Introduction
Congenital malformations of the uterus are the consequence of an anomaly in combination, canalization, and resorption of the septum during the development of Mullerian ducts. Buttram and Gibbons initially classified Mullerian duct anomalies depending on the degree of failure of Mullerian duct development in 1979. This classification was received and revised by the American Society of Reproductive Medicine in 1988, and they classified Mullerian duct anomalies into seven classes.
- Class I- Agenesis/hypoplasia
- Class II- Unicornuate uterus, IIa with a rudimentary communicating horn; IIb with a rudimentary non-communicating horn; II c with a rudimentary horn without a cavity: II d without a rudimentary horn
- Class III- Didelphys uterus
- Class IV- Bicornuate uterus( IV a is complete, IV b is partial)
- Class V- Septate uterus
- Class VI- Arcuate uterus
- Class VII- Diethylstilbestrol-related anomalies.[1]
American Society of Reproductive Medicine further reorganized the classification in 2016 and included arcuate uterus and healthy uterus in a single class.[2]
A bicornuate uterus is a result of a partial fusion of Mullerian ducts resulting in a heart-shaped uterus instead of a pear shape. It is further segregated into two types depending upon the partition of the cervix:
- Bicornuate unicollis
- Bicornuate bicollis
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
The etiology behind variation in the fusion of Mullerian ducts is still unclear though it could be multifactorial comprising of genetic as well as environmental factors. Studies have linked the uterine anomalies to an abnormality in gene expression. Several genes, such as Pax, Lim1, Emx2, Wnt4, Wnt9b, are involved in the development of Mullerian ducts during morphogenesis. A subset of homeobox genes, along with Wnt7a, regulates the differentiation of Mullerian ducts. Wnt7a is responsible for the expression of Hoxa 10 and Hoxa 11 genes. This segmental expression of Hoxa 9, 10, 11, and 13 along the Mullerian duct in a linear pattern is essential for the differentiation of ducts into fallopian tubes, uterus, cervix, and vagina.[3] HOXA13 mutations have also been found in hand-foot-genital syndrome, the characteristic features of this condition are limb malformations along with urogenital abnormalities, including Mullerian fusion anomalies.
The environmental factors can also be responsible; for instance, the development of T shaped uterus (class VII of the American Society of Reproductive Medicine) is attributed to the exposure of diethylstilbestrol during pregnancy, especially during pregnancies in the 1940s to the 1970s. Nonetheless, the incidence of this disease is decreasing as the drug is no longer in use now.[4]
Epidemiology
A survey of 94 observational studies was done to determine the pervasiveness of congenital anomalies in the general population in contrast to the women with a history of infertility or miscarriages. It reports an 8.0% prevalence of uterine abnormalities among women with infertility, 13.3% in women with a background of miscarriage, 24.5% in those with miscarriage and infertility. The most widely recognized anomaly is arcuate uterus in the general population and septate uterus among those in the populace with infertility and miscarriage. The bicornuate uterus is 0.4% prevalent in the general population, 1.1% in women with infertility, 2.1 % in those with miscarriage, and 4.7% in women with infertility along with miscarriage.[5]
Pathophysiology
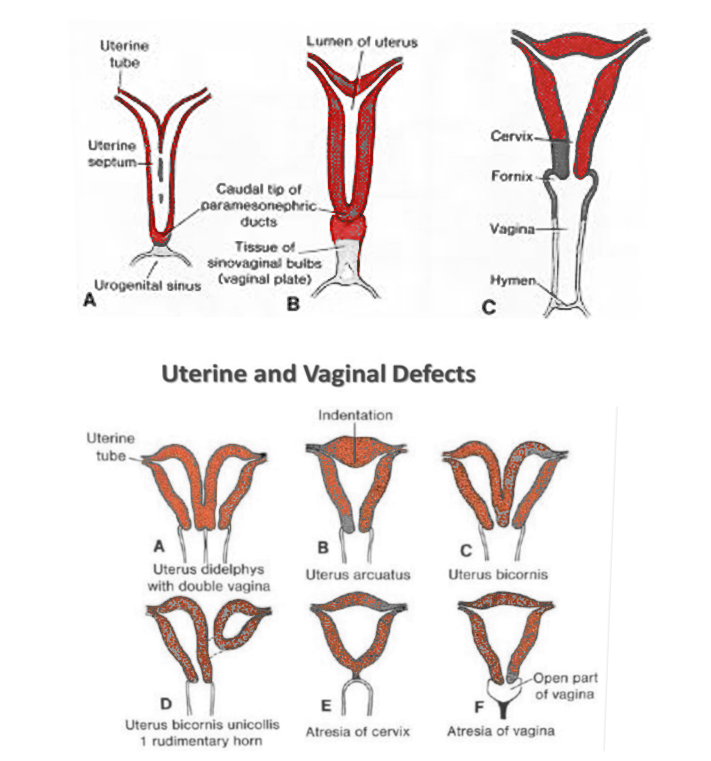
Two genital ducts lead to the formation of the female genital tract, one is the mesonephric duct/Wolffian duct, and the other is the paramesonephric duct/ Mullerian duct. The entire procedure happens in three phases-
- Morphogenesis- The development of a mesonephric duct occurs at around the sixth week of gestation. Paramesonephric duct is formed in the seventh-week as an invagination in celomic mesothelium in the upper lateral portion of the intermediate cell mass.[6]
- Differentiation- SRY gene situated on the Y chromosome produces the antimullerian hormone, which is liable for the relapse of the Mullerian duct in males. The Mullerian duct of both sides grows in a caudal direction parallel to the mesonephric duct. These ducts then cross the Wolffian conduit and fuse in the midline in their caudal end. The central combined portion frames the uterus, and the lower part of the fused portion forms the upper third of the vagina. The cranial portion of the Mullerian ducts stay unfused and gives rise to oviducts in the future.[7]
- Resorption of the septum- The uterus assumes its shape at around 12 weeks; however, the middle fused portion of the ducts is persisting. During the third stage, this central septum gets resorbed, transforming the uterovaginal canal into a single cavity.[8]
The lower fifth of the vagina has its origin from the endoderm of urogenital sinus instead of Mullerian ducts. The caudal tip of fused Mullerian tubes is called Mullerian tubercle. This tubercle interacts with urogenital sinus and prompts the proliferation of endodermal cells of the urogenital sinus. These are called sinovaginal bulbs, which, along with the uterovaginal canal, form a vaginal plate. This plate canalizes to frame the vaginal canal.[4]
If there is impedance during the second stage-fusion of Mullerian ducts leading to partial fusion of the ducts, it results in the bicornuate uterus. This combination can vary. On the off chance that the outcome is a solitary vagina yet separate cervix with separate uterine cavities, it is called bicornuate bicollis. However, it is termed as bicornuate unicollis uterus; the uterine cavities are discrete, but the cervix and vagina are single.
The origins of ovaries are from the genital ridge and are independent of the Mullerian ducts; ovaries are generally not engaged in Mullerian duct anomalies.[9]
History and Physical
A bicornuate uterus can exist as an isolated anomaly or in association with complex uterine and genital malformations. An isolated bicornuate uterus is associated with significant reproductive complications. A meta-analysis study of 25 comparative studies reported a significantly increased risk of
- First and second-trimester loss of pregnancy.
- Preterm delivery <28 weeks.
- Low birth weight babies.
- Malpresentation at delivery.[10]
Most of the patients with bicornuate uterus do not have any symptoms in their adolescence. Some women may present to the clinic with menorrhagia or dysmenorrhea owing to the presence of two uterine cavities. A few women are also diagnosed when they present for routine evaluation during pregnancy. A significant number of women are diagnosed when they present with obstetric complications, as mentioned beforehand. Physical exam in isolated anomalies is usually insignificant.
A longitudinal vaginal septum exists in 25% of cases associated with a bicornuate uterus, which may lead to obstructive symptoms or dyspareunia. The patient may present with an inability to control menstrual bleeding with tampon in such cases.[11] The physical exam reveals a vaginal septum, which on removal exposes a double cervix and uterus.
Renal anomalies are frequently found in affiliation with Mullerian anomalies due to the interlinked development of mesonephric and Mullerian ducts along with urogenital sinus. The most common defect is renal agenesis associated with didelphys uterus. However, it can also present along with the bicornuate uterus. An ectopic ureter can also be found.[12]
Bicornuate uterus with non-communicating uterine cavities, associated with renal agenesis and blind hemivagina, present with acute urinary retention, pelvic pain, and dysmenorrhea. On a physical exam, a bulge can be found in the vagina, making it challenging to explore the cervix.[13]
In another variant, a communicating bicornuate uterus can exist with renal agenesis and hemivagina. In such cases, patients present with a Gartner duct pseudocyst. On physical exam, a cyst is present in the anterolateral wall of the vagina, which indeed is blind hemivagina. A bicornuate uterus can also exist with the non-communicating uterine horn. Such patients can present with infertility in consequence of endometriosis due to retrograde menstruation.[14]
Evaluation
Imaging plays an essential role in the diagnosis and management of bicornuate uterus. There are multiple modalities available for this purpose which are as follows-
- Hysterosalpingography- This is the most seasoned and most broadly utilized method for assessment of an instance of infertility. During hysterosalpingography, an oil or water-based contrast is infused through an endocervical catheter into the uterine cavity. This contrast opacifies the uterus and will portray the endometrial orifice of the uterus. Under fluoroscopic direction, different pictures of the uterus and uterine tubes are taken, which can help in surveying the endometrial cavity, patency of the fallopian tubes, and sophisticated communications with Mullerian anomalies. While applying to the conclusion of bicornuate uterus, the intercornual angle is estimated. If two horns of uterus are evident with an intercornual point more noteworthy than 105 degrees, it is named as the bicornuate uterus.[15] However, the utilization of hysterosalpingography is restricted in such conditions as it can't survey the serosal surface of the uterus, which is essential to differentiate septate uterus from the bicornuate uterus. Its application is additionally constrained within sight of the vaginal septum, prompting obstructive Mullerian variations from the norm. The contrast can't enter the blocked uterine cavity, in this way misclassifying twofold cavity irregularities into single cavity anomalies.[8]
- Ultrasound-This is the mainline imaging performed when a patient first visits the facility; it is reasonable and noninvasive with no introduction to radiation. 2D ultrasonography is accessible at each training; however, 3D ultrasonography is increasingly precise and reliable. 3D ultrasonography has a sensitivity of 99% and specificity of 100% in the separation of the bicornuate uterus from the septate uterus. Another examination done saw 3D ultrasonography as profoundly dependable. A progression of 2D pictures is taken during ultrasonography to make a 3D volume of the uterus. Coronal views are the most significant as they give data about the endometrial pit alongside the serosal surface of the uterus. The serosal surface appraisal assumes a remarkable job in the separation of the septate uterus from the bicornuate uterus. Z strategy is the most dominating method used to acquire midcoronal views.[16] The ultrasound should be done during the secretory phase of the menstrual cycle as the endometrium is thick and echogenic, which can help separate bicornuate uterus accurately.[15] After getting a coronal view of the uterus, draw a line from the interstitial portion of one fallopian tube to the other. Then another perpendicular line is drawn from this line to the point of maximum indentation of the uterus. If the indent is >10 mm, it is bicornuate; however, with an indent of <10 mm, the uterus is classified as arcuate/subseptate.[16] The confinement of ultrasound lies in that it is a profoundly modern procedure and requires a specialist supposition. Another impediment is in its powerlessness to distinguish rudimentary horns of the uterus or unicornuate uterus.
- MRI- MRI It is the best quality level for the conclusion of the bicornuate uterus because of its noninvasiveness, absence of radiations, and producing pictures in numerous planes. It is equipped for giving data about the uterine structure and its fundal surface, the anatomy of the vagina, cervix, along the entire pelvis. A precise methodology has been formulated for differential diagnosis of Mullerian peculiarities in MRI. The initial phase in this procedure is to decide whether Mullerian hypoplasia is available. With hypoplasia, the differential can be Mayer Rokitansky Kuster syndrome or unicornuate uterus. If no hypoplasia is seen, the subsequent stage is an evaluation of the uterine fundal form. On the off chance that a split more than10 mm is found in the fundus, it is a fusion anomaly (bicornuate/didelphys uterus). Be that as it may, with parted less than 10 mm, it focuses on a resorption peculiarity (septate/arcuate uterus). Further separation among bicornuate and didelphys uterus is finished by taking a look at the tissue between two horns. If it is available, the uterus is bicornuate, while its nonattendance focuses on didelphys uterus. Twofold cervix shows up as owl eyes on MRI in bicornuate bicollis uterus.[17]
Treatment / Management
The decision to manage a patient with the bicornuate uterus is pertinent to the presentation of the patient. A patient can present to a facility in 2 different ways:
If a woman presents for a routine evaluation during her pregnancy gets diagnosed with a bicornuate uterus, then aggressive prenatal monitoring is indicated to prevent obstetric complications. Pay attention to the signs of preterm labor, malpresentation.[11](B3)
A patient can additionally present with a history of recurrent abortions or preterm labor in preceding pregnancies. The presentation mentioned above is an indication for the surgical unification of uterus, Strassman metroplasty.[11] The procedure was first illustrated in 1907 by Strassman in 4 stages. The procedure initiates by making a transverse incision over the fundus of the uterus, staying away from uterotubal junctions to avoid injury. Subsequently, the uterine cavity is opened, and the septum is removed after splitting the partition. Thus the procedure transforms the double cavity into a single cavity. Ultimately the cavity is closed by vertical suturing to prevent endometrial adhesions.[18](B3)
The laparoscopic approach is being preferred in the present in place of abdominal metroplasty. The laparoscopic approach provides leverage in terms of less bleeding and decreased rate of infections. It also reports significantly reduced postoperative adhesion formation, which can be credited to a decrease in tissue handling and drying of tissues.[19](B2)
Differential Diagnosis
- A bicornuate uterus is with a longitudinal vaginal septum in some cases, which makes it difficult to differentiate from uterus didelphys. In such cases, look for the presence of soft tissue between 2 uterine cavities, which establishes the diagnosis of a bicornuate uterus.[17]
- It is challenging to distinguish bicornuate uterus from the septate uterus on hysterosalpingography. The differentiation of bicornuate from the septate uterus is critical due to the contrast in their management approach. A septate uterus is managed via hysteroscopic resection as opposed to bicornuate, which requires a unification of uterus. In such a case, MRI helps differentiate fusion anomalies from resorption anomalies.[14]
Prognosis
A bicornuate uterus is associated with adverse reproductive outcomes like preterm labor, recurrent abortions, malpresentation.
Strassman metroplasty is indicated in women with recurrent abortions. In a prospective study done over seven years, open Strassman's metroplasty reports improvement in the fetal viability from 0 % to 80%.[20]
Laparoscopic metroplasty has now replaced abdominal metroplasty in most places. A case series performed on patients post-laparoscopic metroplasty reported an 85% pregnancy achievement rate. Seven women with bicornuate uterus carried the pregnancy from twelve weeks to the term.Laparoscopic metroplasty is also associated with a decrease in uterine adhesions and increased uterus compliance, thus decreasing the chances of rupture.[19]
Complications
Patients with a bicornuate uterus can present with several unfortunate complications.
The most common complication associated with the bicornuate uterus is preterm labor. A cervical length of less than 25 mm on transvaginal ultrasound has 13 times higher risk of preterm delivery.[21] Cervical cerclage is an effective method to prevent a non-viable preterm delivery in women.[22]
A pregnancy in a bicornuate uterus can also be succeeded by postpartum hemorrhage. Postpartum hemorrhage is manageable by an assortment of surgical and nonsurgical alternatives. The nonsurgical methodology of utilizing Bakri Balloon is proved to be viable in the bicornuate uterus. However, the surgical approach, for instance, B lynch suturing, and conduit ligation can harm the neighboring structures. Moreover, ligation may not be effective with the collateral blood supply of uterus in pregnancy.[23]
A bicornuate uterus is a risk factor for rupture of the uterus even in a primigravida. The explanation could be an abnormal development of the lower fragment of the uterus or the presence of a fibrous band between the corpora of the uterus. This band restrains the uterus, unable to expand, and hence gets inclined to rupture.[24]
Due to the association of uterine anomalies with renal anomalies, a woman is at high risk for pregnancy-induced hypertension during her gestational period. Thus it is essential to monitor blood pressure during pregnancy in a woman with bicornuate uterus.
Although bicornuate uterus is not an independent risk factor for endometrial cancer but cancer in endometrium can go undetected. If taken from the healthy uterine cavity, a biopsy can give false-negative results, leading to a delay in diagnosis and worsening the prognosis of the patient. MRI can play an essential role in diagnosing the disease if a patient with bicornuate uterus presents with uterine bleeding.[25]
Deterrence and Patient Education
A bicornuate uterus is a rare oddity; it is fundamental to educate a woman diagnosed with this anomaly. A vast majority of the women are diagnosed during gestation, so it is crucial to counsel them about reproductive outcomes. Along with aggressive prenatal monitoring, she should be educated about the signs of preterm labor. Cautioning her about uterine rupture during labor is additionally fundamental. Malpresentations being very common during pregnancy in the double uterus, she should be advised to prepare for a cesarean section in the future.
Enhancing Healthcare Team Outcomes
A primary care provider should maintain a high suspicion for anomalies in adolescents who exhibit menorrhagia with dysmenorrhea. This can lead to early diagnosis of the abnormality and, thus, better reproductive outcomes in the future.
Management of bicornuate uterus requires clear differentiation of this anomaly from septate and didelphys uterus. Only an experienced radiologist with knowledge about embryology of the uterine anomalies and different associations can make a definite diagnosis.
A woman with a bicornuate uterus can present to the clinic with recurrent pregnancy loss. As a rare anomaly, the role of a gynecologist is to increase the patient's awareness regarding her diagnosis and make her aware of the linked complications. Comprehensive counseling of women diagnosed with the bicornuate uterus is very crucial. Women who experience recurrent pregnancy loss can suffer from anxiety or depression. The psychiatrist plays an essential position in such a circumstance.
Media
(Click Image to Enlarge)
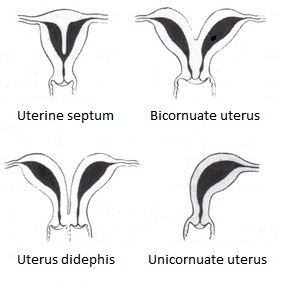
Types of Bicornuate Uterus Malformation. A uterine malformation is a type of female genital malformation resulting from abnormal Müllerian duct(s) development during embryogenesis. The prevalence of uterine malformation is estimated to be 6%-7% in the human female population. They may cause premature birth. Didelphis and bicornuate uterus are the normal anatomy in some mammals, just not in humans.
EternamenteAprendiz, Public Domain, via Wikimedia Commons.
References
. The American Fertility Society classifications of adnexal adhesions, distal tubal occlusion, tubal occlusion secondary to tubal ligation, tubal pregnancies, müllerian anomalies and intrauterine adhesions. Fertility and sterility. 1988 Jun:49(6):944-55 [PubMed PMID: 3371491]
Ludwin A, Pfeifer SM. Reproductive surgery for müllerian anomalies: a review of progress in the last decade. Fertility and sterility. 2019 Sep:112(3):408-416. doi: 10.1016/j.fertnstert.2019.07.005. Epub [PubMed PMID: 31446900]
Roly ZY, Backhouse B, Cutting A, Tan TY, Sinclair AH, Ayers KL, Major AT, Smith CA. The cell biology and molecular genetics of Müllerian duct development. Wiley interdisciplinary reviews. Developmental biology. 2018 May:7(3):e310. doi: 10.1002/wdev.310. Epub 2018 Jan 19 [PubMed PMID: 29350886]
Passos IMPE, Britto RL. Diagnosis and treatment of müllerian malformations. Taiwanese journal of obstetrics & gynecology. 2020 Mar:59(2):183-188. doi: 10.1016/j.tjog.2020.01.003. Epub [PubMed PMID: 32127135]
Chan YY, Jayaprakasan K, Zamora J, Thornton JG, Raine-Fenning N, Coomarasamy A. The prevalence of congenital uterine anomalies in unselected and high-risk populations: a systematic review. Human reproduction update. 2011 Nov-Dec:17(6):761-71. doi: 10.1093/humupd/dmr028. Epub 2011 Jun 24 [PubMed PMID: 21705770]
Level 1 (high-level) evidenceMoncada-Madrazo M, Rodríguez Valero C. Embryology, Uterus. StatPearls. 2023 Jan:(): [PubMed PMID: 31613528]
Rackow BW, Arici A. Reproductive performance of women with müllerian anomalies. Current opinion in obstetrics & gynecology. 2007 Jun:19(3):229-37 [PubMed PMID: 17495638]
Level 3 (low-level) evidenceRobbins JB, Broadwell C, Chow LC, Parry JP, Sadowski EA. Müllerian duct anomalies: embryological development, classification, and MRI assessment. Journal of magnetic resonance imaging : JMRI. 2015 Jan:41(1):1-12. doi: 10.1002/jmri.24771. Epub 2014 Oct 7 [PubMed PMID: 25288098]
Pizzo A, Laganà AS, Sturlese E, Retto G, Retto A, De Dominici R, Puzzolo D. Mayer-rokitansky-kuster-hauser syndrome: embryology, genetics and clinical and surgical treatment. ISRN obstetrics and gynecology. 2013:2013():628717. doi: 10.1155/2013/628717. Epub 2013 Feb 4 [PubMed PMID: 23431465]
Venetis CA, Papadopoulos SP, Campo R, Gordts S, Tarlatzis BC, Grimbizis GF. Clinical implications of congenital uterine anomalies: a meta-analysis of comparative studies. Reproductive biomedicine online. 2014 Dec:29(6):665-83. doi: 10.1016/j.rbmo.2014.09.006. Epub 2014 Sep 21 [PubMed PMID: 25444500]
Level 1 (high-level) evidenceLetterie GS. Management of congenital uterine abnormalities. Reproductive biomedicine online. 2011 Jul:23(1):40-52. doi: 10.1016/j.rbmo.2011.02.008. Epub 2011 Feb 17 [PubMed PMID: 21652266]
Level 3 (low-level) evidenceHall-Craggs MA, Kirkham A, Creighton SM. Renal and urological abnormalities occurring with Mullerian anomalies. Journal of pediatric urology. 2013 Feb:9(1):27-32. doi: 10.1016/j.jpurol.2011.11.003. Epub 2011 Nov 29 [PubMed PMID: 22129802]
Dietrich JE, Millar DM, Quint EH. Obstructive reproductive tract anomalies. Journal of pediatric and adolescent gynecology. 2014 Dec:27(6):396-402. doi: 10.1016/j.jpag.2014.09.001. Epub 2014 Sep 11 [PubMed PMID: 25438708]
Level 1 (high-level) evidenceAcién P, Acién M. Diagnostic imaging and cataloguing of female genital malformations. Insights into imaging. 2016 Oct:7(5):713-26. doi: 10.1007/s13244-016-0515-4. Epub 2016 Aug 9 [PubMed PMID: 27507534]
Behr SC, Courtier JL, Qayyum A. Imaging of müllerian duct anomalies. Radiographics : a review publication of the Radiological Society of North America, Inc. 2012 Oct:32(6):E233-50. doi: 10.1148/rg.326125515. Epub [PubMed PMID: 23065173]
Deutch TD, Abuhamad AZ. The role of 3-dimensional ultrasonography and magnetic resonance imaging in the diagnosis of müllerian duct anomalies: a review of the literature. Journal of ultrasound in medicine : official journal of the American Institute of Ultrasound in Medicine. 2008 Mar:27(3):413-23 [PubMed PMID: 18314520]
Yoo RE, Cho JY, Kim SY, Kim SH. A systematic approach to the magnetic resonance imaging-based differential diagnosis of congenital Müllerian duct anomalies and their mimics. Abdominal imaging. 2015 Jan:40(1):192-206. doi: 10.1007/s00261-014-0195-9. Epub [PubMed PMID: 25070770]
Level 1 (high-level) evidenceStrassmann EO. Fertility and unification of double uterus. Fertility and sterility. 1966 Mar-Apr:17(2):165-76 [PubMed PMID: 5907041]
Alborzi S, Asefjah H, Amini M, Vafaei H, Madadi G, Chubak N, Tavana Z. Laparoscopic metroplasty in bicornuate and didelphic uteri: feasibility and outcome. Archives of gynecology and obstetrics. 2015 May:291(5):1167-71. doi: 10.1007/s00404-014-3520-1. Epub 2014 Nov 6 [PubMed PMID: 25373708]
Level 2 (mid-level) evidenceRechberger T, Monist M, Bartuzi A. Clinical effectiveness of Strassman operation in the treatment of bicornuate uterus. Ginekologia polska. 2009 Feb:80(2):88-92 [PubMed PMID: 19338203]
Airoldi J, Berghella V, Sehdev H, Ludmir J. Transvaginal ultrasonography of the cervix to predict preterm birth in women with uterine anomalies. Obstetrics and gynecology. 2005 Sep:106(3):553-6 [PubMed PMID: 16135586]
Yassaee F, Mostafaee L. The role of cervical cerclage in pregnancy outcome in women with uterine anomaly. Journal of reproduction & infertility. 2011 Oct:12(4):277-9 [PubMed PMID: 23926514]
Abraham C. Bakri balloon placement in the successful management of postpartum hemorrhage in a bicornuate uterus: A case report. International journal of surgery case reports. 2017:31():218-220. doi: 10.1016/j.ijscr.2017.01.055. Epub 2017 Jan 24 [PubMed PMID: 28189983]
Level 3 (low-level) evidenceNitzsche B, Dwiggins M, Catt S. Uterine rupture in a primigravid patient with an unscarred bicornuate uterus at term. Case reports in women's health. 2017 Jul:15():1-2. doi: 10.1016/j.crwh.2017.03.004. Epub 2017 Mar 28 [PubMed PMID: 29593991]
Level 3 (low-level) evidenceGao J, Zhang J, Tian W, Teng F, Zhang H, Zhang X, Wang Y, Xue F. Endometrial cancer with congenital uterine anomalies: 3 case reports and a literature review. Cancer biology & therapy. 2017 Mar 4:18(3):123-131. doi: 10.1080/15384047.2017.1281495. Epub 2017 Jan 24 [PubMed PMID: 28118070]
Level 3 (low-level) evidence