOP has been shown to begin after 4 hours, peak after 12 hours, and maintain stability in the decrease in IOP for 24 hours.[67] Clinical trials of 1-year duration demonstrate that bimatoprost effectively lowers IOP long-term and is generally well tolerated.[68] A tissue can remove excess therapy around the lids and periorbital region to decrease the adverse local effects.
Applying Bimatoprost Ophthalmic Solution for Hypotrichosis
Bimatoprost ophthalmic solution 0.03% is most commonly used and associated with increased growth and prominence of eyelashes with once-daily administration. The medication and included applicators are currently available commercially as a bottled solution. Before application, patients should remove contact lenses. Applying any solution directly to the eye is not recommended. Every evening, a drop of the solution is applied to the single-use-per-eye applicator and brushed against the base of the upper eyelid margin. The applicator is to be used with only one eye to prevent contamination.
Evidence suggests that the solution should be applied for at least 16 weeks for maximum eyelash prominence.[2] Bimatoprost solution on the scalp is recommended to treat alopecia, which can be combined with other therapy types.[69]
Application of a New Bimatoprost Implant for Open-angled Glaucoma
Although the topical ophthalmic solution of bimatoprost is highly effective for reducing IOP, there are also serious concerns about disease management due to poor adherence by the patients.[70] Factors associated with decreased adherence to the recommended treatments include difficulty with eye drop administration, frequent dosing, adverse side effects, false perception of the disease, and forgetfulness.[70]
Intracameral drug delivery systems have been developed to circumvent these concerns.[71] A bimatoprost sustained-release implant was designed and approved in the USA in 2020 for patients with ocular hypertension and open-angle glaucoma.[72] The intracameral implant is a single, sterile, rod-shaped solid polymer that consists of a 10 or 15 μg dose of bimatoprost lasting 4 to 6 months.[73] The drug is preloaded into a single-used applicator and then injected into the anterior chamber, allowing the implant to deliver the prostaglandin analog directly to the iris-ciliary body.
A recent phase 3 trial demonstrated that after 3 administrations of the implants at day 1, week 16, and week 32, most patients did not require additional treatment after 12 months and demonstrated noninferiority to topical ophthalmic solutions for OAG.[74][45] Studies have shown that the 2020 FDA-approved intracameral sustained-release implant of bimatoprost (Durysta) provided advantages related to patient adherence, ocular surface and periorbital adverse effects, and IOP reduction in patients with glaucoma.[72][75] The main safety concern with these intracameral implants remains the gradual, irreversible loss of corneal endothelial cells, especially in repeat implants.[76]
The bimatoprost ocular ring is another sustained release method of ocular drug delivery.[53] A polypropylene and silicone ring ranging in diameter from 24 to 29 mm is impregnated with bimatoprost. The ring is positioned between the lower and upper fornices and provides continuous bimatoprost delivery for up to 6 months, with IOP comparable to daily bimatoprost topical drops.[77]
The use of contact lenses with micelle-laden drugs is being studied as an alternative drug delivery system. However, research tends to be limited to animal models, and the risks related to long-term contact use remain an essential limitation.[78] Nonasponges with bimatoprost and injection of drug-laden microspheres have been reported in the literature; they remain preliminary and experimental in animal studies.[79]
Adverse Effects
Numerous trials have demonstrated the high level of safety and tolerance of bimatoprost when applied to the ocular surface or eyelids.[68][80]
The most common adverse events related to the local use of bimatoprost eye drops include:
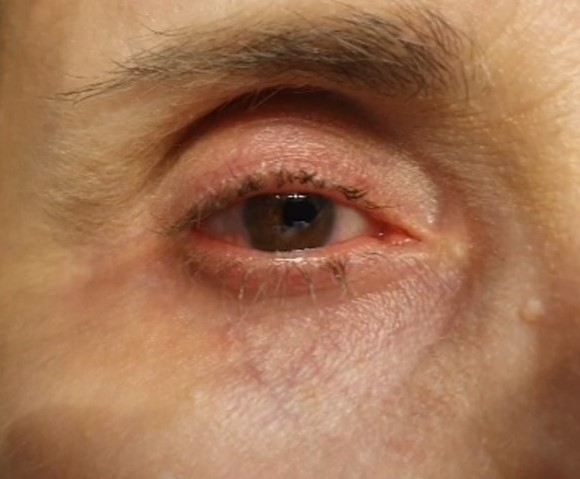
- Conjunctival hyperemia (see Image. Adverse Affects) which peaks by day 15, then tends to decrease after several months (up to 45% of patients)
- Eyelash growth and darkening (see Image. Adverse Affects) (15% to 35% of patients)
- Eye pruritus and erythema (see Image. Adverse Affects) (about 10% of patients)
- Blepharitis
- Conjunctivitis
- Dry eye syndrome
- Burning irritation
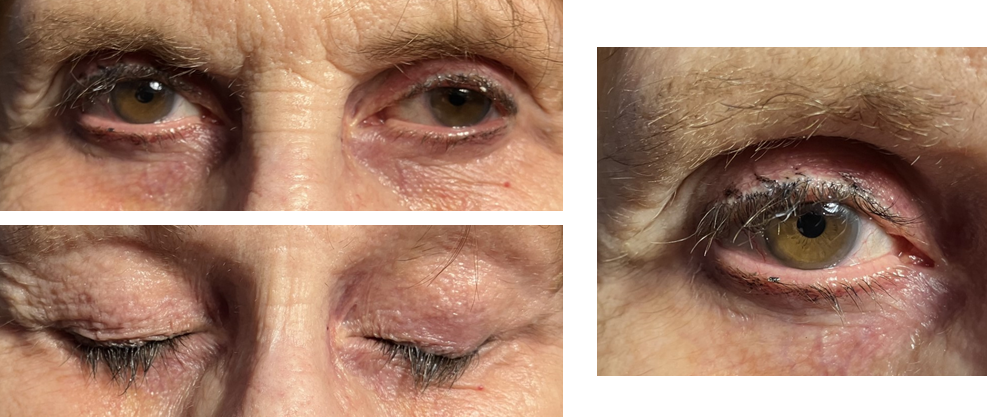
- Pigmentation of eyelid margins and periocular regions (see Image. Mild Adverse Affects)
- Iris pigmentation (see Image. Adverse Affects)
- Visual disturbances
- Eye stinging and pain
- Foreign body sensations
- Tearing
- Photophobia
- Superficial punctate keratits
- Periorbital fat atrophy and deepening of upper eyelid sulcus [25]
Studies have highlighted prostaglandin-associated periorbitopathy commonly seen in prolonged use of topical prostaglandin analogs, especially with bimatoprost.[81] Periorbitopathy is typically seen clinically as periorbital fat atrophy, mild ptosis, and deepening of the upper lid sulcus (seen in 80% of patients using bimatoprost).[82][83] A review noted an underreported incidence of discomfort and prostaglandin-associated periorbitopathy, possibly due to the topic being dominated by industry-sponsored studies.[84]
These adverse effects of periorbital fat atrophy with bimatoprost are being considered in treating thyroid eye disease (TED). This disorder is due to an inflammatory response that can cause soft tissue expansion within the orbit, leading to exophthalmos. Preliminary clinical studies have shown potential benefits for TED-induced exophthalmos in patients taking bimatoprost.[85]
Several case reports have illustrated an association between bimatoprost use and the reactivation of herpetic keratitis.[86][87] One significant but rare side effect is developing or exacerbating anterior non-granulomatous uveitis.[88][89] The incidence of drug-induced uveitis is very low and tends to manifest clinically as anterior iridocyclitis.[90]
Bimatoprost applied as an eye drop has also been associated with the alteration of iris pigmentation. The change and darkening of the iris are caused by increased melanosomes within the melanocytes in the iris stroma, not an increase in the number of melanocytes.[91] Brown and hazel-colored irises are more at risk of hyperpigmentation with bimatoprost, which can be irreversible.[91] Darkening of the iris tends to occur within the first year of treatment and increases in time with continual use of the drug.
Multiple case reports have suggested various prostaglandin analogs, such as bimatoprost and latanoprost, can be associated with cystoid macular edema (CME) due to their pro-inflammatory effects.[18] CME is a form of thickening of the macula characterized by intraretinal cystic fluid-filled spaces visible with optical coherence tomography (OCT). Prostaglandins, especially bimatoprost, are thought to induce blood-aqueous barrier disruption and thus favor the formation of CME after surgery or in patients with other forms of ocular inflammation.[92] Studies have documented a potential association between using ophthalmic prostaglandin analogs and CME after cataract surgery in patients with glaucoma. This justifies the temporary suspension of bimatoprost and modification in glaucoma therapy in patients undergoing surgery and with ocular infections and inflammation.[93] Carbonic anhydrase inhibitors have shown to be a valid alternative to bimatoprost during the early pre and postoperative periods or in patients with inflammation at risk of CME.[94][95]
Regarding the cosmetic use of bimatoprost, studies have shown that the safety profile for dermal application of bimatoprost ophthalmic solution 0.03% is more favorable than that seen with topical eye drop application.[58] The difference in dose may explain the significant difference in the likelihood of adverse effects between the 2 methods of bimatoprost application. A single application of a bimatoprost 0.03% dermally results in only about 5% of the amount applied for treatment for glaucoma.[59] The following adverse effects have been reported in fewer than 4% of patients following dermal application:[59]
- Conjunctival hyperemia (most common in 3.6% of patients)
- Eye pruritus
- Skin hyperpigmentation
- Ocular irritation
- Dry eye syndrome
- Eyelid erythema
Studies including "before-and-after" images of patients using bimatoprost have shown the potential risks of developing prostaglandin-associated periorbitopathy due to periorbital fat atrophy and deepening of the upper eyelid sulcus.[96] Patients must know the possible adverse side effects of lash-lengthening serums with bimatoprost and other prostaglandins.
Contraindications
Bimatoprost is contraindicated in patients with a medical history of bimatoprost hypersensitivity or hypersensitivity to ingredients within the formulation. These include preservatives such as benzalkonium chloride.
Bimatoprost may not be appropriate for some cases of closed-angle glaucoma and inflammatory or neovascular glaucoma. Contact lenses should be removed before the bimatoprost ophthalmic solution is applied. They can be reinserted after 15 minutes.
The safety and efficacy of bimatoprost ophthalmic 0.03% dermal application for hypotrichosis have not been studied in children, neonates, and infants younger than 5 years old. The application should only be considered if the potential benefits justify the potential calculated risks.
Bimatoprost is considered a category-C drug for pregnancy. Bimatoprost ophthalmic solution has not been adequately studied in pregnant women and pediatric patients; it should be avoided or used cautiously in these cases unless deemed necessary.[11] The presence or absence of the drug in breast milk has not been established, considering the short half-life; it should be avoided during nursing if possible.[97] Pressure can be placed over the tear duct for about a minute, and removal of excess therapy with a tissue can be considered after applying ocular drops to decrease the amount of drug in the bloodstream.[98] Intraocular application of bimatoprost ophthalmic solution for glaucoma is contraindicated in neonates, infants, and children due to concern for pigmentation changes after long-term use.
Bimatoprost ophthalmic drops should be used with caution in patients with macular edema, uveitis, ocular inflammation, aphakia, complicated pseudophakia, a history of herpes simplex virus keratitis, and ocular inflammatory conditions.[10]
Monitoring
To treat glaucoma and ocular hypertension, bimatoprost ophthalmic solution should be initially monitored with regular clinic visits every 2 to 4 weeks until the patient's individualized target IOP is achieved. After reaching the IOP goal, the patient can be reassessed every 6 months or at the clinician's discretion. Additional parameters to monitor include ocular peri-ocular pigmentation changes.
When using bimatoprost ophthalmic solution 0.03% dermally for hypotrichosis, monitoring for potential side effects is advised. When bimatoprost is discontinued, eyelashes may return to their original pre-treatment length and thickness.
Toxicity
Currently, no studies have been performed to evaluate bimatoprost overdose in humans. If an overdose were to occur, manufacturers recommend supportive symptomatic treatment.
Enhancing Healthcare Team Outcomes
Bimatoprost is among the most effective first-line treatments for open-angle glaucoma and eyelash hypotrichosis. Several large, double-masked randomized control trials have shown bimatoprost’s superior efficacy in lowering IOP in primary open-angle glaucoma and ocular hypertension.[6][80] The efficacy of treatment of conditions including hypotrichosis and different forms of alopecia has been demonstrated in the literature.[99]
Successful treatment of glaucoma is highly dependent on interprofessional efforts to educate, communicate, and monitor for compliance with consistent eye drop use. Patients educated on the importance of the daily bimatoprost intervention were significantly more likely to report lower IOP values, which is critically important to preserve vision.[100] Evidence demonstrates that most patients are not bothered by bimatoprost’s most common side effect of hyperemia, especially if a healthcare team member has explained the importance of IOP-lowering medications.
Ultimately, collaboration and communication amongst an interprofessional team of ophthalmologists, ophthalmic technicians, nurses, and the patient will ensure the best care for the patient. A well-educated patient will, in turn, improve patient acceptance and compliance with the prescribed medication, resulting in optimized outcomes.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Li T, Lindsley K, Rouse B, Hong H, Shi Q, Friedman DS, Wormald R, Dickersin K. Comparative Effectiveness of First-Line Medications for Primary Open-Angle Glaucoma: A Systematic Review and Network Meta-analysis. Ophthalmology. 2016 Jan:123(1):129-40. doi: 10.1016/j.ophtha.2015.09.005. Epub 2015 Oct 31 [PubMed PMID: 26526633]
Level 2 (mid-level) evidenceCohen JL. Enhancing the growth of natural eyelashes: the mechanism of bimatoprost-induced eyelash growth. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2010 Sep:36(9):1361-71. doi: 10.1111/j.1524-4725.2010.01522.x. Epub [PubMed PMID: 20384750]
Barrón-Hernández YL, Tosti A. Bimatoprost for the treatment of eyelash, eyebrow and scalp alopecia. Expert opinion on investigational drugs. 2017 Apr:26(4):515-522. doi: 10.1080/13543784.2017.1303480. Epub 2017 Mar 16 [PubMed PMID: 28264599]
Level 3 (low-level) evidenceWirta D, Baumann L, Bruce S, Ahluwalia G, Weng E, Daniels S. Safety and Efficacy of Bimatoprost for Eyelash Growth in Postchemotherapy Subjects. The Journal of clinical and aesthetic dermatology. 2015 Apr:8(4):11-20 [PubMed PMID: 26060513]
Jones D. Enhanced eyelashes: prescription and over-the-counter options. Aesthetic plastic surgery. 2011 Feb:35(1):116-21. doi: 10.1007/s00266-010-9561-3. Epub 2010 Aug 21 [PubMed PMID: 20730536]
Moon SB, Han SB. Efficacy and safety of fixed-combination bimatoprost/timolol ophthalmic solution. Patient preference and adherence. 2017:11():1069-1070. doi: 10.2147/PPA.S141868. Epub 2017 Jun 26 [PubMed PMID: 29200831]
Lee D, Mantravadi AV, Myers JS. Patient considerations in ocular hypertension: role of bimatoprost ophthalmic solution. Clinical ophthalmology (Auckland, N.Z.). 2017:11():1273-1280. doi: 10.2147/OPTH.S118689. Epub 2017 Jul 10 [PubMed PMID: 28744094]
Wirta D, Pariser DM, Yoelin SG, Arase S, McMichael A, Weng E, Mao C, Demos G, Vandenburgh A. Bimatoprost 0.03% for the Treatment of Eyelash Hypotrichosis: A Pooled Safety Analysis of Six Randomized, Double-masked Clinical Trials. The Journal of clinical and aesthetic dermatology. 2015 Jul:8(7):17-29 [PubMed PMID: 26203317]
Level 1 (high-level) evidenceWan Z, Woodward DF, Cornell CL, Fliri HG, Martos JL, Pettit SN, Wang JW, Kharlamb AB, Wheeler LA, Garst ME, Landsverk KJ, Struble CS, Stamer WD. Bimatoprost, prostamide activity, and conventional drainage. Investigative ophthalmology & visual science. 2007 Sep:48(9):4107-15 [PubMed PMID: 17724194]
Level 3 (low-level) evidencePatil AJ, Vajaranant TS, Edward DP. Bimatoprost - a review. Expert opinion on pharmacotherapy. 2009 Nov:10(16):2759-68. doi: 10.1517/14656560903292649. Epub [PubMed PMID: 19874254]
Level 3 (low-level) evidenceCurran MP, Orman JS. Bimatoprost/timolol: a review of its use in glaucoma and ocular hypertension. Drugs & aging. 2009:26(2):169-84. doi: 10.2165/0002512-200926020-00008. Epub [PubMed PMID: 19220073]
Doucette LP, Walter MA. Prostaglandins in the eye: Function, expression, and roles in glaucoma. Ophthalmic genetics. 2017 Mar-Apr:38(2):108-116. doi: 10.3109/13816810.2016.1164193. Epub 2016 Apr 12 [PubMed PMID: 27070211]
Giuffrè G. The effects of prostaglandin F2 alpha in the human eye. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 1985:222(3):139-41 [PubMed PMID: 3856545]
Level 1 (high-level) evidenceAngeli A, Supuran CT. Prostaglandin receptor agonists as antiglaucoma agents (a patent review 2013 - 2018). Expert opinion on therapeutic patents. 2019 Oct:29(10):793-803. doi: 10.1080/13543776.2019.1661992. Epub 2019 Sep 4 [PubMed PMID: 31462124]
Level 3 (low-level) evidenceSharif NA, Kelly CR, Crider JY, Williams GW, Xu SX. Ocular hypotensive FP prostaglandin (PG) analogs: PG receptor subtype binding affinities and selectivities, and agonist potencies at FP and other PG receptors in cultured cells. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2003 Dec:19(6):501-15 [PubMed PMID: 14733708]
Level 3 (low-level) evidenceWoodward DF, Liang Y, Krauss AH. Prostamides (prostaglandin-ethanolamides) and their pharmacology. British journal of pharmacology. 2008 Feb:153(3):410-9 [PubMed PMID: 17721551]
Level 3 (low-level) evidenceCrowston JG, Lindsey JD, Morris CA, Wheeler L, Medeiros FA, Weinreb RN. Effect of bimatoprost on intraocular pressure in prostaglandin FP receptor knockout mice. Investigative ophthalmology & visual science. 2005 Dec:46(12):4571-7 [PubMed PMID: 16303950]
Level 3 (low-level) evidenceAgange N, Mosaed S. Prostaglandin-induced cystoid macular edema following routine cataract extraction. Journal of ophthalmology. 2010:2010():690707. doi: 10.1155/2010/690707. Epub 2010 Nov 7 [PubMed PMID: 21076526]
Level 3 (low-level) evidenceCamras CB, Sharif NA, Wax MB, Stjernschantz J. Bimatoprost, the prodrug of a prostaglandin analogue. The British journal of ophthalmology. 2008 Jun:92(6):862-3; author reply 863-4 [PubMed PMID: 18523093]
Level 3 (low-level) evidenceWoodward DF, Krauss AH, Chen J, Liang Y, Li C, Protzman CE, Bogardus A, Chen R, Kedzie KM, Krauss HA, Gil DW, Kharlamb A, Wheeler LA, Babusis D, Welty D, Tang-Liu DD, Cherukury M, Andrews SW, Burk RM, Garst ME. Pharmacological characterization of a novel antiglaucoma agent, Bimatoprost (AGN 192024). The Journal of pharmacology and experimental therapeutics. 2003 May:305(2):772-85 [PubMed PMID: 12606640]
Level 3 (low-level) evidenceHellberg MR, Ke TL, Haggard K, Klimko PG, Dean TR, Graff G. The hydrolysis of the prostaglandin analog prodrug bimatoprost to 17-phenyl-trinor PGF2alpha by human and rabbit ocular tissue. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2003 Apr:19(2):97-103 [PubMed PMID: 12804054]
Level 3 (low-level) evidenceCantor LB, Hoop J, Wudunn D, Yung CW, Catoira Y, Valluri S, Cortes A, Acheampong A, Woodward DF, Wheeler LA. Levels of bimatoprost acid in the aqueous humour after bimatoprost treatment of patients with cataract. The British journal of ophthalmology. 2007 May:91(5):629-32 [PubMed PMID: 17135335]
Level 1 (high-level) evidencePrum BE Jr, Lim MC, Mansberger SL, Stein JD, Moroi SE, Gedde SJ, Herndon LW Jr, Rosenberg LF, Williams RD. Primary Open-Angle Glaucoma Suspect Preferred Practice Pattern(®) Guidelines. Ophthalmology. 2016 Jan:123(1):P112-51. doi: 10.1016/j.ophtha.2015.10.055. Epub 2015 Nov 12 [PubMed PMID: 26581560]
Christiansen GA, Nau CB, McLaren JW, Johnson DH. Mechanism of ocular hypotensive action of bimatoprost (Lumigan) in patients with ocular hypertension or glaucoma. Ophthalmology. 2004 Sep:111(9):1658-62 [PubMed PMID: 15350319]
Level 1 (high-level) evidenceSubbulakshmi S, Kavitha S, Venkatesh R. Prostaglandin analogs in ophthalmology. Indian journal of ophthalmology. 2023 May:71(5):1768-1776. doi: 10.4103/IJO.IJO_2706_22. Epub [PubMed PMID: 37203029]
Park KS, Kim KN, Kim KM, Lee HM, Lee SB, Lee NH, Kim CS. Effects of Topical Prostaglandin Analog on Macular Thickness Following Cataract Surgery with Postoperative Topical Bromfenac Treatment. Journal of clinical medicine. 2020 Sep 7:9(9):. doi: 10.3390/jcm9092883. Epub 2020 Sep 7 [PubMed PMID: 32906606]
Romano MR, Lograno MD. Evidence for the involvement of cannabinoid CB1 receptors in the bimatoprost-induced contractions on the human isolated ciliary muscle. Investigative ophthalmology & visual science. 2007 Aug:48(8):3677-82 [PubMed PMID: 17652738]
Toris CB, Gabelt BT, Kaufman PL. Update on the mechanism of action of topical prostaglandins for intraocular pressure reduction. Survey of ophthalmology. 2008 Nov:53 Suppl1(SUPPL1):S107-20. doi: 10.1016/j.survophthal.2008.08.010. Epub [PubMed PMID: 19038618]
Level 3 (low-level) evidenceWang JW, Woodward DF, Stamer WD. Differential effects of prostaglandin E2-sensitive receptors on contractility of human ocular cells that regulate conventional outflow. Investigative ophthalmology & visual science. 2013 Jul 16:54(7):4782-90. doi: 10.1167/iovs.13-12363. Epub 2013 Jul 16 [PubMed PMID: 23766471]
Lim KS, Nau CB, O'Byrne MM, Hodge DO, Toris CB, McLaren JW, Johnson DH. Mechanism of action of bimatoprost, latanoprost, and travoprost in healthy subjects. A crossover study. Ophthalmology. 2008 May:115(5):790-795.e4. doi: 10.1016/j.ophtha.2007.07.002. Epub [PubMed PMID: 18452763]
Level 1 (high-level) evidenceStein JD, Khawaja AP, Weizer JS. Glaucoma in Adults-Screening, Diagnosis, and Management: A Review. JAMA. 2021 Jan 12:325(2):164-174. doi: 10.1001/jama.2020.21899. Epub [PubMed PMID: 33433580]
Tang W, Zhang F, Liu K, Duan X. Efficacy and safety of prostaglandin analogues in primary open-angle glaucoma or ocular hypertension patients: A meta-analysis. Medicine. 2019 Jul:98(30):e16597. doi: 10.1097/MD.0000000000016597. Epub [PubMed PMID: 31348303]
Level 1 (high-level) evidenceYildirim N, Sahin A, Gultekin S. The effect of latanoprost, bimatoprost, and travoprost on circadian variation of intraocular pressure in patients with open-angle glaucoma. Journal of glaucoma. 2008 Jan-Feb:17(1):36-9. doi: 10.1097/IJG.0b013e318133fb70. Epub [PubMed PMID: 18303382]
Level 1 (high-level) evidenceCai Z, Cao M, Liu K, Duan X. Analysis of the Responsiveness of Latanoprost, Travoprost, Bimatoprost, and Tafluprost in the Treatment of OAG/OHT Patients. Journal of ophthalmology. 2021:2021():5586719. doi: 10.1155/2021/5586719. Epub 2021 May 25 [PubMed PMID: 34123413]
Law SK, Song BJ, Fang E, Caprioli J. Feasibility and efficacy of a mass switch from latanoprost to bimatoprost in glaucoma patients in a prepaid Health Maintenance Organization. Ophthalmology. 2005 Dec:112(12):2123-30 [PubMed PMID: 16225924]
Level 2 (mid-level) evidenceCurran MP. Bimatoprost: a review of its use in open-angle glaucoma and ocular hypertension. Drugs & aging. 2009:26(12):1049-71. doi: 10.2165/11203210-000000000-00000. Epub [PubMed PMID: 19929032]
Deshpande SS, Sonty S, Ahmad A. Evaluating intraocular pressure-lowering solutions for the treatment of open-angle glaucoma: comparison between bimatoprost 0.03% and bimatoprost 0.01% - an observational switch study. Clinical ophthalmology (Auckland, N.Z.). 2017:11():1371-1376. doi: 10.2147/OPTH.S137537. Epub 2017 Jul 27 [PubMed PMID: 28794615]
Petricca S, Celenza G, Costagliola C, Tranfa F, Iorio R. Cytotoxicity, Mitochondrial Functionality, and Redox Status of Human Conjunctival Cells after Short and Chronic Exposure to Preservative-Free Bimatoprost 0.03% and 0.01%: An In Vitro Comparative Study. International journal of molecular sciences. 2022 Nov 15:23(22):. doi: 10.3390/ijms232214113. Epub 2022 Nov 15 [PubMed PMID: 36430590]
Level 2 (mid-level) evidenceFigus M, Nardi M, Piaggi P, Sartini M, Guidi G, Martini L, Lazzeri S. Bimatoprost 0.01% vs bimatoprost 0.03%: a 12-month prospective trial of clinical and in vivo confocal microscopy in glaucoma patients. Eye (London, England). 2014 Apr:28(4):422-9. doi: 10.1038/eye.2013.304. Epub 2014 Jan 17 [PubMed PMID: 24434659]
Level 1 (high-level) evidencePillunat LE, Eschstruth P, Häsemeyer S, Thelen U, Foja C, Leaback R, Pfennigsdorf S. Preservative-free bimatoprost 0.03% in patients with primary open-angle glaucoma or ocular hypertension in clinical practice. Clinical ophthalmology (Auckland, N.Z.). 2016:10():1759-65. doi: 10.2147/OPTH.S103084. Epub 2016 Sep 12 [PubMed PMID: 27672307]
Mrukwa-Kominek E, Misiuk-Hojlo M, Csutak A, Stalmans I, Garhofer G. A randomized clinical trial comparing three fixed combinations of bimatoprost with timolol in patients with open-angle glaucoma or ocular hypertension. Current medical research and opinion. 2023 May:39(5):775-783. doi: 10.1080/03007995.2023.2197496. Epub 2023 Apr 17 [PubMed PMID: 37013445]
Level 1 (high-level) evidenceXu KM, Cho R, Chan TYB. Retrospective Analysis of Switching Bimatoprost 0.01% to Bimatoprost 0.03% in Patients with Various Types of Glaucoma and Ocular Hypertension. Clinical ophthalmology (Auckland, N.Z.). 2022:16():2385-2390. doi: 10.2147/OPTH.S368214. Epub 2022 Jul 29 [PubMed PMID: 35936971]
Level 2 (mid-level) evidenceFilippelli M, Campagna G, Ciampa N, Fioretto G, Giannini R, Marino PF, dell'Omo R, Costagliola C. Ocular Tolerability of Bimatoprost 0.1 mg/mL Preservative-Free versus Bimatoprost 0.1 mg/mL with Benzalkonium Chloride or Bimatoprost 0.3 mg/mL Preservative-Free in Patients with Primary Open-Angle Glaucoma. Journal of clinical medicine. 2022 Jun 19:11(12):. doi: 10.3390/jcm11123518. Epub 2022 Jun 19 [PubMed PMID: 35743588]
Gedde SJ, Vinod K, Wright MM, Muir KW, Lind JT, Chen PP, Li T, Mansberger SL, American Academy of Ophthalmology Preferred Practice Pattern Glaucoma Panel. Primary Open-Angle Glaucoma Preferred Practice Pattern®. Ophthalmology. 2021 Jan:128(1):P71-P150. doi: 10.1016/j.ophtha.2020.10.022. Epub 2020 Nov 12 [PubMed PMID: 34933745]
Bacharach J, Tatham A, Ferguson G, Belalcázar S, Thieme H, Goodkin ML, Chen MY, Guo Q, Liu J, Robinson MR, Bejanian M, Wirta DL, ARTEMIS 2 Study Group. Phase 3, Randomized, 20-Month Study of the Efficacy and Safety of Bimatoprost Implant in Patients with Open-Angle Glaucoma and Ocular Hypertension (ARTEMIS 2). Drugs. 2021 Nov:81(17):2017-2033. doi: 10.1007/s40265-021-01624-9. Epub 2021 Nov 1 [PubMed PMID: 34724172]
Level 1 (high-level) evidenceBirt CM, Buys YM, Ahmed II, Trope GE, Toronto Area Glaucoma Society. Prostaglandin efficacy and safety study undertaken by race (the PRESSURE study). Journal of glaucoma. 2010 Sep:19(7):460-7. doi: 10.1097/IJG.0b013e3181c4aeac. Epub [PubMed PMID: 20051890]
Level 1 (high-level) evidenceMenon MG, Goodkin ML. Triple Fixed-Combination Bimatoprost/Brimonidine/Timolol in Glaucoma and Ocular Hypertension in India: A Multicenter, Open-Label, Phase 3 Study. Clinical ophthalmology (Auckland, N.Z.). 2022:16():3559-3569. doi: 10.2147/OPTH.S369626. Epub 2022 Oct 20 [PubMed PMID: 36274674]
Lemmens S, Rossetti L, Oddone F, Sunaric-Mégevand G, Hommer A, Vandewalle E, Francesca Cordeiro M, McNaught A, Montesano G, Stalmans I. Comparison of preserved bimatoprost 0.01% with preservative-free tafluprost: A randomised, investigator-masked, 3-month crossover, multicentre trial, SPORT II. European journal of ophthalmology. 2021 Apr 5:():11206721211006573. doi: 10.1177/11206721211006573. Epub 2021 Apr 5 [PubMed PMID: 33818170]
Level 1 (high-level) evidenceBourne RRA, Kaarniranta K, Lorenz K, Traverso CE, Vuorinen J, Ropo A. Changes in ocular signs and symptoms in patients switching from bimatoprost-timolol to tafluprost-timolol eye drops: an open-label phase IV study. BMJ open. 2019 Apr 2:9(4):e024129. doi: 10.1136/bmjopen-2018-024129. Epub 2019 Apr 2 [PubMed PMID: 30944129]
Kim JH, Kim EJ, Kim YH, Kim YI, Lee SH, Jung JC, Lee KW, Park YJ. In Vivo Effects of Preservative-free and Preserved Prostaglandin Analogs: Mouse Ocular Surface Study. Korean journal of ophthalmology : KJO. 2015 Aug:29(4):270-9. doi: 10.3341/kjo.2015.29.4.270. Epub 2015 Jul 21 [PubMed PMID: 26240512]
Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology. 2014 Nov:121(11):2081-90. doi: 10.1016/j.ophtha.2014.05.013. Epub 2014 Jun 26 [PubMed PMID: 24974815]
Level 1 (high-level) evidencePlosker GL, Keam SJ. Bimatoprost: a pharmacoeconomic review of its use in open-angle glaucoma and ocular hypertension. PharmacoEconomics. 2006:24(3):297-314 [PubMed PMID: 16519552]
Level 3 (low-level) evidenceMohan N, Chakrabarti A, Nazm N, Mehta R, Edward DP. Newer advances in medical management of glaucoma. Indian journal of ophthalmology. 2022 Jun:70(6):1920-1930. doi: 10.4103/ijo.IJO_2239_21. Epub [PubMed PMID: 35647957]
Level 3 (low-level) evidenceAlany RG. Adherence, persistence and cost-consequence comparison of bimatoprost topical ocular formulations. Current medical research and opinion. 2013 Sep:29(9):1187-9. doi: 10.1185/03007995.2013.818968. Epub 2013 Jul 5 [PubMed PMID: 23829677]
Level 3 (low-level) evidenceHolmstrom S, Buchholz P, Walt J, Wickstrøm J, Aagren M. The cost-effectiveness of bimatoprost, latanoprost and timolol in treatment of primary open angle glaucoma in five European countries. Current medical research and opinion. 2006 May:22(5):897-905 [PubMed PMID: 16709311]
Level 3 (low-level) evidenceWoodward DF, Carling RW, Cornell CL, Fliri HG, Martos JL, Pettit SN, Liang Y, Wang JW. The pharmacology and therapeutic relevance of endocannabinoid derived cyclo-oxygenase (COX)-2 products. Pharmacology & therapeutics. 2008 Oct:120(1):71-80. doi: 10.1016/j.pharmthera.2008.08.001. Epub 2008 Aug 6 [PubMed PMID: 18700152]
Level 3 (low-level) evidenceTauchi M, Fuchs TA, Kellenberger AJ, Woodward DF, Paus R, Lütjen-Drecoll E. Characterization of an in vivo model for the study of eyelash biology and trichomegaly: mouse eyelash morphology, development, growth cycle, and anagen prolongation by bimatoprost. The British journal of dermatology. 2010 Jun:162(6):1186-97. doi: 10.1111/j.1365-2133.2010.09685.x. Epub 2010 Mar 12 [PubMed PMID: 20346040]
Level 3 (low-level) evidenceJiang S, Hao Z, Qi W, Wang Z, Zhou M, Guo N. The efficacy of topical prostaglandin analogs for hair loss: A systematic review and meta-analysis. Frontiers in medicine. 2023:10():1130623. doi: 10.3389/fmed.2023.1130623. Epub 2023 Mar 14 [PubMed PMID: 36999072]
Level 1 (high-level) evidenceSmith S, Fagien S, Whitcup SM, Ledon F, Somogyi C, Weng E, Beddingfield FC 3rd. Eyelash growth in subjects treated with bimatoprost: a multicenter, randomized, double-masked, vehicle-controlled, parallel-group study. Journal of the American Academy of Dermatology. 2012 May:66(5):801-6. doi: 10.1016/j.jaad.2011.06.005. Epub 2011 Sep 6 [PubMed PMID: 21899919]
Level 1 (high-level) evidenceLaw SK. Bimatoprost in the treatment of eyelash hypotrichosis. Clinical ophthalmology (Auckland, N.Z.). 2010 Apr 26:4():349-58 [PubMed PMID: 20463804]
Carruthers J, Beer K, Carruthers A, Coleman WP 3rd, Draelos ZD, Jones D, Goldman MP, Pucci ML, VanDenburgh A, Weng E, Whitcup SM. Bimatoprost 0.03% for the Treatment of Eyebrow Hypotrichosis. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2016 May:42(5):608-17. doi: 10.1097/DSS.0000000000000755. Epub [PubMed PMID: 27124878]
Yazdanian N, Mozafarpoor S, Goodarzi A. Phosphodiesterase inhibitors and prostaglandin analogues in dermatology: A comprehensive review. Dermatologic therapy. 2021 Jan:34(1):e14669. doi: 10.1111/dth.14669. Epub 2020 Dec 21 [PubMed PMID: 33314552]
Sarsik SM, El-Amawy HS. Uses of eye drops in dermatology, literature review. The Journal of dermatological treatment. 2022 Sep:33(6):2758-2770. doi: 10.1080/09546634.2022.2079598. Epub 2022 Jun 2 [PubMed PMID: 35652324]
Zaher H, Gawdat HI, Hegazy RA, Hassan M. Bimatoprost versus Mometasone Furoate in the Treatment of Scalp Alopecia Areata: A Pilot Study. Dermatology (Basel, Switzerland). 2015:230(4):308-13. doi: 10.1159/000371416. Epub 2015 Mar 4 [PubMed PMID: 25765294]
Level 3 (low-level) evidenceFukumoto T, Fukumoto R, Magno E, Oka M, Nishigori C, Horita N. Treatments for alopecia areata: A systematic review and network meta-analysis. Dermatologic therapy. 2021 May:34(3):e14916. doi: 10.1111/dth.14916. Epub 2021 Mar 4 [PubMed PMID: 33631058]
Level 1 (high-level) evidenceGupta AK, Bamimore MA, Foley KA. Efficacy of non-surgical treatments for androgenetic alopecia in men and women: a systematic review with network meta-analyses, and an assessment of evidence quality. The Journal of dermatological treatment. 2022 Feb:33(1):62-72. doi: 10.1080/09546634.2020.1749547. Epub 2020 Apr 13 [PubMed PMID: 32250713]
Level 1 (high-level) evidenceEasthope SE, Perry CM. Topical bimatoprost: a review of its use in open-angle glaucoma and ocular hypertension. Drugs & aging. 2002:19(3):231-48 [PubMed PMID: 12027782]
Higginbotham EJ, Schuman JS, Goldberg I, Gross RL, VanDenburgh AM, Chen K, Whitcup SM, Bimatoprost Study Groups 1 and 2. One-year, randomized study comparing bimatoprost and timolol in glaucoma and ocular hypertension. Archives of ophthalmology (Chicago, Ill. : 1960). 2002 Oct:120(10):1286-93 [PubMed PMID: 12365906]
Level 1 (high-level) evidenceAli MS, Hafiz HSA, Ahmed NA, Galal SA. Combined microneedling with topical vitamin D3 or bimatoprost versus microneedling alone in the treatment of alopecia areata: A comparative randomized trial. Journal of cosmetic dermatology. 2023 Apr:22(4):1286-1296. doi: 10.1111/jocd.15569. Epub 2023 Feb 10 [PubMed PMID: 36762396]
Level 1 (high-level) evidenceNewman-Casey PA, Robin AL, Blachley T, Farris K, Heisler M, Resnicow K, Lee PP. The Most Common Barriers to Glaucoma Medication Adherence: A Cross-Sectional Survey. Ophthalmology. 2015 Jul:122(7):1308-16. doi: 10.1016/j.ophtha.2015.03.026. Epub 2015 Apr 24 [PubMed PMID: 25912144]
Level 2 (mid-level) evidenceGautam M, Gupta R, Singh P, Verma V, Verma S, Mittal P, Karkhur S, Sampath A, Mohan RR, Sharma B. Intracameral Drug Delivery: A Review of Agents, Indications, and Outcomes. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2023 Mar:39(2):102-116. doi: 10.1089/jop.2022.0144. Epub 2023 Feb 9 [PubMed PMID: 36757304]
Shirley M. Bimatoprost Implant: First Approval. Drugs & aging. 2020 Jun:37(6):457-462. doi: 10.1007/s40266-020-00769-8. Epub [PubMed PMID: 32447639]
Seal JR, Robinson MR, Burke J, Bejanian M, Coote M, Attar M. Intracameral Sustained-Release Bimatoprost Implant Delivers Bimatoprost to Target Tissues with Reduced Drug Exposure to Off-Target Tissues. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2019 Jan/Feb:35(1):50-57. doi: 10.1089/jop.2018.0067. Epub 2018 Oct 18 [PubMed PMID: 30335560]
Medeiros FA, Walters TR, Kolko M, Coote M, Bejanian M, Goodkin ML, Guo Q, Zhang J, Robinson MR, Weinreb RN, ARTEMIS 1 Study Group. Phase 3, Randomized, 20-Month Study of Bimatoprost Implant in Open-Angle Glaucoma and Ocular Hypertension (ARTEMIS 1). Ophthalmology. 2020 Dec:127(12):1627-1641. doi: 10.1016/j.ophtha.2020.06.018. Epub 2020 Jun 13 [PubMed PMID: 32544560]
Level 1 (high-level) evidenceMiller PE, Eaton JS. Medical anti-glaucoma therapy: Beyond the drop. Veterinary ophthalmology. 2021 Mar:24 Suppl 1():2-15. doi: 10.1111/vop.12843. Epub 2020 Nov 9 [PubMed PMID: 33164328]
Sirinek PE, Lin MM. Intracameral sustained release bimatoprost implants (Durysta). Seminars in ophthalmology. 2022 Apr 3:37(3):385-390. doi: 10.1080/08820538.2021.1985145. Epub 2021 Sep 29 [PubMed PMID: 34586961]
Brandt JD, DuBiner HB, Benza R, Sall KN, Walker GA, Semba CP, Collaborators. Long-term Safety and Efficacy of a Sustained-Release Bimatoprost Ocular Ring. Ophthalmology. 2017 Oct:124(10):1565-1566. doi: 10.1016/j.ophtha.2017.04.022. Epub 2017 May 17 [PubMed PMID: 28528010]
Xu J, Ge Y, Bu R, Zhang A, Feng S, Wang J, Gou J, Yin T, He H, Zhang Y, Tang X. Co-delivery of latanoprost and timolol from micelles-laden contact lenses for the treatment of glaucoma. Journal of controlled release : official journal of the Controlled Release Society. 2019 Jul 10:305():18-28. doi: 10.1016/j.jconrel.2019.05.025. Epub 2019 May 16 [PubMed PMID: 31103677]
Aref AA. Sustained drug delivery for glaucoma: current data and future trends. Current opinion in ophthalmology. 2017 Mar:28(2):169-174. doi: 10.1097/ICU.0000000000000334. Epub [PubMed PMID: 27764023]
Level 3 (low-level) evidenceWilliams RD, Cohen JS, Gross RL, Liu CC, Safyan E, Batoosingh AL, Bimatoprost Study Group. Long-term efficacy and safety of bimatoprost for intraocular pressure lowering in glaucoma and ocular hypertension: year 4. The British journal of ophthalmology. 2008 Oct:92(10):1387-92. doi: 10.1136/bjo.2007.128454. Epub 2008 Jul 11 [PubMed PMID: 18621791]
Level 1 (high-level) evidenceJbara D, Eiger-Moscovich M, Didkovsky E, Keshet Y, Avisar I. In Vivo Effects of Prostaglandin Analogues Application by Topical Drops or Retrobulbar Injections on the Orbital Fat of a Rat Model. Ocular immunology and inflammation. 2023 Feb:31(2):298-303. doi: 10.1080/09273948.2022.2026977. Epub 2022 Jan 26 [PubMed PMID: 35081015]
Kucukevcilioglu M, Bayer A, Uysal Y, Altinsoy HI. Prostaglandin associated periorbitopathy in patients using bimatoprost, latanoprost and travoprost. Clinical & experimental ophthalmology. 2014 Mar:42(2):126-31. doi: 10.1111/ceo.12163. Epub 2013 Aug 4 [PubMed PMID: 23844550]
Level 2 (mid-level) evidenceShrirao N, Khurana M, Mukherjee B. Prostaglandin-associated periorbitopathy. Indian journal of ophthalmology. 2016 Jun:64(6):459. doi: 10.4103/0301-4738.187676. Epub [PubMed PMID: 27488155]
Steinsapir KD, Steinsapir SMG. Revisiting the Safety of Prostaglandin Analog Eyelash Growth Products. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2021 May 1:47(5):658-665. doi: 10.1097/DSS.0000000000002928. Epub [PubMed PMID: 33625141]
Eiger-Moscovich M, Stiebel-Kalish H, Yassur I, Barash D, Gaton D, Avisar I. Prostaglandin analogue drops for the treatment of soft tissue expansion and exophthalmos in patients with inactive thyroid eye disease. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 2019 Aug:54(4):426-430. doi: 10.1016/j.jcjo.2018.09.003. Epub 2018 Nov 23 [PubMed PMID: 31358139]
Kroll DM, Schuman JS. Reactivation of herpes simplex virus keratitis after initiating bimatoprost treatment for glaucoma. American journal of ophthalmology. 2002 Mar:133(3):401-3 [PubMed PMID: 11860979]
Level 3 (low-level) evidenceKothari MT, Mehta BK, Asher NS, Kothari KJ. Recurrence of bilateral herpes simplex virus keratitis following bimatoprost use. Indian journal of ophthalmology. 2006 Mar:54(1):47-8 [PubMed PMID: 16531673]
Level 3 (low-level) evidenceWoodward JA, Haggerty CJ, Stinnett SS, Williams ZY. Bimatoprost 0.03% gel for cosmetic eyelash growth and enhancement. Journal of cosmetic dermatology. 2010 Jun:9(2):96-102. doi: 10.1111/j.1473-2165.2010.00487.x. Epub [PubMed PMID: 20618554]
Level 1 (high-level) evidencePacker M, Fine IH, Hoffman RS. Bilateral nongranulomatous anterior uveitis associated with bimatoprost. Journal of cataract and refractive surgery. 2003 Nov:29(11):2242-3 [PubMed PMID: 14670442]
Level 3 (low-level) evidenceCano Parra J, Díaz-Llopis M. [Drug induced uveitis]. Archivos de la Sociedad Espanola de Oftalmologia. 2005 Mar:80(3):137-49 [PubMed PMID: 15803424]
Kapur R, Osmanovic S, Toyran S, Edward DP. Bimatoprost-induced periocular skin hyperpigmentation: histopathological study. Archives of ophthalmology (Chicago, Ill. : 1960). 2005 Nov:123(11):1541-6 [PubMed PMID: 16286616]
Holló G, Aung T, Cantor LB, Aihara M. Cystoid macular edema related to cataract surgery and topical prostaglandin analogs: Mechanism, diagnosis, and management. Survey of ophthalmology. 2020 Sep-Oct:65(5):496-512. doi: 10.1016/j.survophthal.2020.02.004. Epub 2020 Feb 22 [PubMed PMID: 32092363]
Level 3 (low-level) evidenceYeh PC, Ramanathan S. Latanoprost and clinically significant cystoid macular edema after uneventful phacoemulsification with intraocular lens implantation. Journal of cataract and refractive surgery. 2002 Oct:28(10):1814-8 [PubMed PMID: 12388034]
Level 2 (mid-level) evidencePepple KL, Nguyen MH, Pakzad-Vaezi K, Williamson K, Odell N, Lee C, Leveque TK, Van Gelder RN. RESPONSE OF INFLAMMATORY CYSTOID MACULAR EDEMA TO TREATMENT USING ORAL ACETAZOLAMIDE. Retina (Philadelphia, Pa.). 2019 May:39(5):948-955. doi: 10.1097/IAE.0000000000002044. Epub [PubMed PMID: 29346240]
van der Valk R, Webers CA, Schouten JS, Zeegers MP, Hendrikse F, Prins MH. Intraocular pressure-lowering effects of all commonly used glaucoma drugs: a meta-analysis of randomized clinical trials. Ophthalmology. 2005 Jul:112(7):1177-85 [PubMed PMID: 15921747]
Level 1 (high-level) evidenceJamison A, Okafor L, Ullrich K, Schiedler V, Malhotra R. Do Prostaglandin Analogue Lash Lengtheners Cause Eyelid Fat and Volume Loss? Aesthetic surgery journal. 2022 Oct 13:42(11):1241-1249. doi: 10.1093/asj/sjac156. Epub [PubMed PMID: 35700523]
Level 3 (low-level) evidenceBelkin A, Chen T, DeOliveria AR, Johnson SM, Ramulu PY, Buys YM, American Glaucoma Society and the Canadian Glaucoma Society. A Practical Guide to the Pregnant and Breastfeeding Patient with Glaucoma. Ophthalmology. Glaucoma. 2020 Mar-Apr:3(2):79-89. doi: 10.1016/j.ogla.2019.12.004. Epub [PubMed PMID: 32672600]
Blumen-Ohana E, Sellem E. [Pregnancy & glaucoma: SFO-SFG recommendations]. Journal francais d'ophtalmologie. 2020 Jan:43(1):63-66. doi: 10.1016/j.jfo.2019.11.001. Epub 2019 Dec 5 [PubMed PMID: 31813552]
Fagien S, Walt JG, Carruthers J, Cox SE, Wirta D, Weng E, Beddingfield FC 3rd. Patient-reported outcomes of bimatoprost for eyelash growth: results from a randomized, double-masked, vehicle-controlled, parallel-group study. Aesthetic surgery journal. 2013 Aug 1:33(6):789-98. doi: 10.1177/1090820X13495887. Epub 2013 Jul 19 [PubMed PMID: 23873891]
Level 1 (high-level) evidenceTrattler W, Noecker RJ, Earl ML. A multicentre evaluation of the effect of patient education on acceptance of hyperaemia associated with bimatoprost therapy for glaucoma or ocular hypertension. Advances in therapy. 2008 Mar:25(3):179-89. doi: 10.1007/s12325-008-0029-7. Epub [PubMed PMID: 18351298]
Level 1 (high-level) evidence