Introduction
The intricacy of the coronary venous system highlights both the complexity and beauty of the human heart's anatomical features. As biomedical science and technology advance, coronary veins have gained significant clinical importance for treatment and intervention for cardiac patients globally. Coronary veins are responsible for draining deoxygenated blood from the myocardium into the cardiac chambers. Comprised of two venous systems, coronary veins classify into either the greater cardiac venous system or the smaller cardiac venous system. The greater cardiac venous system drains the majority of the deoxygenated blood, while the smaller cardiac venous system drains a smaller portion of the deoxygenated blood to its respective heart chambers. Physiological variability of coronary veins also exists among a diverse patient population. Gaining a comprehensive understanding of the anatomy of the coronary venous vasculature is thus vital to enhance clinical knowledge and aid informed physician decision-making for future cardiovascular patients.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
The primary physiological function of the coronary veins is to carry deoxygenated blood from the myocardium and empty them into the chambers of the heart. Coronary veins can be organized into two groups: the greater and smaller cardiac venous system.
Greater Cardiac Venous System
The greater cardiac venous system returns three-quarters of the deoxygenated blood from the myocardium to the cardiac chambers. The main venous vasculature of this system includes the coronary sinus and its tributaries, marginal veins, anterior cardiac veins, ventricular veins, and atrial veins.[1][2]
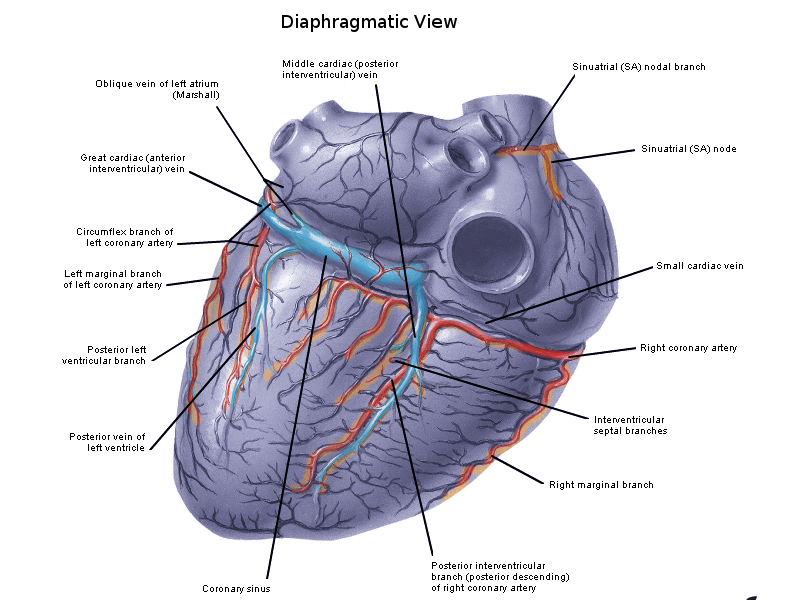
Coronary Sinus
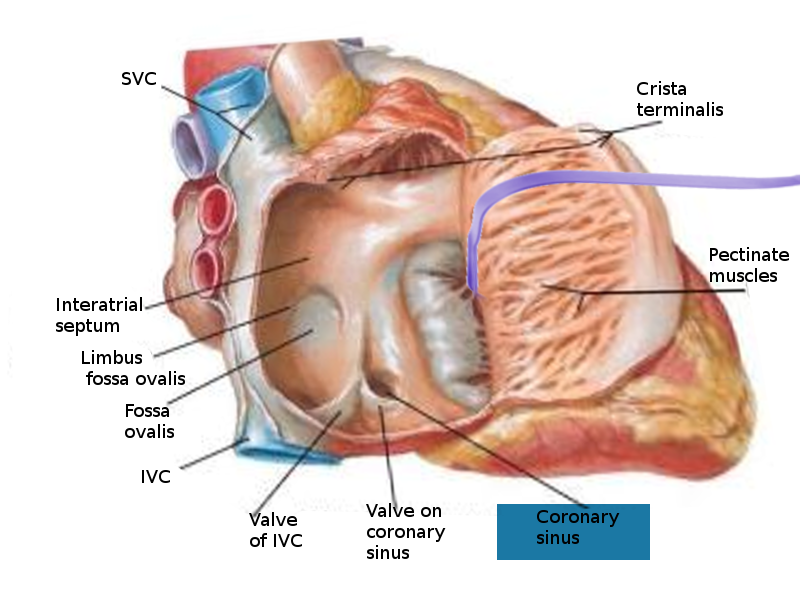
The coronary sinus is the largest cardiac vein with multiple smaller vessels converging into it. Located along the left posterior atrioventricular groove, the coronary sinus empties directly into the right atrium through the coronary sinus orifice. The venous tributaries that merge into the coronary sinus include the great cardiac vein, middle cardiac vein, small cardiac vein, left posterior ventricular, and oblique cardiac vein.[2][3]
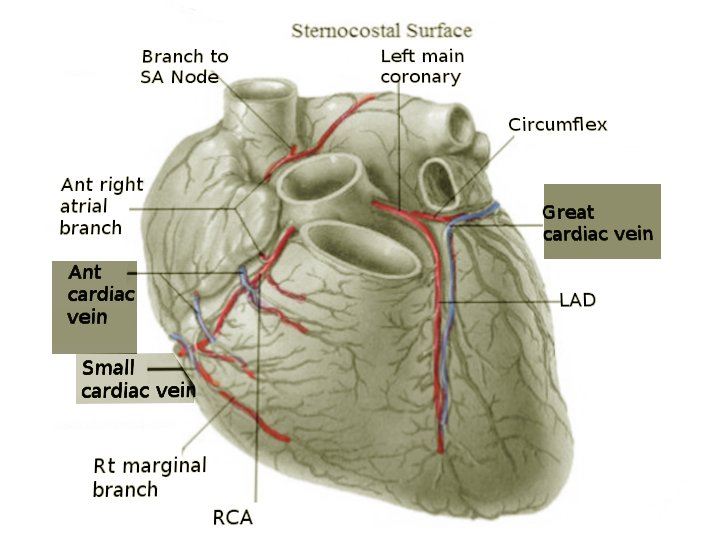
Great Cardiac Vein
Starting from the apex of the heart and running parallel with the anterior interventricular artery, the great cardiac vein travels up along the anterior interventricular sulcus towards the base of the left atrium’s auricle[2]. Once the vein reaches the left margin of the heart, it circumvents to the posterior side and eventually converges with the oblique vein of the left atrium into the coronary sinus. The great cardiac vein empties the blood from both ventricles and the left atrium into the right atrium.[3][4]
Middle Cardiac Vein
The middle cardiac vein originates from the apex of the heart but on the posterior side. The middle cardiac vein travels alongside the posterior interventricular artery in the posterior interventricular sulcus, and empties deoxygenated blood into the coronary sinus.[3]
Small Cardiac Vein
Located in the coronary sulcus between the right atrium and right ventricle, the small cardiac vein eventually coalesces with the coronary sinus on the posterior side of the heart and drains to the right atrium.[3] The small cardiac vein also runs parallel to the right marginal branch of the right coronary artery on the anterior surface of the heart and collects deoxygenated blood from the posterior side of the right chambers.[5]
Right Marginal Vein
The right marginal vein is located on the very inferior margin of the anterior surface of the heart and serves to empty the anterior and diaphragmatic walls of the right ventricle.[3] This small vein is also known for merging with the small cardiac vein in the coronary sulcus.[5]
Anterior Cardiac Veins
Anterior cardiac veins return blood from the anterior right ventricle and drain them directly into the anterior right atrium. The anterior cardiac veins are more superior to the right marginal vein.[4]
Left Marginal Vein
Also known as the obtuse marginal vein, the left marginal vein drains the myocardium of the left ventricle.[2] This vein travels along the left oblique marginal surface of the heart.
Left and Right Ventricular Veins
The left and right ventricular veins vary from person to person, but generally, they function to drain the external walls of the ventricles[6].
Left and Right Atrial Veins
The left atrial veins consist of the septal veins, posterolateral veins, and posterosuperior veins that all carry the deoxygenated blood from the left atrium into the right atrium. The right atrial veins drain the right atrial walls.[7]
Inferior Vein of the Left Ventricle
Also known as the posterior vein of the left ventricle, the inferior left ventricular vein returns the deoxygenated blood from the inferior and lateral walls of the left ventricle into the coronary sinus. This vein travels between the middle and great cardiac veins.[5]
Oblique Vein of the Left Atrium
The oblique vein of the left atrium is a relatively small-sized vein that rounds obliquely and inferiorly towards the posterior left atrium. The vein is also observed to unite with the great cardiac vein to merge with the distal coronary sinus.[4]
Smaller Cardiac Venous System
The smaller cardiac venous system returns one-quarter of the deoxygenated blood from the inner layers of the myocardium to the cardiac chambers. This system is mainly composed of Thebesian veins draining deoxygenated blood from all sections of the myocardium chambers. Thebesian veins also drain the right side of the heart more than the left side.[8]
Embryology
During embryo development, the angiogenesis of coronary veins begins from the sinus venosus, an embryonic vein that returns blood to the heart[9] The venous endothelial cells from the sinus venosus migrate towards the surface of the myocardium and then differentiate into coronary veins and their tributaries.
Blood Supply and Lymphatics
Coronary veins receive oxygenated blood and nourishment from coronary arteries and their tributaries. Additionally, large veins especially receive nutrient-rich blood supply from the vasa vasorum, a network of microvessels. Vasa vasorum are also known as the vessels of the vessels.[10]
Nerves
The heart and its venous vasculature engage in autonomic innervation. Located in the brainstem, the medulla plays a significant role in regulating the sympathetic and parasympathetic nerves to the heart and its blood vessels. In the sympathetic division, the sympathetic nerves exit the medulla and synapse with preganglionic fibers in the upper thoracic spinal cord. The preganglionic fibers then synapse with postganglionic fibers in the sympathetic ganglia, where the postganglionic nerves finally travel to and synapse with the target sites in the heart and coronary veins.[11] In the parasympathetic division, the preganglionic efferent fibers of the vagus nerve exit the medulla and synapse with postganglionic fibers within the vascular venous tissue.[12] Overall, sympathetic innervation causes coronary veins to dilate, while parasympathetic innervation causes coronary veins to constrict.
Muscles
Coronary veins are composed of three structural layers, but only the tunica media or the middle layer of the coronary veins is smooth muscle. Comparatively, the tunica media is thinner in coronary veins than in coronary arteries.[13]
Physiologic Variants
There are numerous anatomical variations of the cardiac venous system from varied numbers of tributaries to diverse diameters and locations of the cardiac veins. Clinically, understanding unique structural variations can prove salient for a patient’s diagnosis and cardiac intervention. The most notable venous variations involve the great cardiac vein and coronary sinus.[2] The great cardiac vein usually travels along the anterior interventricular sulcus, but some patients’ great cardiac veins course intramurally, which can inhibit venous flow[5] Furthermore, other clinical findings have discovered patients with duplicate coronary sinuses, which may also affect the draining of the venous system.[14]
Surgical Considerations
As the largest coronary vein, the coronary sinus is often utilized as an anatomical landmark for orientation during cardiothoracic surgery. Although most cardiac intervention procedures occur for adult patients, infant and pediatric patients suffering from congenital heart defects also undergo cardiac operations. One congenital atrial septal defect, known as the unroofed coronary sinus, involves an anomalous and unclosed connection between the coronary sinus and the left atrium.[15] If left untreated, unroofed coronary sinus can lead to heart disease. Closure between the coronary sinus and left atrium via a stent requires careful surgical management and coordinated teamwork among physicians and medical staff to deliver the best medical care for patients and their families.
Clinical Significance
The coronary venous system encompasses significant clinical value for a wide range of cardiac interventional procedures, including cardiac catheterization, retrograde cardioplegia, reperfusion of cardiac veins, drug delivery, stem cell therapy, and lead placement among many.
For clinical treatment and procedures, the most salient anatomical feature of the cardiac veins is the coronary sinus and its venous tributaries. When patients undergo open-heart surgeries or have obstructions in their coronary arteries, the myocardium must continue to receive nourishment. To prevent ischemia of the operated heart, implementing reperfusion of the coronary sinus and its venous tributaries allows an effective reverse flow of nourishment.[16] The distribution of drugs and cardioplegic solutions via the coronary sinus also holds clinical significance. In several research reports, cardiothoracic surgeons have observed that delivery through cardiac veins can prolong contact time and enhance the efficacy of the treatment even more than the delivery through cardiac arteries can.[17][18]
Due to its optimal location and high visibility, the coronary sinus is also a popular vein for retrograde cardioplegia, temporary cessation of the heart’s activity, and is targeted for left ventricular lead placement.[19] For cardiac catheterization and pacemaker placement, the left ventricular lead advances through the coronary sinus into the posterior or lateral coronary vein to enter the left ventricular wall. This procedure allows physicians to evaluate cardiac conduction and the patient’s overall coronary health.[20] From therapeutic drug delivery to permanent pacemaker insertion, these various cardiac techniques highlight the importance of the coronary venous system and the knowledge of its unique anatomy.
Other Issues
Electrocardiography, computed tomographic (CT) angiography, and magnetic resonance imaging (MRI) are excellent noninvasive diagnostic tools to evaluate the coronary sinus and detect potential cardiac anomalies, such as congenital abnormalities and vessel obstruction.[21]
Media
(Click Image to Enlarge)
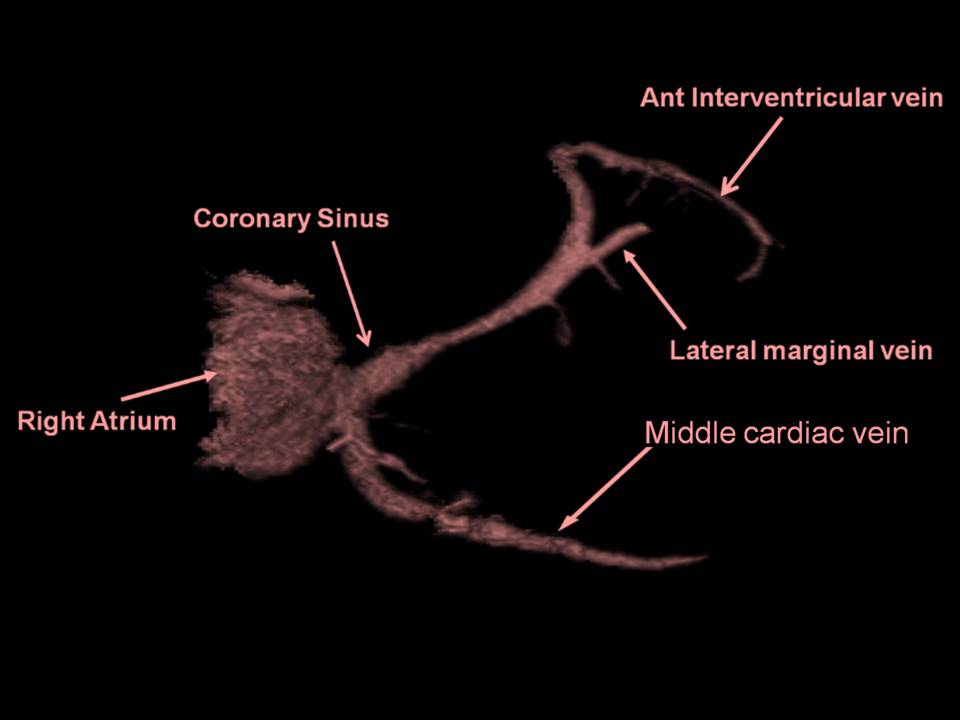
3D volume-rendered computed tomographic reconstruction of the coronary venous system. Contributed by Dr. Amgad Makaryus, from Open access publication: "Catanzaro JN, Makaryus JN, Jadonath R, Makaryus AN. Planning and Guidance of Cardiac Resynchronization Therapy–Lead Implantation by Evaluating Coronary Venous Anatomy A ssessed by Multidetector Computed Tomography. Clinical Medicine Insights: Cardiology 2014:8(S4) 43–50 doi: 10.4137/CMC.S18762".
References
Sirajuddin A, Chen MY, White CS, Arai AE. Coronary venous anatomy and anomalies. Journal of cardiovascular computed tomography. 2020 Jan-Feb:14(1):80-86. doi: 10.1016/j.jcct.2019.08.006. Epub 2019 Aug 7 [PubMed PMID: 31444098]
Loukas M, Bilinsky S, Bilinsky E, el-Sedfy A, Anderson RH. Cardiac veins: a review of the literature. Clinical anatomy (New York, N.Y.). 2009 Jan:22(1):129-45. doi: 10.1002/ca.20745. Epub [PubMed PMID: 19097063]
Ortale JR, Gabriel EA, Iost C, Márquez CQ. The anatomy of the coronary sinus and its tributaries. Surgical and radiologic anatomy : SRA. 2001:23(1):15-21 [PubMed PMID: 11370136]
Spencer JH, Anderson SE, Iaizzo PA. Human coronary venous anatomy: implications for interventions. Journal of cardiovascular translational research. 2013 Apr:6(2):208-17. doi: 10.1007/s12265-012-9443-y. Epub 2013 Jan 11 [PubMed PMID: 23307201]
von Lüdinghausen M. The venous drainage of the human myocardium. Advances in anatomy, embryology, and cell biology. 2003:168():I-VIII, 1-104 [PubMed PMID: 12645157]
Level 3 (low-level) evidenceWhitmore I. Terminologia anatomica: new terminology for the new anatomist. The Anatomical record. 1999 Apr 15:257(2):50-3 [PubMed PMID: 10321431]
von Lüdinghausen M, Ohmachi N, Besch S, Mettenleiter A. Atrial veins of the human heart. Clinical anatomy (New York, N.Y.). 1995:8(3):169-89 [PubMed PMID: 7606591]
F Boeder N, Nef HM, Bauer T. Thebesian veins as drainage to the ventricle: A case report. Cardiovascular revascularization medicine : including molecular interventions. 2017 Apr-May:18(3):213-214. doi: 10.1016/j.carrev.2016.09.007. Epub 2016 Sep 30 [PubMed PMID: 27743821]
Level 3 (low-level) evidenceChen HI, Sharma B, Akerberg BN, Numi HJ, Kivelä R, Saharinen P, Aghajanian H, McKay AS, Bogard PE, Chang AH, Jacobs AH, Epstein JA, Stankunas K, Alitalo K, Red-Horse K. The sinus venosus contributes to coronary vasculature through VEGFC-stimulated angiogenesis. Development (Cambridge, England). 2014 Dec:141(23):4500-12. doi: 10.1242/dev.113639. Epub 2014 Nov 5 [PubMed PMID: 25377552]
Level 3 (low-level) evidenceRitman EL, Lerman A. The dynamic vasa vasorum. Cardiovascular research. 2007 Sep 1:75(4):649-58 [PubMed PMID: 17631284]
Level 3 (low-level) evidenceHasan W. Autonomic cardiac innervation: development and adult plasticity. Organogenesis. 2013 Jul-Sep:9(3):176-93. doi: 10.4161/org.24892. Epub 2013 May 14 [PubMed PMID: 23872607]
Level 3 (low-level) evidenceKapa S, DeSimone CV, Asirvatham SJ. Innervation of the heart: An invisible grid within a black box. Trends in cardiovascular medicine. 2016 Apr:26(3):245-57. doi: 10.1016/j.tcm.2015.07.001. Epub 2015 Jul 9 [PubMed PMID: 26254961]
Melnikova NB, Svitenkov AI, Hose DR, Hoekstra AG. A cell-based mechanical model of coronary artery tunica media. Journal of the Royal Society, Interface. 2017 Jul:14(132):. doi: 10.1098/rsif.2017.0028. Epub [PubMed PMID: 28679664]
Sahinoglu K, Cassell MD, Miyauchi R, Bergman RA. Human persistent left superior vena cava with doubled coronary sinus. Annals of anatomy = Anatomischer Anzeiger : official organ of the Anatomische Gesellschaft. 1994 Oct:176(5):451-4 [PubMed PMID: 7978342]
Level 3 (low-level) evidenceBonardi M, Valentini A, Camporotondo R. Unroofed coronary sinus and persistent left superior vena cava: A case report. Journal of ultrasound. 2012 Sep:15(3):179-82. doi: 10.1016/j.jus.2012.06.001. Epub 2012 Jun 27 [PubMed PMID: 23450671]
Level 3 (low-level) evidenceBECK CS, STANTON E. Revascularization of heart by graft of systemic artery into coronary sinus. Journal of the American Medical Association. 1948 May 29:137(5):436-42 [PubMed PMID: 18862319]
Subayi JB, Menasché P, Piwnica A. [Retrograde cardioplegia through the coronary sinus in the surgery of the aortic valve. A clinical study of 500 patients]. Archives des maladies du coeur et des vaisseaux. 1991 Jul:84(7):943-8 [PubMed PMID: 1929713]
Menasché P, Kucharski K, Mundler O, Veyssié L, Subayi JB, Le Pimpec F, Fauchet M, Piwnica A. Adequate preservation of right ventricular function after coronary sinus cardioplegia. A clinical study. Circulation. 1989 Nov:80(5 Pt 2):III19-24 [PubMed PMID: 2553301]
Morgan JM, Delgado V. Lead positioning for cardiac resynchronization therapy: techniques and priorities. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2009 Nov:11 Suppl 5():v22-8. doi: 10.1093/europace/eup306. Epub [PubMed PMID: 19861387]
Aguilera AL, Volokhina YV, Fisher KL. Radiography of cardiac conduction devices: a comprehensive review. Radiographics : a review publication of the Radiological Society of North America, Inc. 2011 Oct:31(6):1669-82. doi: 10.1148/rg.316115529. Epub [PubMed PMID: 21997988]
Shah SS, Teague SD, Lu JC, Dorfman AL, Kazerooni EA, Agarwal PP. Imaging of the coronary sinus: normal anatomy and congenital abnormalities. Radiographics : a review publication of the Radiological Society of North America, Inc. 2012 Jul-Aug:32(4):991-1008. doi: 10.1148/rg.324105220. Epub [PubMed PMID: 22786990]