Introduction
The dexamethasone suppression test is used to diagnose endogenous Cushing syndrome by assessing the lack of suppression of the hypothalamic-pituitary-adrenal axis in response to exogenous corticosteroids.[1] The first use of dexamethasone for diagnosing Cushing syndrome was in 1960 by Liddle; he developed a test based on the non-suppressibility of endogenous cortisol production in Cushing syndrome versus the physiological suppression in nonaffected individuals achieved by dexamethasone.[2]
Dexamethasone is a potent synthetic corticosteroid (dexamethasone 0.75 mg= prednisone; 5 mg= methylprednisolone; 4 mg= hydrocortisone 20 mg) with high affinity for glucocorticoid receptors and a long duration of action (biological half-life 36-54 hours; plasma half-life 4-5 hours).[3] Furthermore, dexamethasone possesses minimal mineralocorticoid activity, and unlike other glucocorticoids, this corticosteroid does not interfere with cortisol measurement in the plasma, urine, or saliva. These qualities make dexamethasone the preferred steroid for assessing the hypothalamic-pituitary-adrenal axis.[4]
The dexamethasone suppression test is also used to investigate mild autonomous cortisol excess in patients with incidentalomas. (See Image. Dexamethasone Suppression Test.) For diagnosing Cushing syndrome and mild autonomous cortisol excess, a serum cortisol concentration of 1.8 µg/dL (50 nmol/L) is the widely recommended cut-off point that increases the diagnostic sensitivity of the test to approximately 95%.[5] Serum cortisol concentrations less than 1.8 µg/dL suggest adequate hypothalamic-pituitary-adrenal axis suppression after dexamethasone administration, effectively excluding Cushing syndrome.[6]
Inappropriately low serum dexamethasone levels during the 1-mg dexamethasone suppression test may result in false-positive results. This phenomenon is associated with recurrent use of cytochrome P450 3A4 (CYP3A4)-inducing drugs or gastrointestinal abnormalities. When serum dexamethasone is undetectable, the primary reason is failure to take the medication appropriately. Chronic use of glucocorticoids can also lead to low dexamethasone levels because exogenous glucocorticoids suppress cortisol and the cortisol receptors, which also bind dexamethasone; this lack of available receptors can cause dexamethasone to be excreted without physiological action.[7] Simultaneous measurement of serum cortisol and dexamethasone allows for dexamethasone suppression test validation, improving accuracy and avoiding unnecessary repetitions. Adherence to verbal or written recommendations is critical for interpreting the test. The measured dexamethasone level should be higher than 200 ng/dL (4.5 nmol/L) for an accurate result.
Etiology and Epidemiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Pathophysiology
The hypothalamic-pituitary-adrenal axis, a primary neuroendocrine system, helps maintain homeostatic function and the stress response.[8] Neurons in the paraventricular nucleus of the hypothalamus synthesize corticotropin-releasing hormone, which is transported through the hypophyseal portal blood to the anterior pituitary, which stimulates adrenocorticotropic hormone production.[9] Adrenocorticotropic hormone is transported through the systemic vasculature to the adrenal glands and stimulates the synthesis and secretion of cortisol by zona fasciculata of the adrenal cortex.[10]
Under basal conditions, only about 5% to 10% of circulating cortisol is free.[11] The rest is bound to proteins, with approximately 85% binding to transcortin (cortisol-binding globulin) and 5% to 10% binding to albumin. Although the free fraction is physiologically active, the albumin-bound fraction is also available due to the low binding affinity of albumin for cortisol.[12] Unbound cortisol is filtered at the glomerulus and is subsequently excreted. This fraction constitutes less than 1% of the total cortisol synthesized daily; the rest is excreted as soluble metabolites and glucuronide conjugates.[13] The excretory products of cortisol that contain the dihydroxyacetone group are known as 17-hydroxycorticosteroids.[14] Urinary 17-hydroxycorticosteroid was previously used as an indirect measure of cortisol secretion rate. Urinary cortisol reflects free, circulating cortisol levels in the blood.[15]
Serum cortisol, also called the stress hormone, exerts negative feedback on both the hypothalamus and the anterior pituitary, inhibiting the secretion of corticotropin-releasing hormone and adrenocorticotropic hormone, respectively. This positive or negative feedback mechanism helps regulate serum cortisol levels and the stress response.[16] When the hypothalamic-pituitary-adrenal axis is intact, exogenously administered corticosteroids exert feedback inhibition on serum corticotropin-releasing hormone and adrenocorticotropic hormone production by binding to the hypothalamic and pituitary glucocorticoid receptors, respectively, which subsequently leads to the suppression of serum cortisol synthesis and secretion.[8] However, in pathological hypercortisolism, the hypothalamic-pituitary-adrenal axis becomes partially or entirely resistant to feedback inhibition by exogenous steroids.[17]
Specimen Requirements and Procedure
Both serum and heparinized plasma are suitable specimens for measuring cortisol in the blood.[18] Specimens can be stored at 2 °C to 8 °C for 2 days. For more extended storage, specimens must be frozen. Freeze-thaw cycles have not been found to alter cortisol concentrations significantly.[19] Immunoassays are the main methods for estimating cortisol in clinical laboratories. These immunoassays are competitive immunoassays, where cortisol in the sample competes with labeled cortisol for a limited number of anti-cortisol antibodies.[20] Many commercial kits are available for the determination of plasma and urinary cortisol. Many immunoassays are now automated and available on semi-automated or automated analyzer platforms.[21] These immunoassays can be either heterogeneous assays, where a separation step is required to distinguish the bound from unbound fractions, or homogenous assays, where no separation is required.[22] The specificity of immunoassays depends mainly on the properties of the antibody.[23]
Chromatographic methods are an alternative to immunoassay for the determination of cortisol. These methods have the advantage of less interference but are time-consuming and unsuitable for routine analysis.[24] Among the chromatographic techniques, clinical laboratories often use high-performance liquid chromatography.[25] All high-performance liquid chromatography methods require either liquid-liquid or solid-phase extraction columns to extract cortisol from the sample. Both reversed-phase and normal-phase columns are used for chromatographic analyses, and the column effluents are monitored by either ultraviolet (254 nm) or fluorescence spectroscopy.[26]
Testing Procedures
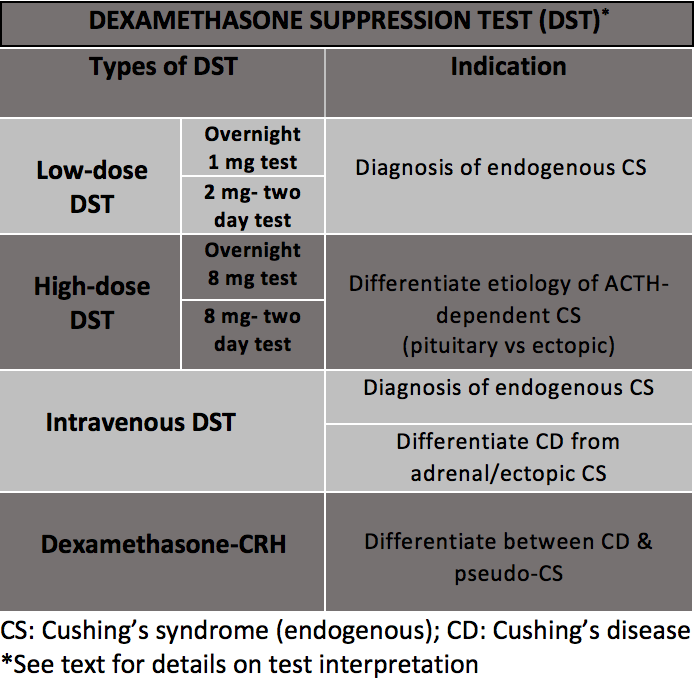
Types of Dexamethasone Suppression Tests
There are 4 different types of dexamethasone suppression tests.
Low-dose dexamethasone suppression test: This test in the initial diagnosis of Cushing syndrome as either a screening or a confirmatory test.[27] The test can be performed in 2 different formats—overnight or as a 2-day test.
- Overnight, 1 mg test: The overnight test is the most commonly used screening test due to ease and convenience. Dexamethasone 1 mg is administered orally between 11 PM and midnight. Serum cortisol levels are drawn between 8 AM and 9 AM the next morning.[28][29]
- Two-day, 2 mg test: Dexamethasone 0.5 mg is administered orally every 6 hours, typically at 9 AM, 3 PM, 9 PM, and 3 AM, for 2 days, resulting in a total dose of 4 mg. Serum cortisol levels are drawn 6 hours, typically at 9 AM, after the last administered dose.[2]
High-dose dexamethasone suppression test: Once the diagnosis of Cushing syndrome is confirmed, the next step is to categorize adrenocorticotropic hormone–independent versus adrenocorticotropic hormone–dependent Cushing syndrome by checking the plasma adrenocorticotropic hormone levels. In adrenocorticotropic hormone–independent Cushing syndrome, the plasma adrenocorticotropic hormone is low or undetectable, indicating an adrenal etiology (causing pituitary suppression of adrenocorticotropic hormone). In adrenocorticotropic hormone–dependent Cushing syndrome, plasma adrenocorticotropic hormone is inappropriately normal or high, suggesting either a pituitary or an ectopic source.
In adrenocorticotropic hormone–dependent Cushing syndrome, the high-dose dexamethasone suppression test can help distinguish a pituitary source, that is, Cushing disease) from an ectopic source of adrenocorticotropic hormone overproduction. The principle behind the high-dose dexamethasone suppression test is that the overproduction of adrenocorticotropic hormone observed in Cushing disease, but not ectopic tumors, can undergo either partial or complete suppression by high doses of dexamethasone (approximately 8 mg). Like the low-dose dexamethasone suppression test, the high-dose dexamethasone suppression test utilizes oral dexamethasone as either an overnight or 2-day test.
- Overnight, 8 mg test: Baseline morning serum cortisol is measured, and oral dexamethasone 8 mg is administered between 11 PM and midnight. Repeat serum cortisol levels are drawn the following morning, typically between 8 AM and 9 AM.[30]
- Two-day, 8 mg test: On day 1, baseline morning serum cortisol or 24-hour urinary-free cortisol from the previous day is obtained. Dexamethasone 2 mg is administered orally every 6 hours, typically at 9 AM, 3 PM, 9 PM, and 3 AM, for 2 days to complete a total dose of 16 mg over days 1 and 2. A urine sample for urinary-free cortisol is collected simultaneously with dexamethasone administration. In addition, serum cortisol levels are checked 6 hours after the last dose of dexamethasone, typically at 9 AM on day 2.[2]
Intravenous dexamethasone suppression test: This test helps in the initial diagnosis of Cushing syndrome while overcoming concerns of drug compliance and malabsorption.[31] In addition, it is useful when differentiating Cushing disease from adrenocorticotropic hormone–dependent ectopic tumors and adrenocorticotropic hormone–independent adrenal etiologies.[32] After a baseline morning serum cortisol (between 8 AM and 9 AM) is obtained, an infusion of intravenous dexamethasone at 1 mg/h for 4 to 7 hours is administered. Repeat serum cortisol levels are measured at the end of the infusion (day 1) and 23 to 24 hours later (day 2).[33]
Dexamethasone suppressed corticotropin-releasing hormone test: Based on the rationale that glucocorticoid suppression of the hypothalamic-pituitary-adrenal axis can be overcome by corticotropin-releasing hormone stimulation in Cushing disease and not in pseudo-Cushing syndrome, also known as physiological hypercortisolism, this test helps distinguish the two entities. Dexamethasone 0.5 mg every 6 hours, typically at 12 PM, 6 PM, 12 AM, and 6 AM, is given orally for 48 hours. Two hours after the last dose of dexamethasone, an intravenous corticotropin-releasing hormone dose of 1 mcg/kg is administered, typically at 8 AM, and serum cortisol levels are drawn 15 minutes later.[34]
Interfering Factors
Considerations and factors relevant to the evaluation and diagnosis of hypercortisolemia and Cushing syndrome include:
- Iatrogenic hypercortisolism can lead to exogenous Cushing syndrome.[35][36] Biochemical testing shows elevated serum cortisol levels (due to the cross-reactivity of most exogenous steroids with cortisol immunoassays) and depressed adrenocorticotropic hormone (ACTH) adrenocorticotropic hormone levels, as is typically usually observed in ACTH-independent adrenocorticotropic hormone-independent Cushing syndrome.[37][38] Individuals receiving exogenous corticosteroids—irrespective of the route of administration, eg, inhaled, topical, parenteral, intra-articular—must be identified before pursuing a work-up for pathological hypercortisolemia.[36]
- Pseudo-Cushing syndrome, also called physiological or non-neoplastic hypercortisolism, is seen in conditions such as alcohol use disorder, obesity, insulin resistance, and neuropsychiatric disorders due to HPA axis stimulation.[38][39] Obtaining a detailed history and performing a thorough physical examination are pivotal in recognizing many individuals with pseudo-Cushing syndrome. In equivocal cases, midnight serum cortisol, late-night salivary cortisol, dexamethasone-corticotropin-releasing hormone, or desmopressin testing can assist in the distinction.[23]
- In any acute illness strain, whether emotional or physical, a stress response can occur through stimulation of the HPA axis, resulting in elevated ACTH hormone and cortisol levels. Therefore, evaluation for Cushing syndrome should take place after the resolution of acute stress.[40][41]
- Corticosteroid-binding globulin: Approximately 90% of circulating cortisol is protein-bound in this globulin. Available assays measure the total cortisol levels (free and protein-bound). However, certain conditions resulting in elevated levels of circulating corticosteroid-binding globulin (CBG), such as pregnancy or use of estrogen-containing medications, or reduced levels of circulating CBG, such as malnutrition or nephrotic syndrome, may lead to spurious results on DST.[39][42] Late-night salivary cortisol or urinary-free cortisol (UFC) is the preferred test in these situations. Patients receiving estrogen therapy should ideally stop treatment at least 6 weeks before the DST.
- Dexamethasone bioavailability: MalabsorptionDexamethasone bioavailability can be affected by factors such as malabsorption, altered metabolism, or non-compliance with taking the medication can result in variable bioavailability of dexamethasone, confounding the variable results. The liver metabolizes dexamethasone through CYP3A4.[41][43] Thus, CYP3A4 inducers such as phenytoin and carbamazepine or CYP3A4 inhibitors (such as itraconazole, fluoxetine, and ritonavir) may result in decreased or increased clearance of the dexamethasone, risking false positive or negative results, respectively.[42][43] This error can be overcome by measuring serum dexamethasone levels simultaneously with serum cortisol levels. Most laboratories that conduct this test provide reference ranges based on dexamethasone dose and interval of blood drawn.
- Improper urine collection: An inadequately collected urine sample for urinary-free cortisol leads to diagnostic errors. For this reason, the urine sample should include testing for 24-hour urinary creatinine excretion in addition to cortisol.[43][44] In adults younger than 50, the 24-hour urinary creatinine excretion is approximately 15 to 20 mg/kg/day/d in women and 20 to 25 mg/kg/day/d in men. In those older than 50, there is a progressive decline in muscle mass; creatinine excretion can be as low as 10 mg/kg/d.
Results, Reporting, and Critical Findings
Low-Dose Dexamethasone Suppression Test
In either of the low-dose dexamethasone suppression tests (overnight or 2-day), a serum cortisol level of 1.8 mcg/dL (50 nmol/L) is the recommended cut-off value that increases the diagnostic sensitivity of the test to approximately 95%.[5] However, at this cut-off value, the specificity of the 2-day test is superior compared to that of the overnight test (97%-100% vs 86%). A recent meta-analysis showed that abnormal and expected results for the 1 mg overnight test had positive and negative likelihood ratios of 11.6 and 0.09, respectively.[45] On the other hand, for the 2-day 2-mg test, abnormal and normal results had positive and negative likelihood ratios of 7.3 and 0.8, respectively. Serum cortisol levels under 1.8 mcg/dL suggest adequate hypothalamic-pituitary-adrenal axis suppression by dexamethasone and exclude Cushing syndrome. Levels greater than 1.8 mcg/dL should be verified with a second test (24-hour urinary-free cortisol or late-night salivary cortisol) before establishing a confirmed diagnosis of Cushing syndrome.[29]
High-Dose Dexamethasone Suppression Test
A reduction in urinary-free cortisol or serum cortisol greater than 50% overnight during a 2-day high-dose dexamethasone suppression test makes Cushing disease the likely source of adrenocorticotropic hormone-dependent Cushing syndrome. At a cut-off value of 50% suppression, the high-dose dexamethasone suppression test provides a sensitivity and specificity of 60% to 100%, respectively. Increasing the cut-off value to greater than 90% cortisol suppression increases the specificity of diagnosing Cushing disease to almost 100%, albeit at a much-reduced sensitivity.[44] Because of this limitation, the high-dose dexamethasone suppression test is not recommended unless the pituitary magnetic resonance imaging and bilateral inferior petrosal venous sampling are either negative or logistically challenging. Even in this scenario, the high-dose dexamethasone suppression test is performed with a corticotropin-releasing hormone stimulation test to enhance diagnostic accuracy.[46]
Intravenous Dexamethasone Suppression Test
Cushing syndrome is diagnosed if the day 2 serum cortisol level is greater than 20% of the baseline value or greater than 4.7 mcg/dL (130 nmol/L) with a sensitivity of 100% and specificity of 96%.[31][32] In addition, in Cushing disease, unlike other causes of Cushing syndrome, the serum cortisol level at the end of the infusion on day 1 typically shows greater than 70% suppression from the baseline, followed by rebound hypercortisolism in 24 hours.[33]
Dexamethasone Suppressed Corticotropin-Releasing Hormone Test
Serum cortisol levels greater than 1.4 mcg/dL (39 nmol/L) at 15 minutes suggest Cushing disease with a 90% to 100% sensitivity and 50% to 100% specificity.[34][47] Raising the cut-off value to greater than 3.8 mcg/dL (87 nmol/L) increases the specificity to 100% at the cost of a slightly reduced sensitivity at 94%.[48] The cumbersomeness of this test limits its extensive application in the ambulatory setting.[49]
Clinical Significance
Iatrogenic hypercortisolism is the most common cause of Cushing syndrome. Recognizing it before these individuals are subject to further diagnostic testing is crucial; instead, the focus should be on titrating down or discontinuing, if feasible, the prescribed steroid dosage.[50] The dexamethasone suppression test must be performed and interpreted in the light of the pretest probability based on a detailed history and thorough physical examination. In addition, clinicians should be mindful of diagnostic accuracy, limitations, and interfering factors associated with all tests.[51] In cases with high clinical suspicion of Cushing syndrome but equivocal or negative test results, repeat testing should occur in 3 to 6 months, as untreated hypercortisolemia has detrimental consequences.[52]
Quality Control and Lab Safety
For nonwaived tests, laboratory regulations require, at the minimum, analysis of at least 2 levels of control materials once every 24 hours. If necessary, laboratories can assay quality control (QC) samples more frequently to ensure accurate results. QC samples should be assayed after calibration or maintenance of an analyzer to verify the correct method performance.[53] To minimize QC when performing tests for which manufacturers' recommendations are less than those required by the regulatory agency, such as once per month, the laboratories can develop an individualized quality control plan (IQCP) that involves performing a risk assessment of potential sources of error in all phases of testing and putting in place a QC plan to reduce the likelihood of errors.[54]
Westgard multi-rules are used to evaluate the QC runs. If a run is declared out of control, the system, including instruments, standards, and controls, should be investigated to identify the underlying issue. Any analysis should not be performed until the problem has been resolved.[55]
All specimens, control materials, and calibrator materials are considered potentially infectious. The standard precautions required for handling all laboratory reagents should be exercised. Disposal of all waste materials should be in accordance with local guidelines.[56] Gloves, a lab coat, and safety glasses should be worn when handling human blood specimens. All plastic tips, sample cups, and gloves that come into contact with blood should be placed in a biohazard waste container. All disposable glassware should be discarded into sharps waste containers.[57] All work surfaces should be protected with disposable absorbent bench top paper, which should be discarded into biohazard waste containers weekly or whenever blood contamination occurs. Finally, all work surfaces should be wiped weekly.[58]
Enhancing Healthcare Team Outcomes
Primary care providers may initially evaluate patients with suspected Cushing syndrome before referring them to an endocrinologist for a more comprehensive evaluation. This collaborative approach requires clear communication and careful coordination between the clinician and patient. The accurate implementation of the dexamethasone suppression test and proper sample collection are critical aspects of this process, as they can significantly influence subsequent diagnostic results.
Effective communication ensures that all stakeholders understand the importance of adhering to the testing protocol and the implications of accurate test performance. Coordination among the team members ensures that the dexamethasone suppression test is conducted correctly and that samples are collected and handled appropriately to minimize the risk of errors and ensure reliable results. This collaborative effort maximizes the diagnostic accuracy of the dexamethasone suppression test and facilitates the timely and appropriate management of patients with suspected Cushing syndrome.
Media
(Click Image to Enlarge)
References
Keevil BG. Improving the Dexamethasone Suppression Test. Clinical chemistry. 2021 Jul 6:67(7):929-931. doi: 10.1093/clinchem/hvab076. Epub [PubMed PMID: 34125167]
LIDDLE GW. Tests of pituitary-adrenal suppressibility in the diagnosis of Cushing's syndrome. The Journal of clinical endocrinology and metabolism. 1960 Dec:20():1539-60 [PubMed PMID: 13761950]
Williams DM. Clinical Pharmacology of Corticosteroids. Respiratory care. 2018 Jun:63(6):655-670. doi: 10.4187/respcare.06314. Epub [PubMed PMID: 29794202]
Tanaka K, Toriumi M, Itoh S, Ogino Y. [Dexamethasone suppression test]. Nihon rinsho. Japanese journal of clinical medicine. 1997 Apr:55 Suppl 2():353-6 [PubMed PMID: 9172546]
Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM. The diagnosis of Cushing's syndrome: an Endocrine Society Clinical Practice Guideline. The Journal of clinical endocrinology and metabolism. 2008 May:93(5):1526-40. doi: 10.1210/jc.2008-0125. Epub 2008 Mar 11 [PubMed PMID: 18334580]
Level 1 (high-level) evidenceFleseriu M, Auchus R, Bancos I, Ben-Shlomo A, Bertherat J, Biermasz NR, Boguszewski CL, Bronstein MD, Buchfelder M, Carmichael JD, Casanueva FF, Castinetti F, Chanson P, Findling J, Gadelha M, Geer EB, Giustina A, Grossman A, Gurnell M, Ho K, Ioachimescu AG, Kaiser UB, Karavitaki N, Katznelson L, Kelly DF, Lacroix A, McCormack A, Melmed S, Molitch M, Mortini P, Newell-Price J, Nieman L, Pereira AM, Petersenn S, Pivonello R, Raff H, Reincke M, Salvatori R, Scaroni C, Shimon I, Stratakis CA, Swearingen B, Tabarin A, Takahashi Y, Theodoropoulou M, Tsagarakis S, Valassi E, Varlamov EV, Vila G, Wass J, Webb SM, Zatelli MC, Biller BMK. Consensus on diagnosis and management of Cushing's disease: a guideline update. The lancet. Diabetes & endocrinology. 2021 Dec:9(12):847-875. doi: 10.1016/S2213-8587(21)00235-7. Epub 2021 Oct 20 [PubMed PMID: 34687601]
Level 3 (low-level) evidenceFarinelli DG, Oliveira KC, Hayashi LF, Kater CE. Overnight 1-mg Dexamethasone Suppression Test for Screening Cushing Syndrome and Mild Autonomous Cortisol Secretion (MACS): What Happens when Serum Dexamethasone Is Below Cutoff? How Frequent Is it? Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2023 Dec:29(12):986-993. doi: 10.1016/j.eprac.2023.09.007. Epub 2023 Sep 20 [PubMed PMID: 37734596]
Spencer RL, Deak T. A users guide to HPA axis research. Physiology & behavior. 2017 Sep 1:178():43-65. doi: 10.1016/j.physbeh.2016.11.014. Epub 2016 Nov 18 [PubMed PMID: 27871862]
Aguilera G, Liu Y. The molecular physiology of CRH neurons. Frontiers in neuroendocrinology. 2012 Jan:33(1):67-84. doi: 10.1016/j.yfrne.2011.08.002. Epub 2011 Aug 18 [PubMed PMID: 21871477]
Level 3 (low-level) evidenceBrownstein MJ. Adrenocorticotropic hormone (ACTH) in the central nervous system. Advances in biochemical psychopharmacology. 1980:22():93-9 [PubMed PMID: 6249088]
Level 3 (low-level) evidenceTanaka K. [Cortisol]. Nihon rinsho. Japanese journal of clinical medicine. 1995 Mar:53 Su Pt 2():437-40 [PubMed PMID: 8753275]
Hodes A, Meyer J, Lodish MB, Stratakis CA, Zilbermint M. Mini-review of hair cortisol concentration for evaluation of Cushing syndrome. Expert review of endocrinology & metabolism. 2018 Sep:13(5):225-231. doi: 10.1080/17446651.2018.1517043. Epub 2018 Sep 20 [PubMed PMID: 30234410]
Sederberg-Olsen P, Binder C, Kehlet H. Urinary excretion of free cortisol in impaired renal function. Acta endocrinologica. 1975 Jan:78(1):86-90 [PubMed PMID: 1172895]
Billon-Rey S, Beylot M, Mathian B, Patricot MC, Berthezene F, Mornex R, Revol A. [Comparison between the value of urinary free cortisol and 17-hydroxycorticosteroids for the diagnosis of Cushing's syndrome]. Presse medicale (Paris, France : 1983). 1986 May 24:15(21):965-8 [PubMed PMID: 2942852]
Mengden T, Hubmann P, Müller J, Greminger P, Vetter W. Urinary free cortisol versus 17-hydroxycorticosteroids: a comparative study of their diagnostic value in Cushing's syndrome. The Clinical investigator. 1992 Jul:70(7):545-8 [PubMed PMID: 1327327]
Level 2 (mid-level) evidenceLee DY, Kim E, Choi MH. Technical and clinical aspects of cortisol as a biochemical marker of chronic stress. BMB reports. 2015 Apr:48(4):209-16 [PubMed PMID: 25560699]
Level 3 (low-level) evidenceLowrance SA, Ionadi A, McKay E, Douglas X, Johnson JD. Sympathetic nervous system contributes to enhanced corticosterone levels following chronic stress. Psychoneuroendocrinology. 2016 Jun:68():163-70. doi: 10.1016/j.psyneuen.2016.02.027. Epub 2016 Feb 26 [PubMed PMID: 26974501]
Maidana P, Bruno OD, Mesch V. [A critical analysis of cortisol measurements: an update]. Medicina. 2013:73(6):579-84 [PubMed PMID: 24356273]
Gatti R, Antonelli G, Prearo M, Spinella P, Cappellin E, De Palo EF. Cortisol assays and diagnostic laboratory procedures in human biological fluids. Clinical biochemistry. 2009 Aug:42(12):1205-17. doi: 10.1016/j.clinbiochem.2009.04.011. Epub 2009 May 4 [PubMed PMID: 19414006]
El-Farhan N, Rees DA, Evans C. Measuring cortisol in serum, urine and saliva - are our assays good enough? Annals of clinical biochemistry. 2017 May:54(3):308-322. doi: 10.1177/0004563216687335. Epub 2017 Mar 16 [PubMed PMID: 28068807]
Turpeinen U, Hämäläinen E. Determination of cortisol in serum, saliva and urine. Best practice & research. Clinical endocrinology & metabolism. 2013 Dec:27(6):795-801. doi: 10.1016/j.beem.2013.10.008. Epub 2013 Oct 25 [PubMed PMID: 24275191]
Casals G, Hanzu FA. Cortisol Measurements in Cushing's Syndrome: Immunoassay or Mass Spectrometry? Annals of laboratory medicine. 2020 Jul:40(4):285-296. doi: 10.3343/alm.2020.40.4.285. Epub [PubMed PMID: 32067427]
Alwani RA, Schmit Jongbloed LW, de Jong FH, van der Lely AJ, de Herder WW, Feelders RA. Differentiating between Cushing's disease and pseudo-Cushing's syndrome: comparison of four tests. European journal of endocrinology. 2014 Apr:170(4):477-86. doi: 10.1530/EJE-13-0702. Epub 2014 Mar 8 [PubMed PMID: 24394725]
Mezzullo M, Fanelli F, Fazzini A, Gambineri A, Vicennati V, Di Dalmazi G, Pelusi C, Mazza R, Pagotto U, Pasquali R. Validation of an LC-MS/MS salivary assay for glucocorticoid status assessment: Evaluation of the diurnal fluctuation of cortisol and cortisone and of their association within and between serum and saliva. The Journal of steroid biochemistry and molecular biology. 2016 Oct:163():103-12. doi: 10.1016/j.jsbmb.2016.04.012. Epub 2016 Apr 22 [PubMed PMID: 27108942]
Level 1 (high-level) evidenceHariharan M, Naga S, VanNoord T, Kindt EK. Simultaneous assay of corticosterone and cortisol in plasma by reversed-phase liquid chromatography. Clinical chemistry. 1992 Mar:38(3):346-52 [PubMed PMID: 1547550]
Mezzullo M, Fazzini A, Gambineri A, Di Dalmazi G, Mazza R, Pelusi C, Vicennati V, Pasquali R, Pagotto U, Fanelli F. Parallel diurnal fluctuation of testosterone, androstenedione, dehydroepiandrosterone and 17OHprogesterone as assessed in serum and saliva: validation of a novel liquid chromatography-tandem mass spectrometry method for salivary steroid profiling. Clinical chemistry and laboratory medicine. 2017 Aug 28:55(9):1315-1323. doi: 10.1515/cclm-2016-0805. Epub [PubMed PMID: 28076306]
Level 1 (high-level) evidenceWood PJ, Barth JH, Freedman DB, Perry L, Sheridan B. Evidence for the low dose dexamethasone suppression test to screen for Cushing's syndrome--recommendations for a protocol for biochemistry laboratories. Annals of clinical biochemistry. 1997 May:34 ( Pt 3)():222-9 [PubMed PMID: 9158818]
Cronin C, Igoe D, Duffy MJ, Cunningham SK, McKenna TJ. The overnight dexamethasone test is a worthwhile screening procedure. Clinical endocrinology. 1990 Jul:33(1):27-33 [PubMed PMID: 2401096]
NUGENT CA, NICHOLS T, TYLER FH. DIAGNOSIS OF CUSHING'S SYNDROME; SINGLE DOSE DEXAMETHASONE SUPPRESSION TEST. Archives of internal medicine. 1965 Aug:116():172-6 [PubMed PMID: 14315650]
Bruno OD, Rossi MA, Contreras LN, Gómez RM, Galparsoro G, Cazado E, Kral M, Leber B, Arias D. Nocturnal high-dose dexamethasone suppression test in the aetiological diagnosis of Cushing's syndrome. Acta endocrinologica. 1985 Jun:109(2):158-62 [PubMed PMID: 2990131]
Biemond P, de Jong FH, Lamberts SW. Continuous dexamethasone infusion for seven hours in patients with the Cushing syndrome. A superior differential diagnostic test. Annals of internal medicine. 1990 May 15:112(10):738-42 [PubMed PMID: 2158760]
Jung C, Alford FP, Topliss DJ, Burgess JR, Long F, Gome JJ, Stockigt JR, Inder WJ. The 4-mg intravenous dexamethasone suppression test in the diagnosis of Cushing's syndrome. Clinical endocrinology. 2010 Jul:73(1):78-84. doi: 10.1111/j.1365-2265.2009.03756.x. Epub 2009 Dec 18 [PubMed PMID: 20039897]
Tran HA, Petrovsky N. Dexamethasone infusion testing in the diagnosis of Cushing's syndrome. Endocrine journal. 2005 Feb:52(1):103-9 [PubMed PMID: 15758565]
Level 1 (high-level) evidenceYanovski JA, Cutler GB Jr, Chrousos GP, Nieman LK. Corticotropin-releasing hormone stimulation following low-dose dexamethasone administration. A new test to distinguish Cushing's syndrome from pseudo-Cushing's states. JAMA. 1993 May 5:269(17):2232-8 [PubMed PMID: 8386285]
Raff H, Carroll T. Cushing's syndrome: from physiological principles to diagnosis and clinical care. The Journal of physiology. 2015 Feb 1:593(3):493-506. doi: 10.1113/jphysiol.2014.282871. Epub 2015 Jan 5 [PubMed PMID: 25480800]
Level 3 (low-level) evidencePektas SD, Dogan G, Cinar N. Iatrogenic Cushing's Syndrome with Subsequent Adrenal Insufficiency in a Patient with Psoriasis Vulgaris Using Topical Steroids. Case reports in endocrinology. 2017:2017():8320254. doi: 10.1155/2017/8320254. Epub 2017 Nov 13 [PubMed PMID: 29259830]
Level 3 (low-level) evidenceBansal V, El Asmar N, Selman WR, Arafah BM. Pitfalls in the diagnosis and management of Cushing's syndrome. Neurosurgical focus. 2015 Feb:38(2):E4. doi: 10.3171/2014.11.FOCUS14704. Epub [PubMed PMID: 25639322]
Chabre O. The difficulties of pseudo-Cushing's syndrome (or "non-neoplastic hypercortisolism"). Annales d'endocrinologie. 2018 Jun:79(3):138-145. doi: 10.1016/j.ando.2018.04.017. Epub 2018 Apr 30 [PubMed PMID: 29716734]
Bae YJ, Kratzsch J. Corticosteroid-binding globulin: modulating mechanisms of bioavailability of cortisol and its clinical implications. Best practice & research. Clinical endocrinology & metabolism. 2015 Oct:29(5):761-72. doi: 10.1016/j.beem.2015.09.001. Epub 2015 Sep 11 [PubMed PMID: 26522460]
Stephens MA, Wand G. Stress and the HPA axis: role of glucocorticoids in alcohol dependence. Alcohol research : current reviews. 2012:34(4):468-83 [PubMed PMID: 23584113]
Meikle AW. Dexamethasone suppression tests: usefulness of simultaneous measurement of plasma cortisol and dexamethasone. Clinical endocrinology. 1982 Apr:16(4):401-8 [PubMed PMID: 7094363]
Rush AJ, Schlesser MA, Giles DE, Crowley GT, Fairchild C, Altshuler KZ. The effect of dosage on the dexamethasone suppression test in normal controls. Psychiatry research. 1982 Dec:7(3):277-85 [PubMed PMID: 6962436]
Meinardi JR, Wolffenbuttel BH, Dullaart RP. Cyclic Cushing's syndrome: a clinical challenge. European journal of endocrinology. 2007 Sep:157(3):245-54 [PubMed PMID: 17766705]
Flack MR, Oldfield EH, Cutler GB Jr, Zweig MH, Malley JD, Chrousos GP, Loriaux DL, Nieman LK. Urine free cortisol in the high-dose dexamethasone suppression test for the differential diagnosis of the Cushing syndrome. Annals of internal medicine. 1992 Feb 1:116(3):211-7 [PubMed PMID: 1728204]
Level 2 (mid-level) evidenceElamin MB, Murad MH, Mullan R, Erickson D, Harris K, Nadeem S, Ennis R, Erwin PJ, Montori VM. Accuracy of diagnostic tests for Cushing's syndrome: a systematic review and metaanalyses. The Journal of clinical endocrinology and metabolism. 2008 May:93(5):1553-62. doi: 10.1210/jc.2008-0139. Epub 2008 Mar 11 [PubMed PMID: 18334594]
Level 1 (high-level) evidenceAron DC, Raff H, Findling JW. Effectiveness versus efficacy: the limited value in clinical practice of high dose dexamethasone suppression testing in the differential diagnosis of adrenocorticotropin-dependent Cushing's syndrome. The Journal of clinical endocrinology and metabolism. 1997 Jun:82(6):1780-5 [PubMed PMID: 9177382]
Pecori Giraldi F, Pivonello R, Ambrogio AG, De Martino MC, De Martin M, Scacchi M, Colao A, Toja PM, Lombardi G, Cavagnini F. The dexamethasone-suppressed corticotropin-releasing hormone stimulation test and the desmopressin test to distinguish Cushing's syndrome from pseudo-Cushing's states. Clinical endocrinology. 2007 Feb:66(2):251-7 [PubMed PMID: 17223996]
Level 1 (high-level) evidenceTirabassi G, Papa R, Faloia E, Boscaro M, Arnaldi G. Corticotrophin-releasing hormone and desmopressin tests in the differential diagnosis between Cushing's disease and pseudo-Cushing state: a comparative study. Clinical endocrinology. 2011 Nov:75(5):666-72. doi: 10.1111/j.1365-2265.2011.04096.x. Epub [PubMed PMID: 21554373]
Level 2 (mid-level) evidenceFindling JW, Raff H. DIAGNOSIS OF ENDOCRINE DISEASE: Differentiation of pathologic/neoplastic hypercortisolism (Cushing's syndrome) from physiologic/non-neoplastic hypercortisolism (formerly known as pseudo-Cushing's syndrome). European journal of endocrinology. 2017 May:176(5):R205-R216. doi: 10.1530/EJE-16-0946. Epub 2017 Feb 8 [PubMed PMID: 28179447]
Pivonello R, De Martino MC, De Leo M, Lombardi G, Colao A. Cushing's Syndrome. Endocrinology and metabolism clinics of North America. 2008 Mar:37(1):135-49, ix. doi: 10.1016/j.ecl.2007.10.010. Epub [PubMed PMID: 18226734]
Nieman LK, Ilias I. Evaluation and treatment of Cushing's syndrome. The American journal of medicine. 2005 Dec:118(12):1340-6 [PubMed PMID: 16378774]
Nieman LK. Diagnosis of Cushing's Syndrome in the Modern Era. Endocrinology and metabolism clinics of North America. 2018 Jun:47(2):259-273. doi: 10.1016/j.ecl.2018.02.001. Epub [PubMed PMID: 29754631]
Poh DKH, Lim CY, Tan RZ, Markus C, Loh TP. Internal quality control: Moving average algorithms outperform Westgard rules. Clinical biochemistry. 2021 Dec:98():63-69. doi: 10.1016/j.clinbiochem.2021.09.007. Epub 2021 Sep 14 [PubMed PMID: 34534518]
Level 2 (mid-level) evidenceKearney E. Internal quality control. Methods in molecular biology (Clifton, N.J.). 2013:1065():277-89. doi: 10.1007/978-1-62703-616-0_18. Epub [PubMed PMID: 23996371]
Level 2 (mid-level) evidenceBayat H. Selecting multi-rule quality control procedures based on patient risk. Clinical chemistry and laboratory medicine. 2017 Oct 26:55(11):1702-1708. doi: 10.1515/cclm-2016-1077. Epub [PubMed PMID: 28236626]
Level 2 (mid-level) evidenceCornish NE, Anderson NL, Arambula DG, Arduino MJ, Bryan A, Burton NC, Chen B, Dickson BA, Giri JG, Griffith NK, Pentella MA, Salerno RM, Sandhu P, Snyder JW, Tormey CA, Wagar EA, Weirich EG, Campbell S. Clinical Laboratory Biosafety Gaps: Lessons Learned from Past Outbreaks Reveal a Path to a Safer Future. Clinical microbiology reviews. 2021 Jun 16:34(3):e0012618. doi: 10.1128/CMR.00126-18. Epub 2021 Jun 9 [PubMed PMID: 34105993]
Munson E, Bowles EJ, Dern R, Beck E, Podzorski RP, Bateman AC, Block TK, Kropp JL, Radke T, Siebers K, Simmons B, Smith MA, Spray-Larson F, Warshauer DM. Laboratory Focus on Improving the Culture of Biosafety: Statewide Risk Assessment of Clinical Laboratories That Process Specimens for Microbiologic Analysis. Journal of clinical microbiology. 2018 Jan:56(1):. doi: 10.1128/JCM.01569-17. Epub 2017 Dec 26 [PubMed PMID: 29118166]
Ionescu G, Neguţ M, Combiescu AA. [Biosafety and biosecurity in the medical laboratory. Update and trends]. Bacteriologia, virusologia, parazitologia, epidemiologia (Bucharest, Romania : 1990). 2007 Jul-Dec:52(3-4):91-9 [PubMed PMID: 19326721]