Introduction
The word atelectasis originates from Greek, combining ateles and ektasis, thus translating to "incomplete expansion." The concept was first introduced by German physician Eduard Jörg in 1832 in his work De morbo pulmonum organico ex respiratione neonatorum imperfecta orto. Atelectasis occurs due to the partial or complete, reversible collapse of the small airways, resulting in impaired exchange of CO2 and O2, causing an intrapulmonary shunt (perfusion without ventilation). The incidence of atelectasis in patients undergoing general anesthesia can be as high as 90%, underscoring its clinical significance.[1]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Atelectasis, the collapse of lung tissue, generally falls into 2 broad types: obstructive (resorptive) or nonobstructive. Both obstructive and nonobstructive atelectasis occur by any single or combination of 3 different pathophysiologic mechanisms: increased pleural pressure, low alveolar pressure, and surfactant impairment.[2][3]
Obstructive (resorptive) atelectasis: results from airway obstructions that prevent air from reaching the alveoli, and air distal to the obstruction is reabsorbed from the nonventilated alveoli, leading to the collapse of the airways.
- Intrathoracic tumors, mucous plugs, and foreign bodies can partially or completely block airways. Children are particularly susceptible due to their less-developed collateral ventilation pathways. Conversely, adults with chronic obstructive pulmonary disease (COPD), who have extensive collateral airways, are less likely to develop obstructive atelectasis.
- A high fraction of inspired oxygen (FiO2) can lead to absorption atelectasis, as oxygen is more rapidly absorbed into the blood than nitrogen, reducing alveolar patency.
Nonobstructive atelectasis: includes types of atelectasis not directly caused by airway obstruction.
- Compression atelectasis is caused by increased external pressure on the lung. Pleural effusions, pneumothorax, abdominal distension, or tumors may cause compression atelectasis. In these cases, the transmural pressure gradient (alveolar pressure minus intrapleural pressure) across the alveolus decreases, leading to alveolar collapse.
- Surfactant-related atelectasis (adhesive atelectasis) occurs due to surfactant impairment. Surfactant reduces alveolar surface tension, and its impairment leads to increased tension and alveolar collapse, as seen in conditions like acute respiratory distress syndrome (ARDS) or neonatal respiratory distress syndrome (RDS).
- Other Subtypes include cicatrization atelectasis from lung scarring (eg, tuberculosis, fibrosis), relaxation atelectasis from loss of lung-to-chest wall contact (eg, pneumothorax), and replacement atelectasis where tumors replace alveoli, typically seen in bronchioalveolar carcinoma.
Special cases
- Postoperative atelectasis occurs within 72 hours of surgery under general anesthesia. This type of atelectasis is often a mix of obstructive and nonobstructive mechanisms.[4]
- Rounded atelectasis is less common, often associated with asbestosis, and involves the folding of atelectatic lung tissue onto the pleura.
- Middle lobe syndrome involves recurrent or fixed atelectasis of the right middle lobe and lingula caused by extraluminal and intraluminal bronchial obstruction, nonobstructive inflammatory processes, or anatomical defects.
Epidemiology
Atelectasis does not preferentially affect either sex. There is also no increased incidence of atelectasis in people with COPD, asthma, or advanced age.[5]
Atelectasis is more common in patients who are immobilized and mechanically ventilated, such as patients undergoing general anesthesia or in the intensive care unit. Atelectasis is more prominent after cardiac surgery with cardiopulmonary bypass than after other types of surgery, including thoracotomies; however, patients undergoing abdominal and thoracic procedures are at increased risk of developing atelectasis.[4] Additionally, patients with obesity and those who are pregnant are more likely to develop atelectasis due to a decreased functional residual capacity (FRC).
Pathophysiology
Administration of general anesthesia, use of muscle relaxants, obesity, pregnancy, inadequate pain control, and thoracic or cardiopulmonary procedures increase the risk of developing atelectasis in the perioperative period.
The incidence of atelectasis in patients undergoing general anesthesia is 90%.[1] Research has shown that atelectasis appears in the dependent regions of both lungs within five minutes of induction of anesthesia, and up to 15% to 20% of the lung at its base collapses during uneventful anesthesia before any surgical intervention.[6] Atelectasis is seen with general anesthesia regardless of whether or not muscle paralysis is used. Ketamine, when used as a sole agent, is the only anesthetic agent that does not increase the risk of developing atelectasis.[6] High FiO2 during induction and maintenance of general anesthesia also contributes to atelectasis via absorption atelectasis.
FRC is reduced in all patients under general anesthesia and in those in the supine position. This reduction leads to compressive atelectasis in dependent areas of the lungs.
Inadequate pain control can contribute to the development of atelectasis by inducing shallow breathing or splinting. On the other hand, the use of opioids for pain control leads to respiratory depression and inhibition of coughing, also leading to atelectasis.[7]
History and Physical
Typically, atelectasis is asymptomatic if only a few alveoli are affected. As more alveoli become involved, however, the individual may report dyspnea, cough with or without sputum production, and pleuritic chest pain. The physical examination of a patient with atelectasis may reveal tachypnea, increased work of breathing, diminished or absent breath sounds, crackles, and decreased chest expansion.
Evaluation
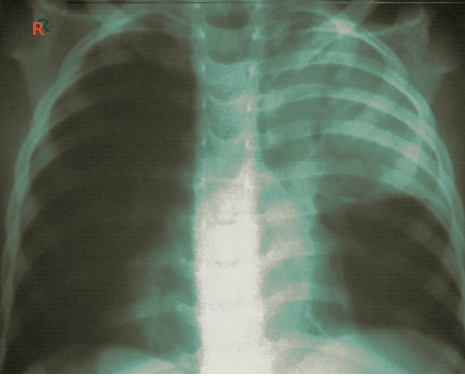
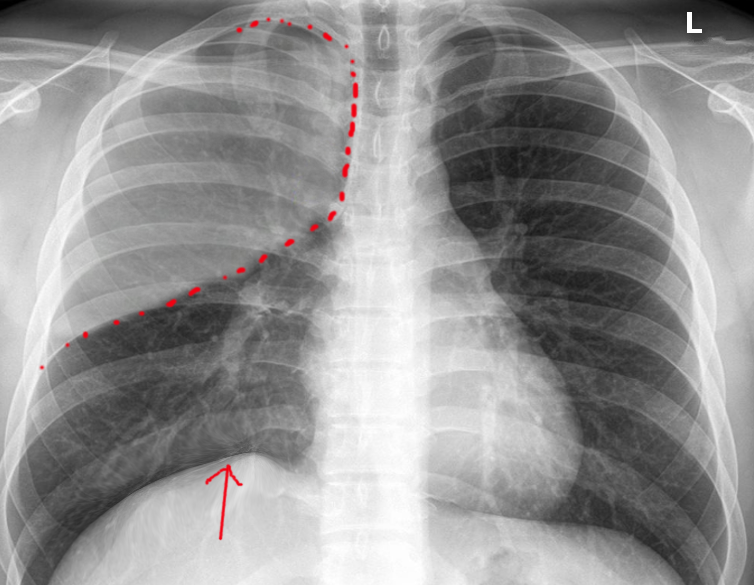
Atelectasis is usually a clinical diagnosis in a patient with known risk factors. If imaging is warranted, chest radiography, chest computed tomography, or thoracic ultrasonography are useful when diagnosing atelectasis. A chest x-ray will reveal platelike, horizontal lines in the area of atelectatic lung tissue and may result in the displacement of interlobar fissures, pulmonary opacification, or tracheal shift toward the affected side.[8]. Atelectasis is not typically evident on conventional chest radiographs until it is significant.
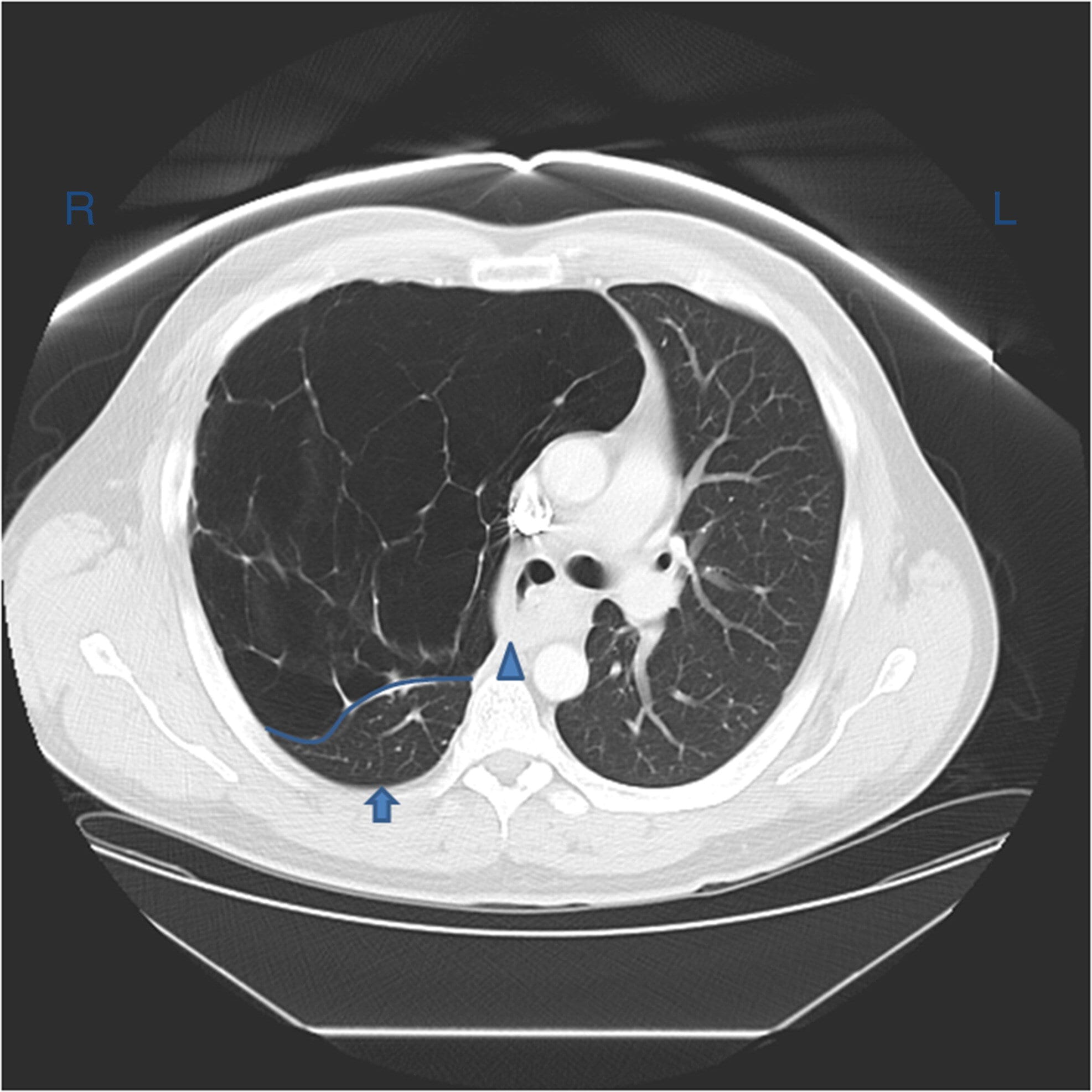
Computed tomography of the chest performed in patients with atelectasis often reveals dependent lung densities and loss of volume in the affected side of the chest.
Atelectasis may also be directly visible with fiberoptic bronchoscopy. Fiberoptic bronchoscopy can be both diagnostic and therapeutic, often revealing the cause of any obstruction contributing to the atelectasis, such as a tumor, mucous plug, or foreign body.
An arterial blood gas may reveal arterial hypoxemia and respiratory alkalosis. The PaCO2 is often normal but may be decreased secondary to increased minute ventilation, which often accompanies atelectasis.
Treatment / Management
Most atelectasis that appears during general anesthesia leads to transient lung dysfunction that resolves within 24 hours after surgery. Nevertheless, some patients develop significant perioperative respiratory complications that can lead to increased morbidity and mortality if not treated. Atelectasis is preventable through avoidance of general anesthesia, early mobilization, adequate pain control, and minimizing parenteral opioid administration. When general anesthesia is unavoidable, continuous positive airway pressure, the lowest possible FiO2 during induction and maintenance, positive end-expiratory pressure (PEEP), lung recruitment maneuvers, and low tidal volumes of 6 to 10 mL/kg will help prevent the development of atelectasis.[7][9] One study showed that intraoperative alveolar recruitment with a vital capacity maneuver followed by PEEP 10 cm H2O is effective at preventing lung atelectasis in patients with morbid obesity; this also correlated with better oxygenation, shorter postanesthesia care unit (PACU) stay, and fewer pulmonary complications in the postoperative period.[10](A1)
Changing position from supine to upright increases FRC and decreases atelectasis.[11] Encouraging patients to take deep breaths, early ambulation, incentive spirometry, use of an acapella device, chest physiotherapy, tracheal suctioning in intubated patients, and positive pressure ventilation have been shown to decrease atelectasis. The mechanism behind these measures is a transient increase in transmural pressure that allows for the reexpansion of collapsed lung segments. Prophylactic measures, such as incentive spirometry, should be taught and instituted before surgery and continued hourly following surgery until discharge to obtain the maximal benefit.[7]
Mucolytic agents, like N-acetylcysteine, have been tried with limited evidence that they prevent postoperative atelectasis. However, in certain populations, such as pediatric patients and those with cystic fibrosis, recombinant human dornase alpha may help alleviate atelectasis due to mucus plugging.[7][12] (B2)
Fiberoptic bronchoscopy also plays a role in the management of atelectasis. In one study, single-suction fiberoptic bronchoscopy improved lung function and reversed atelectasis in 76% of cases. Bronchoscopy should always be the intervention when there is a high suspicion of a mechanically obstructed bronchus and coughing and suctioning have not been successful. Bronchoscopy is also indicated when less invasive efforts, such as early ambulation, incentive spirometry, bronchodilators, and humidity, prove unsuccessful. Employing early preventative strategies and valuing prompt recognition of patients with atelectasis will improve patient outcomes and significantly decrease healthcare costs.[13]
Differential Diagnosis
The differential diagnosis of atelectasis should include:
- Neoplasm
- Pneumonia
- Pleural effusion
- Pulmonary embolism
- Foreign body.
Prognosis
For patients with atelectasis, the prognosis varies greatly, and the primary prognostic determinators are the underlying etiology and patient comorbidities. Untreated atelectasis can result in lung infections, including recurrent pneumonia. Atelectasis may result in impaired oxygen exchange, causing hypoxemia and respiratory distress. With time, recurrent instances of atelectasis may result in the formation of lung scarring or fibrosis and decrease overall lung reserve. Early detection and appropriate management are crucial to prevent these potentially serious complications.
Complications
Atelectasis is one of the most common respiratory complications in the perioperative period, and may contribute to significant morbidity and mortality, including the development of pneumonia and acute respiratory failure.
Postoperative and Rehabilitation Care
Postoperative fever has historically been attributed to atelectasis, but there is no evidence supporting the finding that atelectasis is a causative mechanism for fever.[14] A systematic search by Mavros et al included 8 eligible studies involving 998 patients who underwent cardiac, abdominal, and maxillofacial surgery. The authors found a poor association between atelectasis and early postoperative fever. However, this study was limited by significant heterogeneity, and further large-scale studies are required.[14]
Deterrence and Patient Education
The definition of atelectasis is a partial collapse of the lung. It can cause people to feel short of breath. It can be a consequence of several different processes, most commonly when there is a poor inspiratory effort, an obstruction blocking airflow into the lung, extra pressure exerted on the outside of the lung, or deficient production or function of a specific protein in the lung. Treatment aims to address the underlying cause of atelectasis but mainly involves supportive measures, such as deep breathing exercises, incentive spirometry, and supplemental O2.
Patients should be advised to seek immediate medical attention if they are short of breath. Atelectasis is a common postoperative complication. Deep breathing exercises and devices to assist with clearing mucus and secretions can help expand the lungs and improve ventilation. Smoking can be harmful, and patients must be advised against smoking to promote lung healing.
Enhancing Healthcare Team Outcomes
Preventing atelectasis is vital to improving patient outcomes in the postoperative period. However, despite employing these strategies, atelectasis is not always preventable. Therefore, early recognition and treatment are equally important. This will decrease the length of hospital stay and cost and improve patient outcomes.
Both prevention and treatment of atelectasis need to be an interprofessional team effort. Physicians, especially surgeons and anesthesiologists, need to be aware of the role of anesthesia in atelectasis. The nursing staff will monitor the patient during and after the procedure. In the event of medical management, the pharmacist can provide recommendations on opioids and mucolytics. The nursing staff will administer these medications and can report to the physicians on the effectiveness of therapy and any adverse events that may lead to dose or agent changes or other interventions. The nursing staff should assist the clinicians in educating the patient and family in incentive spirometry and other techniques to minimize risk. In summary, atelectasis management needs to be an interprofessional team collaboration to optimize patient outcomes.
Media
(Click Image to Enlarge)
References
Lundquist H, Hedenstierna G, Strandberg A, Tokics L, Brismar B. CT-assessment of dependent lung densities in man during general anaesthesia. Acta radiologica (Stockholm, Sweden : 1987). 1995 Nov:36(6):626-32 [PubMed PMID: 8519574]
Peroni DG, Boner AL. Atelectasis: mechanisms, diagnosis and management. Paediatric respiratory reviews. 2000 Sep:1(3):274-8 [PubMed PMID: 12531090]
Zeng C, Lagier D, Lee JW, Vidal Melo MF. Perioperative Pulmonary Atelectasis: Part I. Biology and Mechanisms. Anesthesiology. 2022 Jan 1:136(1):181-205. doi: 10.1097/ALN.0000000000003943. Epub [PubMed PMID: 34499087]
Magnusson L, Spahn DR. New concepts of atelectasis during general anaesthesia. British journal of anaesthesia. 2003 Jul:91(1):61-72 [PubMed PMID: 12821566]
Gunnarsson L, Tokics L, Gustavsson H, Hedenstierna G. Influence of age on atelectasis formation and gas exchange impairment during general anaesthesia. British journal of anaesthesia. 1991 Apr:66(4):423-32 [PubMed PMID: 2025468]
Tokics L, Hedenstierna G, Strandberg A, Brismar B, Lundquist H. Lung collapse and gas exchange during general anesthesia: effects of spontaneous breathing, muscle paralysis, and positive end-expiratory pressure. Anesthesiology. 1987 Feb:66(2):157-67 [PubMed PMID: 3813078]
Lagier D, Zeng C, Fernandez-Bustamante A, Vidal Melo MF. Perioperative Pulmonary Atelectasis: Part II. Clinical Implications. Anesthesiology. 2022 Jan 1:136(1):206-236. doi: 10.1097/ALN.0000000000004009. Epub [PubMed PMID: 34710217]
Woodring JH, Reed JC. Types and mechanisms of pulmonary atelectasis. Journal of thoracic imaging. 1996 Spring:11(2):92-108 [PubMed PMID: 8820021]
Hartland BL, Newell TJ, Damico N. Alveolar recruitment maneuvers under general anesthesia: a systematic review of the literature. Respiratory care. 2015 Apr:60(4):609-20. doi: 10.4187/respcare.03488. Epub 2014 Nov 25 [PubMed PMID: 25425708]
Level 1 (high-level) evidenceTalab HF, Zabani IA, Abdelrahman HS, Bukhari WL, Mamoun I, Ashour MA, Sadeq BB, El Sayed SI. Intraoperative ventilatory strategies for prevention of pulmonary atelectasis in obese patients undergoing laparoscopic bariatric surgery. Anesthesia and analgesia. 2009 Nov:109(5):1511-6. doi: 10.1213/ANE.0b013e3181ba7945. Epub [PubMed PMID: 19843790]
Level 1 (high-level) evidenceCraig DB, Wahba WM, Don HF, Couture JG, Becklake MR. "Closing volume" and its relationship to gas exchange in seated and supine positions. Journal of applied physiology. 1971 Nov:31(5):717-21 [PubMed PMID: 5117187]
den Hollander B, Linssen RSN, Cortjens B, van Etten-Jamaludin FS, van Woensel JBM, Bem RA, Dutch Collaborative PICU Research Network. Use of dornase alfa in the paediatric intensive care unit: current literature and a national cross-sectional survey. European journal of hospital pharmacy : science and practice. 2022 May:29(3):123-128. doi: 10.1136/ejhpharm-2020-002507. Epub 2020 Oct 29 [PubMed PMID: 33122405]
Level 2 (mid-level) evidenceRestrepo RD, Braverman J. Current challenges in the recognition, prevention and treatment of perioperative pulmonary atelectasis. Expert review of respiratory medicine. 2015 Feb:9(1):97-107. doi: 10.1586/17476348.2015.996134. Epub 2014 Dec 26 [PubMed PMID: 25541220]
Mavros MN, Velmahos GC, Falagas ME. Atelectasis as a cause of postoperative fever: where is the clinical evidence? Chest. 2011 Aug:140(2):418-424. doi: 10.1378/chest.11-0127. Epub 2011 Apr 28 [PubMed PMID: 21527508]