Introduction
Monitoring the depth of anesthesia is very important during any procedure. Anesthesia that's too deep can cause hemodynamic changes, and anesthesia that is too light carries the risk of recall or awareness during anesthesia.[1] Awareness during anesthesia is a very serious complication with potential long-term psychological sequelae such as anxiety and posttraumatic stress disorder.[2] When the bispectral index (BIS) value is maintained below 60, it decreases the incidence of anesthesia awareness.[2] Several neuro monitors based on the processed electroencephalogram or evoked potentials have been developed to evaluate the anesthetic depth. The BIS monitor is the first FDA-approved method to assess the hypnotic effects of drugs.[3]
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
The BIS represents a significant development in the objective assessment of the depth of anesthesia, providing valuable real-time feedback to anesthesia providers.[4]
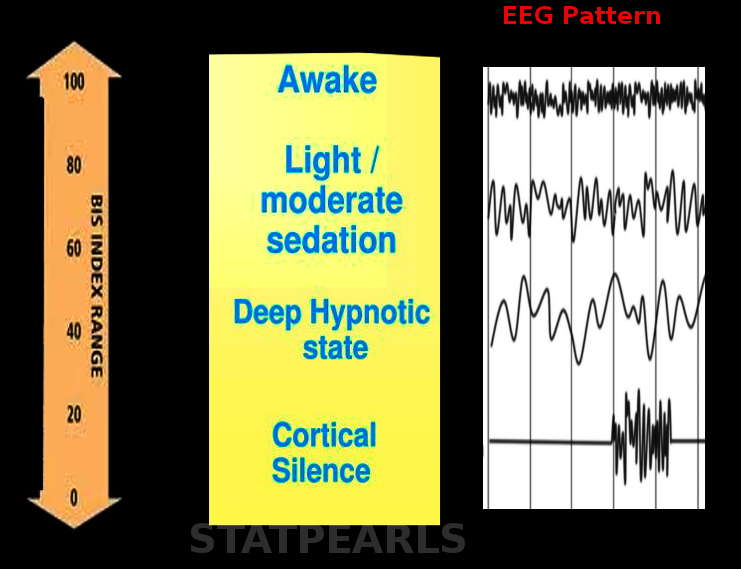
The BIS is a dimensionless number that ranges from 0, indicative of complete brain inactivity, to 100, indicative of an awake and alert patient. The number is obtained by analyzing the electroencephalogram (EEG) of patients undergoing anesthesia. The BIS device employs an algorithm to analyze the EEG waveform, considering factors such as frequency, phase, and power spectrum to generate the BIS value. BIS monitoring provides the anesthesia provider with an objective assessment of the patient's level of consciousness, improving the ability to tailor anesthetic delivery and avoid unnecessary deep anesthesia or inadvertent awareness under anesthesia (see Image. Bispectral Index).[4]
While the physiological principles of the EEG underpin the utility of the BIS, the BIS algorithm's complex mathematical manipulation presents inherent time delays in response to anesthesia induction and changes in anesthetic depth.[4] In an observational study, the authors noted a delay of up to 104 seconds in BIS response to anesthetic induction.[4] This delay may complicate the interpretation of BIS values and has potential implications for anesthetic management, warranting further study and consideration in clinical practice.
The BIS monitoring system is used in anesthesiology to assess the depth of anesthesia. It relies on power concentration in the 40 to 47 Hz frequency range, which correlates with the "traveling peak" theory of anesthetic action. Gamma waves, associated with cognitive function, memory, and information processing, are affected by anesthetics and contribute to the BIS score. The electromyogram (EMG) frequency response overlap with the gamma band explains why neuromuscular blockade decreases the BIS score even in awake individuals. The concentration of power and Wiener entropy, inversely proportional in the EEG dataset, are used to estimate information content and distribution in the frequency spectrum.[5]
Indications
One significant application of BIS is its role in preventing intraoperative awareness, a traumatic event characterized by the patient's consciousness during anesthesia. In a systematic review, authors found evidence suggesting that BIS-guided anesthesia may reduce the risk of intraoperative awareness compared to standard practice without BIS monitoring.[6] By providing an objective measure of anesthetic depth, BIS monitoring allows clinicians to minimize the likelihood of this distressing event.
Another area of BIS application concerns postoperative cognitive dysfunction (POCD) and postoperative delirium (POD). Other authors performed a systematic review and meta-analysis exploring the effect of anesthetic depth on the risk of POCD and POD in high-risk patients.[7] Their findings indicated that using BIS to guide anesthesia depth potentially decreases the incidence of POD and POCD in these patients. It suggests that BIS monitoring may allow for more balanced anesthesia, preventing the adverse cognitive outcomes associated with excessive anesthetic depth.
Furthermore, BIS monitoring may also facilitate early postoperative recovery. A Cochrane review found that using BIS for anesthetic depth guidance may lead to faster emergence from anesthesia and shortened stay in the recovery room.[6] This application demonstrates the potential of BIS to enhance resource utilization in the healthcare system.
The BIS presents a sophisticated tool that adds significant value to anesthetic management. Providing a real-time, objective measure of anesthetic depth can enhance patient safety by reducing the risk of intraoperative awareness, mitigating the risk of postoperative cognitive complications, and expediting postoperative recovery. Nevertheless, its application is not without challenges. Clinicians must be mindful of the inherent time delay in BIS response to anesthesia changes, ensuring optimal anesthetic care. Future studies should continue to refine our understanding of BIS and its application, thereby enhancing its potential to improve anesthesia outcomes and patient experience.
A recent meta-analysis of a total of 26 randomized controlled trials with 10,743 patients showed that based on the depth of anesthesia monitoring, deep anesthesia was associated with lower pain scores at rest within the first hour postoperatively but increased the incidence of postoperative delirium.[8] A meta-analysis of 15 studies involving 5,392 patients revealed that aiming for relatively high BIS was associated with reduced POD and POCD at 3 months; however, no significant differences were found in the length of stay or mortality.[7][9]
Equipment
The BIS monitor processes EEG signals to obtain a value reflecting the patient's consciousness level. It collects this data through its sensors and uses an algorithm to analyze and interpret it. The data displays as a number on the BIS view monitor and ranges from 0 to 100. A value of 0 represents the absence of brain activity, and 100 represents an awake state. Values less than 40 represent a deep hypnotic state. BIS values between 40 and 60 represent adequate general anesthesia for surgery and prevent awareness under anesthesia.
Technique or Treatment
BIS monitoring involves the application of 4 electrodes on the forehead. The skin on the forehead is first cleaned with an alcohol swab, and then 2 to 5 seconds of digital pressure is applied over the sensor leads.[10] The sensor is comprised of disposable wet gel electrodes. EMG activity of the frontalis muscle is measured by lead 4, which is the ground electrode as well. The 2-channel monitor includes a user-configurable display. The 4-channel monitor has enhanced bihemispheric capabilities. BIS-extended sensors are available for use in ICU where patients require long-term monitoring. To measure brain activity in the pediatric population, a BIS pediatric sensor is available, which utilizes 'zipper' technology and can capture low voltage EEG.
The BIS view monitor displays:
- BIS number
- Trend graph of BIS values over time
- Raw EEG waveforms in real-time
- Various signal quality indicators such as signal-quality-index (SQI) and EMG
- Alarm indicators and messages
The SQI bar predicts the reliability of the signal; the higher the SQI, the more reliable the BIS number. The EMG bar indicates EMG activity, which reflects muscle stimulation caused by increased muscle tone or movement.[11]
The BIS monitoring technique uses a specialized sensor attached to the patient's forehead and temple. This sensor records the brain's electrical activity via EEG, with the obtained data analyzed by a proprietary algorithm that calculates the BIS value.[4] This BIS value is subsequently displayed on a monitor, providing real-time information regarding the depth of anesthesia.
While the BIS system is designed to be user-friendly, certain factors may interfere with its accuracy. For example, using EMG can cause an increase in BIS values, thereby leading to misinterpretation of the patient's level of consciousness. Therefore, proper application and securing of the BIS sensor are vital to ensure accurate readings. Additionally, certain medications, such as ketamine and nitrous oxide, can increase BIS values despite causing significant levels of sedation. This highlights the necessity of interpreting BIS values within the overall clinical context, using it as an adjunct rather than the sole guide to anesthetic depth.[4]
Complications
Limitations and Factors Affecting BIS Monitoring
- Anesthetic agents: The anesthetic agents used affect BIS values. The patient anesthetized with 1 anesthetic drug may be more sedated than another patient with the same score anesthetized with a different combination of drugs. BIS monitor is unreliable with certain anesthetics, such as ketamine and nitrous oxide. In 2017, Mishra et al studied the effect of nitrous oxide on the bispectral index and found that the BIS value increases with the addition of nitrous oxide. This effect could be due to the decrease in the suppressant effect of the inhalational anesthetic agents on EEG with the use of nitrous oxide and its neuro-stimulant properties. Nitrous oxide increases cerebral blood flow velocities along with an increase in cerebral consumption of oxygen.[12]
- Age: Titrating anesthetic agents in infants younger than 6 months with BIS is challenging. This could be due to a difference in EEG in this population from older children as brain maturation and synapse formation occur during that period. In 2001, Bannister et al found no significant differences between the standard practice and BIS groups in anesthetic use or recovery measures in children aged 6 months to 3 years.[13]
- Hypothermia: Doi et al studied 12 patients undergoing hypothermia for a cardiopulmonary bypass (CPB) pump and noticed a wide variation in BIS values during the decrease in body temperature.[14] The BIS decreases by 1.12 units for each degree Celsius reduction in body temperature. Reduction in temperature results in a reduced cerebral metabolic rate for oxygen, reflected on the EEG by the progression to an isoelectric/burst suppression pattern dominated by isoelectric periods. Aortic cannulation causes EEG slowing. The onset of CPB produces a transient EEG depression, most likely due to perfusion of the brain with a crystalloid prime solution. EEG slowing that may persist into the post-CPB period is seen during decreased CPB flow rates with the aortic cross-clamp released. Reduced CPB flow rates requested by the surgeon during aortic cross-clamp release often correlate with EEG slowing that may persist into the post-CPB period.[15]
- Neurological impairment: Neurological impairment alters the ability of the BIS to monitor the depth of consciousness; therefore, it is not a reliable tool to assess the level of consciousness in this patient population. Generally, BIS values are lower in patients with neurological impairment. BIS values reflect the activity of the cortical structure of the brain but do not reflect the activity of subcortical structures such as the spinal cord.
- Interference with medical devices: Interference from medical devices causes artifacts and impairs the ability of the BIS monitor to assess the changes in the depth of anesthesia accurately. Chan et al found that SQI decreases during the use of electrosurgical cautery.[16]
Clinical Significance
Alternative technologies to monitor the depth of anesthesia are as follows:[17]
- SNAP EEG monitor system
- Auditory evoked potential monitor
- 4-channel processed EEG
- EEG monitor
- Spectral edge frequency 95
- Automated responsiveness test
While the BIS is a widely recognized tool for monitoring the depth of anesthesia, several alternative methods have been developed to serve similar clinical needs. Each alternative exhibits distinct advantages and drawbacks, thus requiring judicious selection and use in specific clinical contexts.
Spectral entropy monitoring is 1 such alternative. Spectral entropy, like BIS, utilizes EEG data to provide 2 indices: response entropy (RE) and state entropy (SE). RE incorporates both EEG and EMG data, providing an understanding of both cortical and muscular activity, while SE uses only EEG data to represent cortical activity specifically. As a result, spectral entropy can potentially offer a more nuanced understanding of the patient's consciousness state.[18]
Another alternative to BIS is the Narcotrend index. This monitor also uses EEG data but with a different algorithm. It classifies the anesthetic depth into 6 stages, from A (awake) to F (general anesthesia with a high probability of burst suppression). The Narcotrend index has demonstrated potential in guiding anesthetic delivery and predicting intraoperative awareness, although further studies are needed to establish its precise role in anesthetic practice.[18]
The patient state index (PSI) is an additional EEG-derived index developed to assess anesthetic depth. PSI operates on a scale of 0 to 100, with lower numbers reflecting deeper levels of anesthesia. A study suggested that PSI might provide an adequate estimation of anesthetic depth and was relatively stable in response to surgical stimulation.[18]
Finally, the A-line autoregressive index (AAI) is another technique for evaluating anesthetic depth. It uses auditory evoked potentials (AEP) rather than EEG data. AEPs reflect the electrical activity of the auditory pathways and cortex following auditory stimulation. Research has indicated that AEP-derived indices like the AAI can be useful in anesthetic management, providing an alternative to EEG-based techniques.[18]
In conclusion, while the BIS remains a valuable tool in assessing the depth of anesthesia, many viable alternatives are available. Each approach has unique strengths and weaknesses, and the choice of technique will depend on the specific clinical context, highlighting the need for continued research and education in this area.
Enhancing Healthcare Team Outcomes
BIS monitoring is used in operating rooms, ICUs, and some emergency departments. In 2007, Carlos et al found that the time between extubating and discharging from the operating room and the time in the PACU are reduced using BIS monitoring. The risk of postoperative nausea and vomiting was decreased by 12% in patients monitored with BIS.[19] Tong et al, in 2017, demonstrated that the addition of BIS monitoring could result in economic benefits by reducing the use of propofol and faster recovery compared to standard practice.[20]
So far, no gold standard exists to span the entire spectrum of anesthetic effects on the central nervous system. BIS is 1 of the most studied monitors derived from EEG used today. BIS has been shown to have a favorable cost-benefit ratio and lower morbidity than more invasive monitoring methods.[21] This technology requires an interprofessional team approach that includes the surgeon, anesthesiologist, nurse anesthetist, surgical nurses, and other support staff to ensure the best possible patient outcomes.[21] Nurses assist in patient care, anesthesia providers monitor BIS numbers and make informed adjustments, and pharmacists ensure optimal drug dosing for anesthesia depth. By harmonizing these aspects, healthcare professionals create a cohesive team, optimize anesthesia management, reduce complications, and advance patient-centered care, ultimately yielding improved outcomes, safety, and overall team performance.
Media
References
Froese L, Dian J, Gomez A, Batson C, Sainbhi AS, Zeiler FA. Association Between Processed Electroencephalogram-Based Objectively Measured Depth of Sedation and Cerebrovascular Response: A Systematic Scoping Overview of the Human and Animal Literature. Frontiers in neurology. 2021:12():692207. doi: 10.3389/fneur.2021.692207. Epub 2021 Aug 16 [PubMed PMID: 34484100]
Level 1 (high-level) evidenceAvidan MS, Zhang L, Burnside BA, Finkel KJ, Searleman AC, Selvidge JA, Saager L, Turner MS, Rao S, Bottros M, Hantler C, Jacobsohn E, Evers AS. Anesthesia awareness and the bispectral index. The New England journal of medicine. 2008 Mar 13:358(11):1097-108. doi: 10.1056/NEJMoa0707361. Epub [PubMed PMID: 18337600]
Level 1 (high-level) evidenceGe SJ, Zhuang XL, Wang YT, Wang ZD, Li HT. Changes in the rapidly extracted auditory evoked potentials index and the bispectral index during sedation induced by propofol or midazolam under epidural block. British journal of anaesthesia. 2002 Aug:89(2):260-4 [PubMed PMID: 12378664]
Ferreira AL, Mendes JG, Nunes CS, Amorim P. [Evaluation of Bispectral Index time delay in response to anesthesia induction: an observational study]. Brazilian journal of anesthesiology (Elsevier). 2019 Jul-Aug:69(4):377-382. doi: 10.1016/j.bjan.2019.03.008. Epub 2019 Jul 29 [PubMed PMID: 31371175]
Level 2 (mid-level) evidenceConnor CW. Open Reimplementation of the BIS Algorithms for Depth of Anesthesia. Anesthesia and analgesia. 2022 Oct 1:135(4):855-864. doi: 10.1213/ANE.0000000000006119. Epub 2022 Jun 27 [PubMed PMID: 35767469]
Lewis SR, Pritchard MW, Fawcett LJ, Punjasawadwong Y. Bispectral index for improving intraoperative awareness and early postoperative recovery in adults. The Cochrane database of systematic reviews. 2019 Sep 26:9(9):CD003843. doi: 10.1002/14651858.CD003843.pub4. Epub 2019 Sep 26 [PubMed PMID: 31557307]
Level 1 (high-level) evidenceLing L, Yang TX, Lee SWK. Effect of Anaesthesia Depth on Postoperative Delirium and Postoperative Cognitive Dysfunction in High-Risk Patients: A Systematic Review and Meta-Analysis. Cureus. 2022 Oct:14(10):e30120. doi: 10.7759/cureus.30120. Epub 2022 Oct 10 [PubMed PMID: 36381936]
Level 1 (high-level) evidenceLong Y, Feng X, Liu H, Shan X, Ji F, Peng K. Effects of anesthetic depth on postoperative pain and delirium: a meta-analysis of randomized controlled trials with trial sequential analysis. Chinese medical journal. 2022 Dec 5:135(23):2805-2814. doi: 10.1097/CM9.0000000000002449. Epub 2022 Dec 5 [PubMed PMID: 36728598]
Level 1 (high-level) evidenceSumner M, Deng C, Evered L, Frampton C, Leslie K, Short T, Campbell D. Processed electroencephalography-guided general anaesthesia to reduce postoperative delirium: a systematic review and meta-analysis. British journal of anaesthesia. 2023 Feb:130(2):e243-e253. doi: 10.1016/j.bja.2022.01.006. Epub 2022 Feb 17 [PubMed PMID: 35183345]
Level 1 (high-level) evidenceJohansen JW. Update on bispectral index monitoring. Best practice & research. Clinical anaesthesiology. 2006 Mar:20(1):81-99 [PubMed PMID: 16634416]
Olson DM, Chioffi SM, Macy GE, Meek LG, Cook HA. Potential benefits of bispectral index monitoring in critical care. A case study. Critical care nurse. 2003 Aug:23(4):45-52 [PubMed PMID: 12961782]
Level 3 (low-level) evidenceMishra RK, Mahajan C, Prabhakar H, Kapoor I, Bithal PK. Effect of nitrous oxide on bispectral index values at equi-minimum alveolar concentrations of sevoflurane and desflurane. Indian journal of anaesthesia. 2017 Jun:61(6):482-485. doi: 10.4103/ija.IJA_363_16. Epub [PubMed PMID: 28655953]
Bannister CF, Brosius KK, Sigl JC, Meyer BJ, Sebel PS. The effect of bispectral index monitoring on anesthetic use and recovery in children anesthetized with sevoflurane in nitrous oxide. Anesthesia and analgesia. 2001 Apr:92(4):877-81 [PubMed PMID: 11273918]
Level 1 (high-level) evidenceDoi M, Gajraj RJ, Mantzaridis H, Kenny GN. Effects of cardiopulmonary bypass and hypothermia on electroencephalographic variables. Anaesthesia. 1997 Nov:52(11):1048-55 [PubMed PMID: 9404165]
Mathew JP, Weatherwax KJ, East CJ, White WD, Reves JG. Bispectral analysis during cardiopulmonary bypass: the effect of hypothermia on the hypnotic state. Journal of clinical anesthesia. 2001 Jun:13(4):301-5 [PubMed PMID: 11435056]
Chan MT, Ho SS, Gin T. Performance of the bispectral index during electrocautery. Journal of neurosurgical anesthesiology. 2012 Jan:24(1):9-13. doi: 10.1097/ANA.0b013e31823058bf. Epub [PubMed PMID: 21946766]
Medical Advisory Secretariat. Bispectral index monitor: an evidence-based analysis. Ontario health technology assessment series. 2004:4(9):1-70 [PubMed PMID: 23074459]
McCulloch TJ, Sanders RD. Depth of anaesthesia monitoring: time to reject the index? British journal of anaesthesia. 2023 Aug:131(2):196-199. doi: 10.1016/j.bja.2023.04.016. Epub 2023 May 15 [PubMed PMID: 37198033]
Oliveira CR, Bernardo WM, Nunes VM. Benefit of general anesthesia monitored by bispectral index compared with monitoring guided only by clinical parameters. Systematic review and meta-analysis. Brazilian journal of anesthesiology (Elsevier). 2017 Jan-Feb:67(1):72-84. doi: 10.1016/j.bjane.2015.09.001. Epub 2016 Apr 14 [PubMed PMID: 28017174]
Level 1 (high-level) evidenceGan TJ, Glass PS, Windsor A, Payne F, Rosow C, Sebel P, Manberg P. Bispectral index monitoring allows faster emergence and improved recovery from propofol, alfentanil, and nitrous oxide anesthesia. BIS Utility Study Group. Anesthesiology. 1997 Oct:87(4):808-15 [PubMed PMID: 9357882]
Level 1 (high-level) evidenceBard JW. The BIS monitor: a review and technology assessment. AANA journal. 2001 Dec:69(6):477-83 [PubMed PMID: 11837151]