Introduction
An electroencephalogram of a newborn shows unique features compared to those of older children and adults. This technique is the only clinically available study that addresses the premature neonate's normal and abnormal functional development week after week. EEG changes result from gradual structural and functional modifications during the last trimester of pregnancy. EEG features are classified according to whether they manifest endogenous generators, such as theta activities that coalesce with a slow wave or delta brushes or originate from a broader network.[1] Neonatal patterns evolve weekly based on the gestational age.[2] Moreover, delta brush, a common characteristic waveform of the human preterm electroencephalogram, represents spontaneous electrical activity. Recent studies suggested that delta brushes are also evoked by external sensory stimulation or spontaneous movement and reflect the activity of subplate neurons, which plays a significant role in early brain development and network organization.[3]
Issues of Concern
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Issues of Concern
In general, abnormalities of neonatal EEG can be classified as follows:
Background Abnormalities
In the preterm neonate between 27 and 30 weeks of postmenstrual age, there is a discontinuity of background rhythm compared to that of more mature newborns. There are high-voltage bursts of fast activity interspersed with lower-voltage activity. With maturity, the inter-burst intervals become shorter.
The direct measure of EEG amplitude (range EEG or rEEG) can be used to detect bursts and inter-burst intervals (IBI), and this automatic measurement serves as a basis for improvement in neonatal brain monitoring.[6]
However, the prolongation of inter-burst intervals (IBI) can indicate diffuse brain injury or structural abnormality of the cortical network. The acceptable duration of the longest IBI within the record varies according to postmenstrual age and is also state-dependent.[7]
Acceptable duration of the longest IBI according to postmenstrual age include the following:
- 26 weeks = 46 sec
- 27 weeks = 36 sec
- Fewer than 30 weeks = 30 to 35 sec
- 31 to 33 weeks = 20 sec
- 34 to 36 weeks = 10 sec
- 37 to 40 weeks = 6 sec
Prolongation of the IBI is, however, non-specific and may be secondary to a neurological insult or can also occur due to medication effects.
Abnormal Voltage and Lack of Differentiation of Background Activity
Loss of faster frequencies in background activity is the earliest change in response to abnormalities in cortical function. Episodic generalized or regional voltage attenuation that is transient shows milder dysfunction compared to persistent, prolonged depression. The complete absence of polyfrequency activity suggests a lack of differentiation along with other abnormalities and can be related to a severe brain insult. These can be noted in diffuse processes such as perinatal hypoxia-ischemia, metabolic encephalopathies, meningitis, encephalitis, cerebral hemorrhages, and intraventricular hemorrhages. Persistently depressed and poorly differentiated activity in follow-up EEG recording after the first 24 hours carries a poor prognosis.
Dyschronism
In simpler terms, dyschronism signifies a lag of maturity of the EEG from the expected postmenstrual age (conception age).
The term "external dyschronism" refers to disordered maturational development where the features of the record in all states demonstrate a lag or discrepancy between the stated postmenstrual age (PMA) and the PMA estimated by a review of the EEG. A gap of longer than 3 weeks suggests significant neurological dysfunction, particularly if associated with attenuation of background activity and multifocal epileptiform activity (sharp waves and spikes).
Internal dyschronism refers to the discrepancy in the postmenstrual age determined during waking and in the phase of deeper stages of quiet sleep (QS). If a lag or discrepancy of more than 3 weeks is present, this may also be a marker of cerebral dysfunction.
Electrocerebral Silence
An isoelectric recording that is invariant over several or serial recordings indicates cortical neuronal death (but not of the brainstem). Of course, this degree of neuronal function depression is not ordinarily encountered in cases of medication-induced coma or encephalopathic states.
The most rigorous technical standards are to be applied with attention to the number of scalp electrodes (8 plus reference), interelectrode distance (>10 cm), polygraphic recording of ECG, EMG, and respiration, application of appropriate intense sensory and painful stimulation, appropriate electrode impedances, time constants, and sensitivity settings (2 μV/mm) to diagnose electrocerebral silence.
Burst-Suppression Pattern
The burst suppression pattern is usually observed in patients with severely reduced brain activity, such as severe cerebral anoxia, drug-induced, toxic-metabolic encephalopathies, severe structural malformations of the brain, and general anesthesia overdose. Studies mention the importance of reliably detecting burst suppression when administering anesthetic or sedative agents, especially for cerebral-protective treatments in different neurosurgical diseases.[8]
Burst suppression is described as brief bursts of spikes, sharp waves, or slow waves of relatively high amplitude alternating with relatively flat EEG or isoelectric periods.[9] The synchronous bursts (0.5-10-second duration) alternating with or followed by isoelectric background activity (2-10 seconds) in an invariant and non-reactive record is considered as fulfilling criteria for a suppression-burst pattern. Studies show that "burst suppression with identical bursts" only occurs after diffuse cerebral ischemia and is associated with poor outcomes.[10]
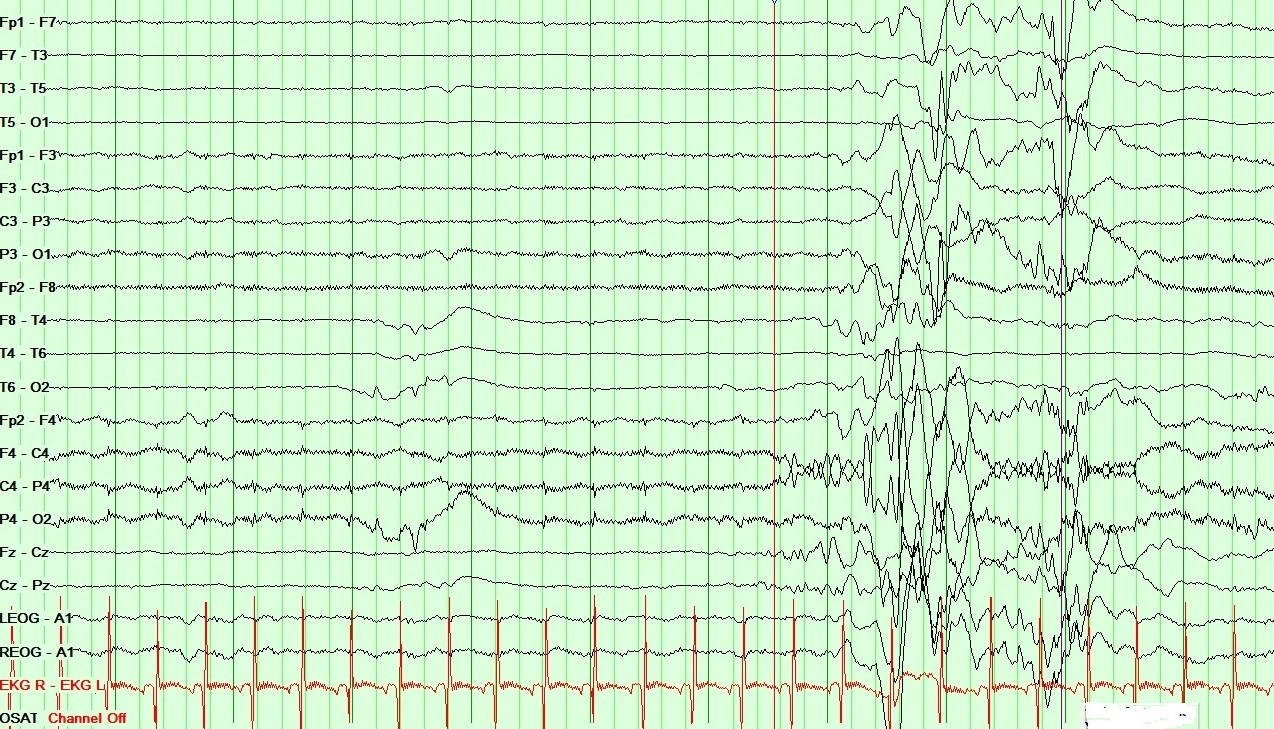
It is also the hallmark of epileptic encephalopathies with burst suppression (Ohtahara syndrome and early myoclonic encephalopathy). In these conditions, the bursts contain very high-amplitude polymorphic variable sharp and spike-wave patterns with or without symmetry, with low-amplitude intervals ranging from 2 to 10 seconds. The pattern persists in all stages of wakefulness and sleep. See Image. Neonatal EEG showing Burst Suppression pattern in Ohtahara syndrome.
Recent studies describe periods of low-amplitude electroencephalographic signal (quiescence) present during anesthetic-induced burst and postictal generalized electroencephalographic suppression (PGES). Quiescent epochs during both PGES and BS have distinct patterns of EEG signals across voltage, frequency, and spatial domains. Thus, these epochs serve as vital neurophysiological markers for clinical outcomes.[11]
Synchronous bursts (0.5-10-second duration) alternating with or followed by isoelectric background activity (2-10 seconds) in an invariant and non-reactive record are considered as fulfilling criteria for a suppression-burst pattern. This pattern can be seen in severe cerebral anoxia, drug-induced, toxic-metabolic encephalopathies, and severe structural malformations of the brain.[12][13]
Excessive Discontinuity
Discontinuous recordings that do not meet the full criteria for burst suppression are perhaps encountered more commonly, particularly in the context of perinatal hypoxic-ischemic encephalopathy. The bursting activity may be of longer duration (10-30 seconds), and IBI occupied by very low amplitude activity of less than 10 μV, less than 10-second duration. These features may be associated with loss of sleep cycling and lability. There may be other variations on this theme of discontinuity, which have been described in the literature.
Recent studies mention the association of prolonged electroencephalographic discontinuity with poor neurodevelopmental outcomes after perinatal asphyxia. Aside from seizure burden, excessive EEG discontinuity is also associated with increased cerebral tissue injury on MRI and is predictive of abnormal neurodevelopmental outcomes in infants treated with therapeutic hypothermia (TH) with hypoxic-ischaemic encephalopathy (HIE). The high positive predictive value of EEG discontinuity at 24 hours may be useful in selecting newborns with HIE for adjunctive treatments.[14] Another study assessed the prognostic effect of an early normal/mildly abnormal conventional EEG (cEEG) on seizure risk in neonates with therapeutic hypothermia. A normal/mildly abnormal cEEG during the first 24 hours indicates a very low risk of subsequent seizures. Therefore, cEEG monitoring can be safely discontinued after 24 hours if it has remained normal or excessively discontinuous and no seizures are detected, limiting the need for this resource-intensive and expensive procedure.[15]
Other Patterns of Uncertain Significance
Positive temporal sharp waves (<400 ms in duration) seen over temporal (T3, T4) electrodes can also be encountered in term neonates and may not be considered to be of clinical significance unless persistent in serial records. Similarly, short-duration central sharp waves (3-7 seconds) may occur in short bursts; these may not stand out from the background activity and are of uncertain clinical significance.
Age-Dependent Patterns of Focal Brain Injury
Focal/unilateral attenuation or voltage depression These findings acquire significance in the context of persistent voltage asymmetry throughout the recording. Focal infarcts, hemorrhage, cystic lesions, accumulation of subdural fluid, and sometimes scalp edema on the dependent side, depending on the infant's position, can be associated with such a focal or unilateral voltage depression.[16]
Positive Central Sharp Waves
Broad-based surface positive sharp waves often localize around C3, C4, and Cz electrodes. These may occur at a frequency of 1 to 2 per minute under pathological conditions. These waves are of higher amplitude and stand out against the background activity; they can sometimes be accompanied by superimposed waves of varying morphology and frequency. These positive central sharp waves are most frequently associated with periventricular white matter injury and consequential later motor impairments but have also been encountered in conditions such as meningitis, hydrocephalus, and perinatal HIE. They must be distinguished from sharp transients that can be encountered over similar locations but are of smaller amplitude and are less prominent.
Persistent Focal Sharp Waves and Spikes
One must consider the clinical significance of focal sharps and spikes, the morphology, polarity, frequency, recurrence rate, and persistence regarding regional expression. This applies to both temporal and extra-temporal expression of focal epileptiform activity. Persistent focal sharps may often be associated with a focal brain injury, while diffuse brain injury can be associated with multifocal expression of sharps or spikes. These findings may or may not be associated with identifiable abnormalities on imaging.
Neonatal Seizures and EEG Correlates
Broadly, neonatal seizures may be classified into these categories:
- Clinical seizures with electrographic correlation (electroclinical seizures)
- Purely electrographic seizures (EEG expression of ictal rhythms without clinical correlation)
- Clinical only seizures (ictal behaviors without EEG correlates)
The EEG features of background activity can provide some direction towards establishing a possible diagnosis and prognosis. In infants suspected of having seizure activity, those with abnormal EEG backgrounds are more likely to have seizures than those with a normal background. On the other hand, those with documented clinical seizure activity have been more likely to have a normal EEG background. Severe background EEG abnormalities such as burst suppression have been associated with early myoclonic encephalopathy (EME) and Ohtahara syndrome (Early Infantile Epileptic Encephalopathy).[17] A theta pointu alternant pattern (generalized sharply contoured theta alternating with periods of generalized attenuation)) has been reported to be associated with benign familial neonatal seizures, as well as during the postictal state.
EEG or aEEG recorded during the first 7 days of life in preterm infants can be a potential predictor for later neurodevelopmental outcomes.[18] Amplitude-integrated EEG (aEEG) is significant in monitoring neonates with acute symptomatic with structural, metabolic, and genetic neonatal-onset EE, being at high risk of electrographic-only and prolonged seizures. EEG/aEEG monitoring has also been identified as an adjunctive method to confirm perinatal hypoxic-ischemic encephalopathy (HIE). Long-term monitoring studies are recommended to define and classify electro-clinical patterns of neonatal-onset EE.[19]
Recent studies mentioned that continuous electroencephalogram (EEG) monitoring is the gold standard for neonatal seizure diagnosis, and most likely, high-risk neonatal populations with known brain injury, such as stroke or hypoxic-ischemic encephalopathy, will benefit from continuous EEG. Recent studies showed that categorizing risk for continuous EEG positively affects clinical care, such as fast and accurate diagnosis and treatment of neonatal seizures, reducing antiseizure medicine usage, and length of hospitalization.[20]
Ictal EEG Activity
Sustained rhythmic activity on the EEG, with highly variable morphology (spikes, sharps, slow waves, mixed waveforms), amplitude, and frequency, lasting for longer than 10 seconds, is considered suggestive of electrical seizure activity. Such activity is rarely documented earlier than 33 to 34 weeks of conception age; with increasing maturity of the neonatal brain, the ability to initiate and sustain seizure activity seems apparent. Another term, BIRDs (brief ictal rhythmic discharges), has been used to describe the rhythmic ictal activity of less than 10 seconds duration. Ictal rhythms are generally focal in onset, can arise in more than one region, tend to spread as the seizure evolves, often to one hemisphere, or may become generalized. The morphology of the discharge can change during a seizure, which helps differentiate it from monomorphic rhythmic patterns that may arise from artifacts. Specialized forms of focal discharges such as lateralized periodic discharges (LPDs) may be seen in neonates and have been described in association with herpes simplex encephalitis, and focal ischemic strokes, and have sometimes been considered indicative of seizures in a depressed brain.
A recent study defined a particular subtype of LPDs called "LPDs-max." The LPDs-max pattern corresponds to an ictal pattern, particularly a focal non-convulsive status epilepticus, sometimes associated with subtle motor signs and epileptic seizures. LPDs-max is described as periodic polyspike-wave activity or focal burst-suppression-like patterns with predominance to the posterior over the temporal-parietal-occipital regions and are refractory to antiseizure drugs.[21]
Generalized ictal rhythms are rare but may be seen in generalized myoclonus (generalized sharps) and spasms (generalized attenuation or electrodecrement). One further term—electroclinical dissociation or decoupling—must be considered here. The term denotes a situation involving the administration of AEDs to infants with electroclinical seizures; the clinical manifestations may stop while the electrical ictal rhythms continue to be expressed.[22] At other times, this may be noted when a sick infant is paralyzed for intubation and mechanical ventilation; there may be no clinical features, but the EEG continues to show ictal rhythmic patterns.
Clinical Significance
The clinical context and results of neuroimaging play key roles in the diagnosis and management, especially for neonatal seizures.[21] Even with current multichannel video electroencephalography, significant difficulty exists in the diagnosis and management of neonatal seizures, compounded by extreme prematurity, non-specific patterns, and seizures without EEG correlation.
Other Issues
Many neonatal units employ much simplified electroencephalographic techniques, amplitude-integrated EEG (aEEG), for rapid screening and monitoring.[23] Whether using aEEGs has improved outcomes of neonatal seizures is unclear.[24]
Recently, actigraphy at a medium threshold has been useful in illustrating sleep among typically developing preterm infants in the NICU; thereby, aEEG may be an adjunct to actigraphy in evaluating sleep/wake states in the NICU setting.[25] Another study assessed the effect of maternal betamethasone administration at 34 to 35 weeks of accelerated neonatal amplitude-integrated EEG (aEEG) maturation. Findings revealed that betamethasone resulted in changes in lower border voltage support accelerated electrical activity.[26]
Multiparametric EEG analysis of brain network dynamics during neonatal seizures was found to be useful over time. The proposed methods were based on analyzing standardized trends of 2 brain network properties: synchronizabilty (S) and degree of phase synchronicity given by the circular omega complexity (COC). These properties differentiate seizure events from interictal periods and seizure-free patients and could also reliably support existing seizure detectors as a post-processing step in uncertain situations.[27]
Enhancing Healthcare Team Outcomes
Neonates with abnormal EEGs require special attention. Early identification can help risk stratify them for more aggressive treatment. A dedicated team of neonatal nurses, pharmacists, neurologists, and neurophysiologists with state-of-the-art technology is essential in diagnosing and managing neonatal seizures.
Recent studies showed that EEG background is highly associated with neurodevelopmental impairment at 2 years old. These findings are helpful to healthcare providers in planning for follow-up care and counseling families for decision-making related to goals of care.[28]
A strategic approach is equally crucial, involving evidence-based strategies to optimize treatment plans and minimize adverse effects. Ethical considerations must guide decision-making, ensuring informed consent and respecting patient autonomy in treatment choices. Effective interprofessional communication is paramount, allowing seamless information exchange and collaborative decision-making among the team members. Care coordination plays a pivotal role in ensuring that the patient's journey from diagnosis to treatment and follow-up is well-managed, minimizing errors and enhancing patient safety. By embracing these principles of skill, strategy, ethics, responsibilities, interprofessional communication, and care coordination, healthcare professionals can deliver patient-centered care, ultimately improving patient outcomes and enhancing team performance in the management of abnormal neonatal EEGs.
Media
(Click Image to Enlarge)
References
Wallois F, Routier L, Heberlé C, Mahmoudzadeh M, Bourel-Ponchel E, Moghimi S. Back to basics: the neuronal substrates and mechanisms that underlie the electroencephalogram in premature neonates. Neurophysiologie clinique = Clinical neurophysiology. 2021 Jan:51(1):5-33. doi: 10.1016/j.neucli.2020.10.006. Epub 2020 Nov 6 [PubMed PMID: 33162287]
Bourel-Ponchel E, Gueden S, Hasaerts D, Héberlé C, Malfilâtre G, Mony L, Vignolo-Diard P, Lamblin MD. Normal EEG during the neonatal period: maturational aspects from premature to full-term newborns. Neurophysiologie clinique = Clinical neurophysiology. 2021 Jan:51(1):61-88. doi: 10.1016/j.neucli.2020.10.004. Epub 2020 Nov 22 [PubMed PMID: 33239230]
Kidokoro H. Delta brushes are not just a hallmark of EEG in human preterm infants. Pediatrics international : official journal of the Japan Pediatric Society. 2021 Feb:63(2):130-136. doi: 10.1111/ped.14420. Epub 2021 Feb 18 [PubMed PMID: 32749014]
André M, Lamblin MD, d'Allest AM, Curzi-Dascalova L, Moussalli-Salefranque F, S Nguyen The T, Vecchierini-Blineau MF, Wallois F, Walls-Esquivel E, Plouin P. Electroencephalography in premature and full-term infants. Developmental features and glossary. Neurophysiologie clinique = Clinical neurophysiology. 2010 May:40(2):59-124. doi: 10.1016/j.neucli.2010.02.002. Epub 2010 Mar 16 [PubMed PMID: 20510792]
Eisermann M, Kaminska A, Moutard ML, Soufflet C, Plouin P. Normal EEG in childhood: from neonates to adolescents. Neurophysiologie clinique = Clinical neurophysiology. 2013 Jan:43(1):35-65. doi: 10.1016/j.neucli.2012.09.091. Epub 2012 Oct 30 [PubMed PMID: 23290174]
Navakatikyan MA, O'Reilly D, Van Marter LJ. Automatic measurement of interburst interval in premature neonates using range EEG. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2016 Feb:127(2):1233-1246. doi: 10.1016/j.clinph.2015.11.008. Epub 2015 Nov 19 [PubMed PMID: 26690783]
Husain AM. Review of neonatal EEG. American journal of electroneurodiagnostic technology. 2005 Mar:45(1):12-35 [PubMed PMID: 15832672]
Liang Z, Wang Y, Ren Y, Li D, Voss L, Sleigh J, Li X. Detection of burst suppression patterns in EEG using recurrence rate. TheScientificWorldJournal. 2014:2014():295070. doi: 10.1155/2014/295070. Epub 2014 Apr 17 [PubMed PMID: 24883378]
Kheder A, Bianchi MT, Westover MB. Burst suppression in sleep in a routine outpatient EEG. Epilepsy & behavior case reports. 2014:2():71-4. doi: 10.1016/j.ebcr.2014.01.003. Epub 2014 Feb 12 [PubMed PMID: 25667874]
Level 3 (low-level) evidenceHofmeijer J, Tjepkema-Cloostermans MC, van Putten MJ. Burst-suppression with identical bursts: a distinct EEG pattern with poor outcome in postanoxic coma. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2014 May:125(5):947-54. doi: 10.1016/j.clinph.2013.10.017. Epub 2013 Oct 26 [PubMed PMID: 24286857]
Level 2 (mid-level) evidenceKafashan M, Brian Hickman L, Labonte AK, Huels ER, Maybrier H, Guay CS, Subramanian S, Farber NB, Ching S, Hogan RE, Kelz MB, Avidan MS, Mashour GA, Palanca BJA. Quiescence during burst suppression and postictal generalized EEG suppression are distinct patterns of activity. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2022 Oct:142():125-132. doi: 10.1016/j.clinph.2022.07.493. Epub 2022 Jul 30 [PubMed PMID: 36030576]
Wusthoff CJ, Sullivan J, Glass HC, Shellhaas RA, Abend NS, Chang T, Tsuchida TN. Interrater agreement in the interpretation of neonatal electroencephalography in hypoxic-ischemic encephalopathy. Epilepsia. 2017 Mar:58(3):429-435. doi: 10.1111/epi.13661. Epub 2017 Feb 6 [PubMed PMID: 28166364]
Bruns N, Blumenthal S, Meyer I, Klose-Verschuur S, Felderhoff-Müser U, Müller H. Application of an Amplitude-integrated EEG Monitor (Cerebral Function Monitor) to Neonates. Journal of visualized experiments : JoVE. 2017 Sep 6:(127):. doi: 10.3791/55985. Epub 2017 Sep 6 [PubMed PMID: 28930990]
Dunne JM, Wertheim D, Clarke P, Kapellou O, Chisholm P, Boardman JP, Shah DK. Automated electroencephalographic discontinuity in cooled newborns predicts cerebral MRI and neurodevelopmental outcome. Archives of disease in childhood. Fetal and neonatal edition. 2017 Jan:102(1):F58-F64. doi: 10.1136/archdischild-2015-309697. Epub 2016 Apr 21 [PubMed PMID: 27103657]
Cornet MC, Pasupuleti A, Fang A, Gonzalez F, Shimotake T, Ferriero DM, Glass HC, Cilio MR. Predictive value of early EEG for seizures in neonates with hypoxic-ischemic encephalopathy undergoing therapeutic hypothermia. Pediatric research. 2018 Sep:84(3):399-402. doi: 10.1038/s41390-018-0040-x. Epub 2018 May 3 [PubMed PMID: 29895836]
Weeke LC, van Ooijen IM, Groenendaal F, van Huffelen AC, van Haastert IC, van Stam C, Benders MJ, Toet MC, Hellström-Westas L, de Vries LS. Rhythmic EEG patterns in extremely preterm infants: Classification and association with brain injury and outcome. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2017 Dec:128(12):2428-2435. doi: 10.1016/j.clinph.2017.08.035. Epub 2017 Oct 3 [PubMed PMID: 29096216]
Olson HE, Kelly M, LaCoursiere CM, Pinsky R, Tambunan D, Shain C, Ramgopal S, Takeoka M, Libenson MH, Julich K, Loddenkemper T, Marsh ED, Segal D, Koh S, Salman MS, Paciorkowski AR, Yang E, Bergin AM, Sheidley BR, Poduri A. Genetics and genotype-phenotype correlations in early onset epileptic encephalopathy with burst suppression. Annals of neurology. 2017 Mar:81(3):419-429. doi: 10.1002/ana.24883. Epub 2017 Feb 14 [PubMed PMID: 28133863]
Fogtmann EP, Plomgaard AM, Greisen G, Gluud C. Prognostic Accuracy of Electroencephalograms in Preterm Infants: A Systematic Review. Pediatrics. 2017 Feb:139(2):. pii: e20161951. doi: 10.1542/peds.2016-1951. Epub [PubMed PMID: 28143915]
Level 1 (high-level) evidenceTrollmann R. Neuromonitoring in Neonatal-Onset Epileptic Encephalopathies. Frontiers in neurology. 2021:12():623625. doi: 10.3389/fneur.2021.623625. Epub 2021 Feb 2 [PubMed PMID: 33603712]
Nguyen T, Wusthoff CJ. Clinical manifestations of neonatal seizures. Pediatrics international : official journal of the Japan Pediatric Society. 2021 Jun:63(6):631-635. doi: 10.1111/ped.14654. Epub 2021 May 22 [PubMed PMID: 33599034]
Gelisse P,Crespel A,Genton P,Jallon P,Kaplan PW, Lateralized Periodic Discharges: Which patterns are interictal, ictal, or peri-ictal? Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2021 Jul; [PubMed PMID: 34034086]
Watanabe K. Neurophysiological aspects of neonatal seizures. Brain & development. 2014 May:36(5):363-71. doi: 10.1016/j.braindev.2014.01.016. Epub 2014 Feb 24 [PubMed PMID: 24581554]
Level 3 (low-level) evidenceZhu X, Guo Y, Liu Y, Liu K. Amplitude-Integrated Electroencephalography for Early Diagnosis and Prognostic Prediction of Hypoxic Encephalopathy in Preterm Infants. Medical science monitor : international medical journal of experimental and clinical research. 2018 Dec 5:24():8795-8802. doi: 10.12659/MSM.909330. Epub 2018 Dec 5 [PubMed PMID: 30514829]
Prempunpong C, Chalak LF, Garfinkle J, Shah B, Kalra V, Rollins N, Boyle R, Nguyen KA, Mir I, Pappas A, Montaldo P, Thayyil S, Sánchez PJ, Shankaran S, Laptook AR, Sant'Anna G. Prospective research on infants with mild encephalopathy: the PRIME study. Journal of perinatology : official journal of the California Perinatal Association. 2018 Jan:38(1):80-85. doi: 10.1038/jp.2017.164. Epub 2017 Nov 2 [PubMed PMID: 29095433]
Ülgen Ö, Barış HE, Aşkan ÖÖ, Akdere SK, Ilgın C, Özdemir H, Bekiroğlu N, Gücüyener K, Özek E, Boran P. Sleep assessment in preterm infants: Use of actigraphy and aEEG. Sleep medicine. 2023 Jan:101():260-268. doi: 10.1016/j.sleep.2022.11.020. Epub 2022 Nov 20 [PubMed PMID: 36459917]
Laptook AR, Chalak L, Pappas A, Davis A, Sanchez PJ, Van Meurs KP, Oh W, Sommers R, Shankaran S, Hensman AM, Rouse DJ, McDonald S, Das A, Goldberg RN, Ambalavanan N, Gyamfi-Bannerman C, Thom EA, Higgins RD, Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network (NRN), Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units (MFMU) Network. The effects of betamethasone on the amplitude integrated EEG of infants born at 34- or 35-weeks gestation. Journal of perinatology : official journal of the California Perinatal Association. 2022 Dec:42(12):1615-1621. doi: 10.1038/s41372-022-01415-4. Epub 2022 May 26 [PubMed PMID: 35618748]
Frassineti L, Parente A, Manfredi C. Multiparametric EEG analysis of brain network dynamics during neonatal seizures. Journal of neuroscience methods. 2021 Jan 15:348():109003. doi: 10.1016/j.jneumeth.2020.109003. Epub 2020 Nov 27 [PubMed PMID: 33249182]
Glass HC, Numis AL, Comstock BA, Gonzalez FF, Mietzsch U, Bonifacio SL, Massey S, Thomas C, Natarajan N, Mayock DE, Sokol GM, Van Meurs KP, Ahmad KA, Maitre N, Heagerty PJ, Juul SE, Wu YW, Wusthoff CJ. Association of EEG Background and Neurodevelopmental Outcome in Neonates With Hypoxic-Ischemic Encephalopathy Receiving Hypothermia. Neurology. 2023 Nov 27:101(22):e2223-e2233. doi: 10.1212/WNL.0000000000207744. Epub 2023 Nov 27 [PubMed PMID: 37816642]