Introduction
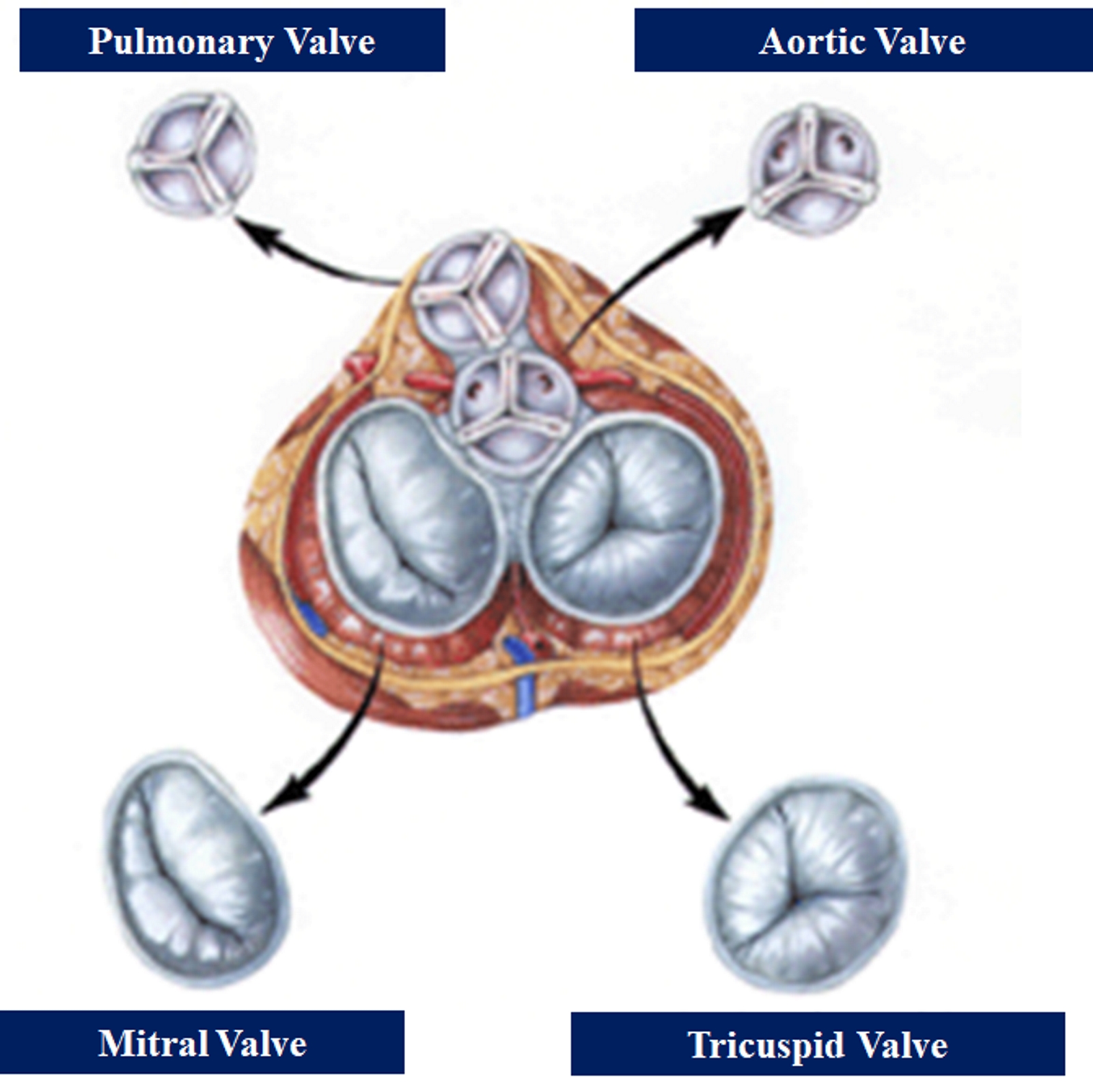
The heart is a four-chambered organ hemodynamically functioning as a reservoir and a pump; it receives deoxygenated blood from the systemic circulation through the superior and inferior vena cava in the right atrium and oxygenated blood from the lung via the four pulmonary veins. The heart pumps deoxygenated blood from the right ventricle to the lungs and at the same time pumps oxygenated blood from the left ventricle into the aorta. These processes are orchestrated by the electric conduction system which coordinates the rhythmic contractions of the atria and ventricles, and the opening and closure of the heart valves. The heart valves are especially important to effectively maintain the systolic and diastolic phase of the cardiac cycle. There are two types of heart valves; the atrioventricular valves (mitral, tricuspid) and the semilunar valves (aortic and pulmonic). The pulmonic valve physically separates the right ventricle from the pulmonary trunk. While there is more literature on the other heart valves, little is known about the intricate function of the pulmonary valve and its role in various disorders.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
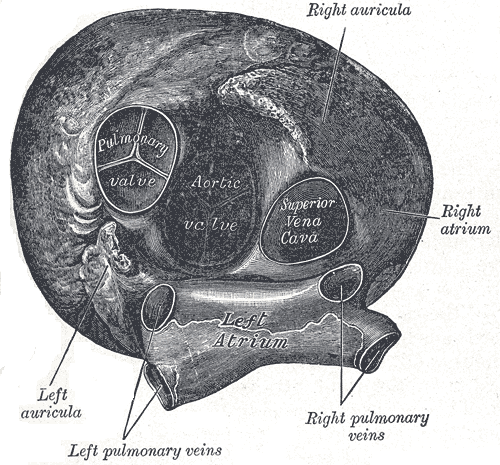
The pulmonary valve is the semilunar valve that separates the right ventricle from the pulmonary trunk. Anatomically, the annulus (ring-like connective tissue) of this valve delimits the right ventricle chamber at the junction of the pulmonary arterial trunk.[1] The annulus and the cardiac fibrous skeleton, a structure which connects the pulmonary valve to other heart valves, plays an essential role in anchoring all the heart valves in the myocardium. The pulmonary valve consists of three cusps; anterior, left, and right cusp. Each of these cusps is separated from one another by a commissure. Below the free margin, these cusps overlap one another for about several millimeters to ensure adequate closure of the lumen. The overlap area is called lunula. At the central portion of the lunula, there is a local fibrous thickening that forms a nodule (nodule of Arantius) that maximize closure of the lumen. At the superior border of the pulmonary valve, there is a pocket formed by the valve cusp and adjacent arterial wall called the sinus of Valsalva.[2]
The pulmonary valve is separated from the tricuspid valve by a muscular fold, known as the ventriculoinfundibular fold. At its septal margin, the fold forms the supraventricular crest and this fold inserts between the anterior and posterior limbs of the septomarginal trabeculation. This same fold also forms the subpulmonary infundibulum of the right ventricular outlet.[1]
Histologically, the pulmonary valve consists of stratified extracellular matrix compartments of four layers which are the arterialis, fibrosa, spongiosa, and the ventricularis layer. The arterialis faces the artery, and the ventricularis layer faces the ventricle.[2][3] Each of these layers has a different composition and function. The arterialis layer is the thinnest layer whose function remains. The fibrosa layer is the backbone of the semilunar valves and consists of circumferentially arranged dense collagen networks that merge with the annulus and the cardiac fibrous skeleton. The spongiosa layer is the layer that allows shear stress between layers during flexure and provides compressive strength because it consists of proteoglycans and glycosaminoglycans. The ventricularis layer contained the most elastic fibers and assists with the elastic recoil of the cusps.[2][4]
The pulmonary valve opens at the systolic phase of the cardiac cycle enabling the deoxygenated blood to be pumped from the right ventricle to the pulmonary circulation. It closes at the diastolic phase of the cardiac cycle, allowing sufficient filling of the right ventricle. The pulmonary valve has a diameter of about 20 mm.
Embryology
The semilunar valves develop as the endocardial cushion forms in the outflow tract (OFT) of the primordial looped heart tube; this is the first sign of valvulogenesis. Later this endocardial cushion fold becomes a primordial heart valve with highly proliferative valve progenitor cells. During remodeling and maturation, the valves are rudimentary. The primordial valve will grow and elongate into a thin fibrous cusp of the semilunar valve. These leaflets will become stratified into compartments consisting of highly organized collagen, proteoglycan, and elastin rich extracellular matrix during late gestation and soon after birth. This valves continues to mature and undergoes remodeling well into the juvenile stages.[3]
The embryonic leaflet from which the valve derives is the splanchnic mesoderm.
Blood Supply and Lymphatics
The pulmonary valve has no distinct blood supply, nor does it possess any lymphatic drainage.[5][6][7]
Nerves
The pulmonary valve receives its innervation from the nerve branch of cardiac ventricular plexus. The entire leaflets contain these nerve terminals except for the coapting edge (the ventricular layer or the lower region of each leaflet). Compared to the aortic valve leaflets, the pulmonary valve leaflets contained more numerous nerve terminals. The activity of acetylcholinesterase or tyrosine hydroxylase and neuropeptide Y are observable in these nerve terminals. With age, the density and distribution of innervation in the aortic valve leaflets decrease, but this does not occur in the pulmonary valve leaflets.[8]
Muscles
The pulmonary valve attaches to infundibulum basally, and the muscle of the subpulmonary infundibulum raises this valve above the ventricular septum; so it is positioned superiorly and becomes the most superior of the cardiac valves. This anatomical configuration enables the surgeon to resect the pulmonary valve and its basal attachment within the infundibulum from the rest of the right ventricular outflow tract (RVOT) safely.[9]
Physiologic Variants
There are several physiologic variants of the pulmonary valve. The valve may be unicuspid, bicuspid, or even atretic. Any abnormality of these components can cause congenital semilunar valve disease (CSVD).[2]
Surgical Considerations
Surgical valvotomy:
There are two types of pulmonary valve valvotomies: closed and open valvotomy. The surgeon performs a closed valvotomy by making an incision at the right ventricular outflow tract, and introducing a cutting valvulotome is introduced. This process allows one to cut the valvar diaphragm transversely, and the stenosed pulmonary valve gets converted into a bicuspid valve. The open valvotomy is done through the pulmonary artery and aided by hypothermia or by cardiopulmonary bypass. The main indication for this surgery is a relief of right ventricular obstruction.[10]
Ross procedure:
The pulmonary valve is usable as a replacement for the diseased mitral or aortic valve.[11] Because this procedure uses the patient's own tissue, it offers growth potential with increasing size of the aortic root according to age; hence, there is no need for anticoagulants. Results indicate that the Ross procedure is more durable than the prosthetic heart valves.[12] Although it is associated with a high reoperation rate, this procedure confers a survival advantage for the pediatric group and young adults.[12][13][14] A common complication of the Ross procedure, autograft dilatation, and regurgitation, has been reported and may be prevented by modifying the surgery technique.[15] A ten-year report about this procedure has revealed positive results in the short and long term, but caution is necessary for the setting of endocarditis.[16]
Clinical Significance
Bicuspid pulmonary valve
This valve anomaly has been reported in several case reports and may present as an isolated finding or associated with other anomalies such as pulmonary artery aneurysm, transposition of great arteries, or an atrial septal defect.[17],[18],[19],[20],[21] The isolated bicuspid pulmonary valve is a benign finding and associated with good clinical outcomes, and no intervention is required if there is no valve obstruction or insufficiency.[22]
Quadricuspid pulmonary valve
This valve anomaly is even rarer, and on autopsy, the incidence of this anomaly varies from 1 to 5 cases per 20000 necropsies.[23] Other reports indicate that this anomaly may be slightly more in males compared to females.[24][25] Based on one review of 121 quadricuspid valves, the anatomical variations of this valve based on frequency from the highest to the lowest as follows: 72 (60%) had 3 equal sized-cusps and 1 smaller cusp; 18 (15%) had 2 equal large cusps and 2 equal small cusps; 7 (6%) had 1 larger size cusp; 2 intermediate cusps and 1 smaller cusp; 4 (3%) had 3 equal cusps and 1 large cusp; 3 (2%) had 2 equal cusps and 2 smaller unequal cusps; 2 valves (2%) had 4 unequal-sized cusps.[26] Because it derives from the same embryology, many patients also have an aortic valve anomaly at the same time.[23][26] With advances in imaging modalities, researchers will discover the real prevalence of quadricuspid pulmonary valve.[27]
Pentacuspid pulmonary valve
To date, there are only two case reports of the pentacuspid pulmonary valve.[28],[29] The first one was found incidentally on the autopsy performed on a 50-year-old man who died because of a ruptured aortic abdominal aorta. On a closer look, he had three equal-sized cusps and two smaller ones.[28] Despite this anomaly, he was clinically and pathologically free of heart failure. The second one was a report about a successful repair of the pentacuspid pulmonary valve of a woman with dyspnea on exertion that caused a pulmonary artery aneurysm and severe regurgitation. The valve consisted of 4 equally-sized cusps and one hypoplastic one.[29]
Pulmonary stenosis
This is a common congenital defect which affects 7% of children with congenital heart disorders.[30][31] There is a female preponderance, and the prevalence is 2% in the absence of genetic disorder.[32] There are three types of pulmonary stenosis: valvular, supravalvular, and subvalvular. Although it can be an isolated lesion, sometimes it may be associated with tetralogy of Fallot, Noonan syndrome, congenital Rubella syndrome, and carcinoid syndrome.[33],[34],[35]
Pulmonary atresia
This is the most severe end of the spectrum for pulmonary stenosis, and it can occur with or without VSD. Other abnormalities such as tricuspid atresia or right ventricular free wall dysplasia usually accompany this disease.[36] Reports exist of functional pulmonary atresia in neonates with normal intracardiac anatomy.[37]
Pulmonary regurgitation, when it occurs as a congenital disorder, may be accompanied by a VSD. Other etiologies such as congenital anomalies (quadricuspid or bicuspid valve), hypoplasia, post-repair of tetralogy of Fallot or prolapse of the pulmonary valve may also be present. Carcinoid syndrome can also produce abnormalities due to thickening and shortening of the valve.[38] When it occurs as an isolated congenital anomaly, pulmonary regurgitation is tolerable for decades, but according to one meta-analysis, symptoms of heart failure developed in 20% of patients after 40 years.[39][40]
Pulmonary valve endocarditis
Pulmonic valve involvement in endocarditis accounts for 1.5 to 2% of total cases.[41] The pulmonary valve shares demographic, clinical, and microbiological characteristics with the more commonly involved tricuspid/aortic valve.[42] Risk factors for endocarditis include congenital anomalies of the heart or injection drug use.[43][44] However, rarely endocarditis can occur in a healthy person without any risk factors.[45] The diagnosis of infective endocarditis is possible with Duke criteria which have proven to be highly specific.[46][47]
Pulmonary balloon valvuloplasty:
Pulmonary balloon valvuloplasty is a procedure to repair the stenotic pulmonary valve of the heart percutaneously. The patient groin is sterilized and draped, and the patient is anticoagulated during the procedure. A catheter is inserted to the left femoral vein and advanced to the right ventricle to monitor its pressure. An 18 gauge cannula is inserted to the left femoral artery to monitor systemic pressure. Then a wedge catheter is inserted to the right femoral vein and advanced into the pulmonary artery. A "J" guidewire is then inserted through this catheter, and once in place, the wedge catheter gets removed. The dilation catheter advanced through the guidewire and positioned across the stenotic pulmonary valve. While being stabilized by the guidewire, the balloon is dilated by hand with dilute contrast material at the pressure of 45 PSI.[48] Although surgical valvotomy produces low longer-term of gradients and reduced number of reinterventions, balloon valvuloplasty is the preferred therapy in isolated valve disease because it is less invasive, less expensive, and shorter length of stays and good long term results.[49],[50]
Electrocardiography:
Based on an electrocardiography study of 100 subjects with isolated pulmonary stenosis, i.e., with an intact atrioventricular septum grouped into 4 (based on right ventricle systolic pressure), no diagnostic pattern was apparent that could aid diagnosis, irrespective of the severity of the lesion. The most common ECG finding is right ventricular hypertrophy.[51]
Echocardiography:
For pulmonary stenosis, this diagnostic tool is useful in detecting the site of the stenosis, determine the cause, quantify the severity, and determine treatment strategies. The transesophageal echocardiography can visualize the pulmonary valve and right ventricular outflow tract more clearly.[52] In pulmonary regurgitation, the role of TEE is limited because the valve is more difficult to image. However, echocardiography can help the evaluation of cusp number, motion, or structure.[38]
On X-ray, the abnormalities that may present are right ventricular enlargement, right atrial enlargement, prominent pulmonary trunk, and signs of congestive heart failure.[53] Chen sign can also be visible on X-ray, which is vascular fullness at the left lung base more than the right lung base because of the preferential flow of the turbulent jet to the left pulmonary artery.[54]
CT & MRI
Nowadays, these modalities are increasingly being used to evaluate the anatomy and functional assessment of the pulmonary valve and right ventricle. CT scan depicts with great detail the pulmonary valve, perivalvular structures, and pulmonary artery branches. Meanwhile, MRI is used postoperatively to evaluate the pulmonary valve function and grading of pulmonary regurgitation.[55]
Other Issues
There are rare cases in which the pulmonary valve is absent; in particular in the absent pulmonary valve syndrome (APVS).[56]
Media
(Click Image to Enlarge)
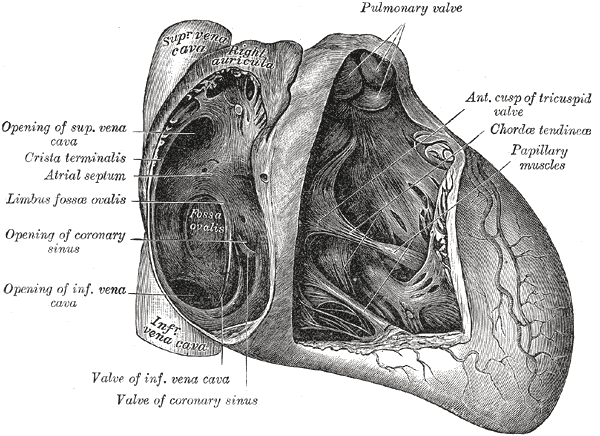
Anatomy of the Heart, Pulmonary valve, Anterior cusp of tricuspid valve, Chordae tendineae, Papillary muscles, Valve of coronary sinus, Valve of Inferior vena cava, Coronary sinus, Limbus fossae ovalis, Crista terminalis, Atrial Septum, Superior Vena cava
Henry Vandyke Carter, Public Domain, via Wikimedia Commons
(Click Image to Enlarge)
(Click Image to Enlarge)
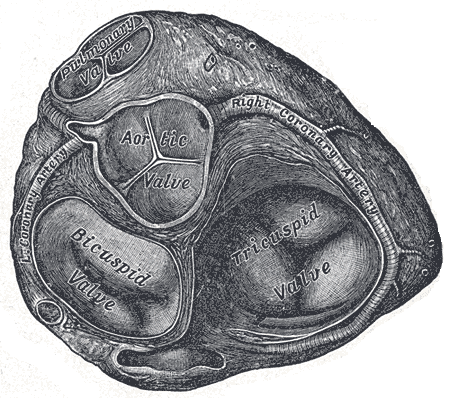
Valves of the Heart. The bicuspid valve, aortic valve, pulmonary valve, tricuspid valve, and right coronary artery are illustrated.
Henry Vandyke Carter, Public Domain, via Wikimedia Commons
(Click Image to Enlarge)
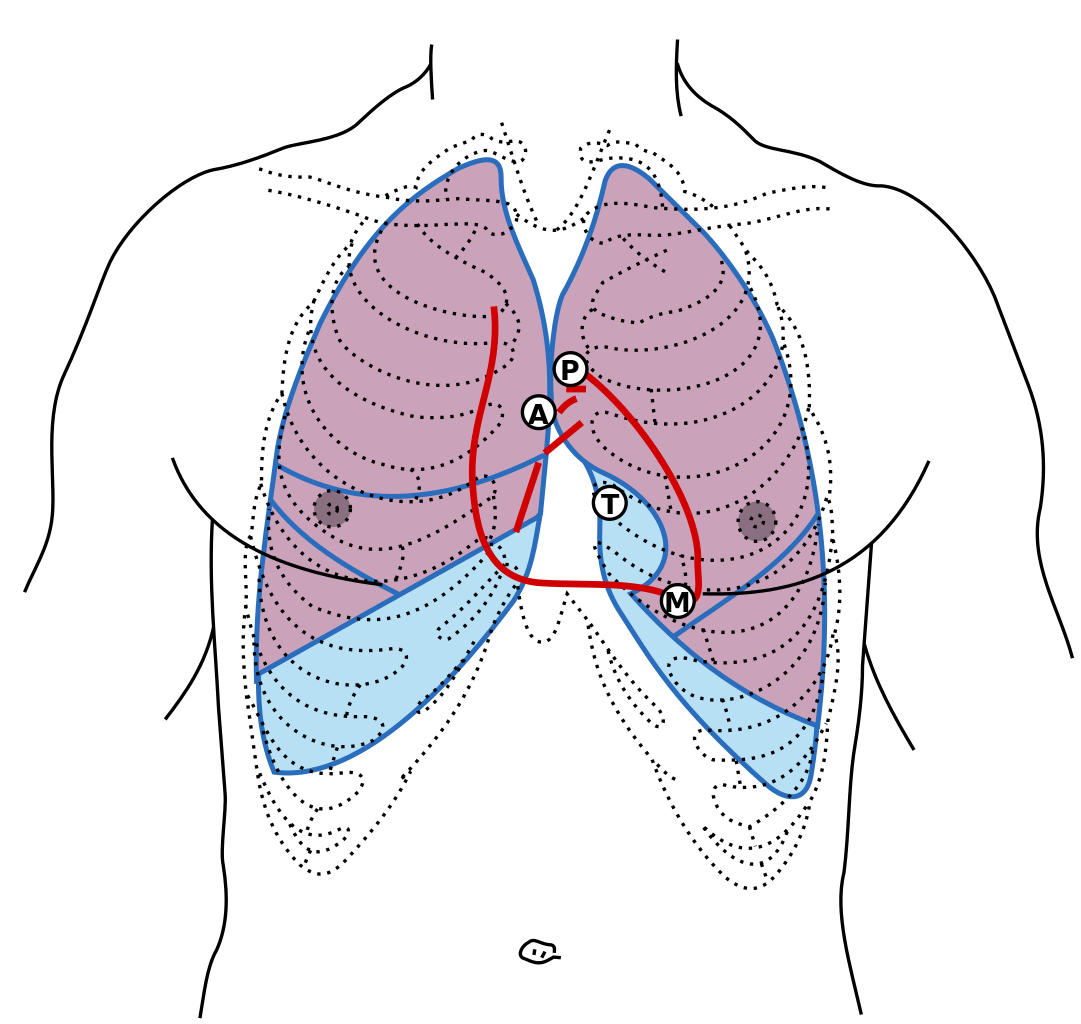
Front of thorax, showing surface relations of bones, lungs (purple), pleura (blue), and heart (red outline). The locations of best auscultation for each heart valve are labeled with "M", "T", "A", and "P". First heart sound: caused by atrioventricular valves - Mitral (M) and Tricuspid (T). Second heart sound caused by semilunar valves -- Aortic (A) and Pulmonary/Pulmonic (P). Contributed by Wikimedia Commons, Henry Gray (Public Domain)
References
Ho SY, Nihoyannopoulos P. Anatomy, echocardiography, and normal right ventricular dimensions. Heart (British Cardiac Society). 2006 Apr:92 Suppl 1(Suppl 1):i2-13 [PubMed PMID: 16543598]
Stephens EH, Kearney DL, Grande-Allen KJ. Insight into pathologic abnormalities in congenital semilunar valve disease based on advances in understanding normal valve microstructure and extracellular matrix. Cardiovascular pathology : the official journal of the Society for Cardiovascular Pathology. 2012 Jan-Feb:21(1):46-58. doi: 10.1016/j.carpath.2011.01.002. Epub 2011 Feb 23 [PubMed PMID: 21349746]
Level 3 (low-level) evidenceCombs MD, Yutzey KE. Heart valve development: regulatory networks in development and disease. Circulation research. 2009 Aug 28:105(5):408-21. doi: 10.1161/CIRCRESAHA.109.201566. Epub [PubMed PMID: 19713546]
Level 3 (low-level) evidenceSchoen FJ. Evolving concepts of cardiac valve dynamics: the continuum of development, functional structure, pathobiology, and tissue engineering. Circulation. 2008 Oct 28:118(18):1864-80. doi: 10.1161/CIRCULATIONAHA.108.805911. Epub [PubMed PMID: 18955677]
Level 3 (low-level) evidenceHarper WF. The blood supply of heart valves in relation to endocarditis. Journal of anatomy. 1938 Oct:73(Pt 1):94-111 [PubMed PMID: 17104751]
Johnson RA, Blake TM. Lymphatics of the heart. Circulation. 1966 Jan:33(1):137-42 [PubMed PMID: 5948106]
Level 3 (low-level) evidenceRatajska A, Gula G, Flaht-Zabost A, Czarnowska E, Ciszek B, Jankowska-Steifer E, Niderla-Bielinska J, Radomska-Lesniewska D. Comparative and developmental anatomy of cardiac lymphatics. TheScientificWorldJournal. 2014:2014():183170. doi: 10.1155/2014/183170. Epub 2014 Jan 27 [PubMed PMID: 24592145]
Level 3 (low-level) evidenceMarron K, Yacoub MH, Polak JM, Sheppard MN, Fagan D, Whitehead BF, de Leval MR, Anderson RH, Wharton J. Innervation of human atrioventricular and arterial valves. Circulation. 1996 Aug 1:94(3):368-75 [PubMed PMID: 8759078]
Anderson RH, Razavi R, Taylor AM. Cardiac anatomy revisited. Journal of anatomy. 2004 Sep:205(3):159-77 [PubMed PMID: 15379923]
Brock SR. THE SURGICAL TREATMENT OF PULMONARY STENOSIS. British heart journal. 1961 Jul:23(4):337-56 [PubMed PMID: 18610157]
Ross DN. Replacement of aortic and mitral valves with a pulmonary autograft. Lancet (London, England). 1967 Nov 4:2(7523):956-8 [PubMed PMID: 4167516]
Stulak JM, Burkhart HM, Sundt TM 3rd, Connolly HM, Suri RM, Schaff HV, Dearani JA. Spectrum and outcome of reoperations after the Ross procedure. Circulation. 2010 Sep 21:122(12):1153-8. doi: 10.1161/CIRCULATIONAHA.109.897538. Epub 2010 Sep 7 [PubMed PMID: 20823390]
Level 2 (mid-level) evidenceAlsoufi B, Al-Halees Z, Manlhiot C, McCrindle BW, Al-Ahmadi M, Sallehuddin A, Canver CC, Bulbul Z, Joufan M, Fadel B. Mechanical valves versus the Ross procedure for aortic valve replacement in children: propensity-adjusted comparison of long-term outcomes. The Journal of thoracic and cardiovascular surgery. 2009 Feb:137(2):362-370.e9. doi: 10.1016/j.jtcvs.2008.10.010. Epub [PubMed PMID: 19185153]
Level 2 (mid-level) evidenceMastrobuoni S, de Kerchove L, Solari S, Astarci P, Poncelet A, Noirhomme P, Rubay J, El Khoury G. The Ross procedure in young adults: over 20 years of experience in our Institution. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2016 Feb:49(2):507-12; discussion 512-3. doi: 10.1093/ejcts/ezv053. Epub 2015 Mar 3 [PubMed PMID: 25736279]
Ungerleider RM, Ootaki Y, Shen I, Welke KF. Modified Ross procedure to prevent autograft dilatation. The Annals of thoracic surgery. 2010 Sep:90(3):1035-7; discussion 1037. doi: 10.1016/j.athoracsur.2009.09.078. Epub [PubMed PMID: 20732551]
Stelzer P, Weinrauch S, Tranbaugh RF. Ten years of experience with the modified Ross procedure. The Journal of thoracic and cardiovascular surgery. 1998 May:115(5):1091-100 [PubMed PMID: 9605079]
Level 2 (mid-level) evidenceFORD AB, HELLERSTEIN HK, WOOD C, KELLY HB. Isolated congenital bicuspid pulmonary valve; clinical and pathologic study. The American journal of medicine. 1956 Mar:20(3):474-86 [PubMed PMID: 13292455]
Angeli E, Gerelli S, Beyler C, Lamerain M, Rochas B, Bonnet D, Vouhé P, Raisky O. Bicuspid pulmonary valve in transposition of the great arteries: impact on outcome. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2012 Feb:41(2):248-55. doi: 10.1016/j.ejcts.2011.03.063. Epub 2011 Dec 12 [PubMed PMID: 21757366]
Level 2 (mid-level) evidenceGoda M, Budts W, Troost E, Meyns B. Bicuspid pulmonary valve with atrial septal defect leading to pulmonary aneurysm. The Annals of thoracic surgery. 2012 May:93(5):1706-8. doi: 10.1016/j.athoracsur.2011.09.063. Epub [PubMed PMID: 22541203]
Level 3 (low-level) evidenceJamis-Dow CA, Barbier GH, Watkins MP, Lanza GM, Caruthers SD, Wickline SA. Bicuspid Pulmonic Valve and Pulmonary Artery Aneurysm. Cardiology research. 2014 Apr:5(2):83-84 [PubMed PMID: 26191115]
Manivarmane R, Taylor R, Khattar R. A case of isolated bicuspid pulmonary valve. Echo research and practice. 2017 Dec 12:5(1):K13-8. doi: 10.1530/ERP-17-0045. Epub 2017 Dec 12 [PubMed PMID: 29233813]
Level 3 (low-level) evidenceVedanthan R, Sanz J, Halperin J. Bicuspid pulmonic valve. Journal of the American College of Cardiology. 2009 Aug 18:54(8):e5. doi: 10.1016/j.jacc.2009.05.027. Epub [PubMed PMID: 19679243]
Level 3 (low-level) evidenceEnoch BA. Quadricuspid pulmonary valve. British heart journal. 1968 Jan:30(1):67-9 [PubMed PMID: 5637560]
Ascione L, lengo R, Tuccillo B, D'Andrea A, De Michele M, Porto A, Ronza F, Del Viscovo L. Quadricuspid pulmonary valve diagnosed by cardiac magnetic resonance. Journal of cardiovascular medicine (Hagerstown, Md.). 2009 Dec:10(12):944-5 [PubMed PMID: 20054880]
Level 3 (low-level) evidenceAkerem Khan SK, Anavekar NS, Araoz PA. Quadricuspid pulmonary valve: computed tomography case series and review of relevant literature. Journal of thoracic imaging. 2012 Nov:27(6):W171-3. doi: 10.1097/RTI.0b013e31822e864c. Epub [PubMed PMID: 23090364]
Level 3 (low-level) evidenceHurwitz LE, Roberts WC. Quadricuspid semilunar valve. The American journal of cardiology. 1973 May:31(5):623-6 [PubMed PMID: 4698133]
Dunay SN, Roberge RA, Avedissian LS. Quadricuspid pulmonic valve found on well exam. Military Medical Research. 2015:2():10. doi: 10.1186/s40779-015-0037-2. Epub 2015 Apr 23 [PubMed PMID: 25922689]
Demircin S, Keles-Coskun N. A case of pentacuspid pulmonary valve. Surgical and radiologic anatomy : SRA. 2010 Jul:32(6):613-5. doi: 10.1007/s00276-009-0607-7. Epub 2009 Dec 19 [PubMed PMID: 20024548]
Level 3 (low-level) evidenceKadowaki T, Yamaguchi H, Takagaki M, Nakamura H, Mitsuyama S, Ando T, Takinami G, Nakao T. Successful Repair of Regurgitant Pentacuspid Pulmonary Valve Combined With Pulmonary Artery Aneurysm. The Annals of thoracic surgery. 2016 May:101(5):1990-2. doi: 10.1016/j.athoracsur.2015.07.038. Epub [PubMed PMID: 27106440]
Samánek M, Slavík Z, Zborilová B, Hrobonová V, Vorísková M, Skovránek J. Prevalence, treatment, and outcome of heart disease in live-born children: a prospective analysis of 91,823 live-born children. Pediatric cardiology. 1989 Fall:10(4):205-11 [PubMed PMID: 2687820]
Stephensen SS, Sigfusson G, Eiriksson H, Sverrisson JT, Torfason B, Haraldsson A, Helgason H. Congenital cardiac malformations in Iceland from 1990 through 1999. Cardiology in the young. 2004 Aug:14(4):396-401 [PubMed PMID: 15680046]
Level 2 (mid-level) evidenceCAMPBELL M. Factors in the aetiology of pulmonary stenosis. British heart journal. 1962 Sep:24(5):625-32 [PubMed PMID: 14018144]
Way RC. Cardiovascular defects and the rubella syndrome. Canadian Medical Association journal. 1967 Nov 25:97(22):1329-34 [PubMed PMID: 6061597]
Ko JM. Genetic Syndromes associated with Congenital Heart Disease. Korean circulation journal. 2015 Sep:45(5):357-61. doi: 10.4070/kcj.2015.45.5.357. Epub 2015 Jul 7 [PubMed PMID: 26413101]
Bhattacharyya S, Davar J, Dreyfus G, Caplin ME. Carcinoid heart disease. Circulation. 2007 Dec 11:116(24):2860-5 [PubMed PMID: 18071089]
Xanthos T, Dalivigkas I, Ekmektzoglou KA. Anatomic variations of the cardiac valves and papillary muscles of the right heart. Italian journal of anatomy and embryology = Archivio italiano di anatomia ed embriologia. 2011:116(2):111-26 [PubMed PMID: 22303639]
Altun G, Babaoğlu K, Binnetoğlu K, Kavas N, Arısoy AE. Functional pulmonary atresia in newborn with normal intracardiac anatomy: Successful treatment with inhaled nitric oxide and pulmonary vasodilators. Annals of pediatric cardiology. 2013 Jan:6(1):83-6. doi: 10.4103/0974-2069.107243. Epub [PubMed PMID: 23626445]
Level 3 (low-level) evidenceLancellotti P, Tribouilloy C, Hagendorff A, Moura L, Popescu BA, Agricola E, Monin JL, Pierard LA, Badano L, Zamorano JL, European Association of Echocardiography. European Association of Echocardiography recommendations for the assessment of valvular regurgitation. Part 1: aortic and pulmonary regurgitation (native valve disease). European journal of echocardiography : the journal of the Working Group on Echocardiography of the European Society of Cardiology. 2010 Apr:11(3):223-44. doi: 10.1093/ejechocard/jeq030. Epub [PubMed PMID: 20375260]
Chaturvedi RR, Redington AN. Pulmonary regurgitation in congenital heart disease. Heart (British Cardiac Society). 2007 Jul:93(7):880-9 [PubMed PMID: 17569817]
Shimazaki Y, Blackstone EH, Kirklin JW. The natural history of isolated congenital pulmonary valve incompetence: surgical implications. The Thoracic and cardiovascular surgeon. 1984 Aug:32(4):257-9 [PubMed PMID: 6207619]
Tariq M, Smego RA Jr, Soofi A, Islam N. Pulmonic valve endocarditis. Southern medical journal. 2003 Jun:96(6):621-3 [PubMed PMID: 12938795]
Level 3 (low-level) evidenceKang N, Smith W, Greaves S, Haydock D. Pulmonary-valve endocarditis. The New England journal of medicine. 2007 May 24:356(21):2224-5 [PubMed PMID: 17522411]
Level 3 (low-level) evidenceSeraj SM, Gill E, Sekhon S. Isolated pulmonary valve endocarditis: truth or myth? Journal of community hospital internal medicine perspectives. 2017:7(5):329-331. doi: 10.1080/20009666.2017.1374808. Epub 2017 Oct 18 [PubMed PMID: 29147479]
Level 3 (low-level) evidenceSwaminath D, Yaqub Y, Narayanan R, Paone RF, Nugent K, Arvandi A. Isolated Pulmonary Valve Endocarditis Complicated With Septic Emboli to the Lung Causing Pneumothorax, Pneumonia, and Sepsis in an Intravenous Drug Abuser. Journal of investigative medicine high impact case reports. 2013 Oct-Dec:1(4):2324709613514566. doi: 10.1177/2324709613514566. Epub 2013 Nov 28 [PubMed PMID: 26425590]
Level 3 (low-level) evidenceRanjith MP, Rajesh KF, Rajesh G, Haridasan V, Bastian C, Sajeev CG, Krishnan MN. Isolated pulmonary valve endocarditis: A case report and review of literature. Journal of cardiology cases. 2013 Nov:8(5):161-163. doi: 10.1016/j.jccase.2013.07.007. Epub 2013 Sep 28 [PubMed PMID: 30534282]
Level 3 (low-level) evidenceBaddour LM, Wilson WR, Bayer AS, Fowler VG Jr, Tleyjeh IM, Rybak MJ, Barsic B, Lockhart PB, Gewitz MH, Levison ME, Bolger AF, Steckelberg JM, Baltimore RS, Fink AM, O'Gara P, Taubert KA, American Heart Association Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young, Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and Stroke Council. Infective Endocarditis in Adults: Diagnosis, Antimicrobial Therapy, and Management of Complications: A Scientific Statement for Healthcare Professionals From the American Heart Association. Circulation. 2015 Oct 13:132(15):1435-86. doi: 10.1161/CIR.0000000000000296. Epub 2015 Sep 15 [PubMed PMID: 26373316]
Hoen B, Béguinot I, Rabaud C, Jaussaud R, Selton-Suty C, May T, Canton P. The Duke criteria for diagnosing infective endocarditis are specific: analysis of 100 patients with acute fever or fever of unknown origin. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 1996 Aug:23(2):298-302 [PubMed PMID: 8842267]
Kan JS, White RI Jr, Mitchell SE, Gardner TJ. Percutaneous balloon valvuloplasty: a new method for treating congenital pulmonary-valve stenosis. The New England journal of medicine. 1982 Aug 26:307(9):540-2 [PubMed PMID: 7099226]
Level 3 (low-level) evidencePeterson C, Schilthuis JJ, Dodge-Khatami A, Hitchcock JF, Meijboom EJ, Bennink GB. Comparative long-term results of surgery versus balloon valvuloplasty for pulmonary valve stenosis in infants and children. The Annals of thoracic surgery. 2003 Oct:76(4):1078-82; discussion 1082-3 [PubMed PMID: 14529989]
Level 2 (mid-level) evidenceMerino-Ingelmo R, Santos-de Soto J, Coserria-Sánchez F, Descalzo-Señoran A, Valverde-Pérez I. Long-term results of percutaneous balloon valvuloplasty in pulmonary valve stenosis in the pediatric population. Revista espanola de cardiologia (English ed.). 2014 May:67(5):374-9. doi: 10.1016/j.rec.2013.08.020. Epub 2014 Feb 5 [PubMed PMID: 24774730]
BENTIVOGLIO LG, MARANHAO V, DOWNING DF. The electrocardiogram in pulmonary stenosis with intact septa. American heart journal. 1960 Mar:59():347-57 [PubMed PMID: 13799028]
Baumgartner H, Hung J, Bermejo J, Chambers JB, Evangelista A, Griffin BP, Iung B, Otto CM, Pellikka PA, Quiñones M, American Society of Echocardiography, European Association of Echocardiography. Echocardiographic assessment of valve stenosis: EAE/ASE recommendations for clinical practice. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2009 Jan:22(1):1-23; quiz 101-2. doi: 10.1016/j.echo.2008.11.029. Epub [PubMed PMID: 19130998]
Rautenburg HW, Askevold IB. [The chest X-ray in children with pulmonic stenosis and intact ventricular septum (author's transl)]. Herz. 1980 Oct:5(5):306-13 [PubMed PMID: 6450150]
Chen JT, Robinson AE, Goodrich JK, Lester RG. Uneven distribution of pulmonary blood flow between left and right lungs in isolated valvular pulmonary stenosis. The American journal of roentgenology, radium therapy, and nuclear medicine. 1969 Oct:107(2):343-50 [PubMed PMID: 5823902]
Saremi F, Gera A, Ho SY, Hijazi ZM, Sánchez-Quintana D. CT and MR imaging of the pulmonary valve. Radiographics : a review publication of the Radiological Society of North America, Inc. 2014 Jan-Feb:34(1):51-71. doi: 10.1148/rg.341135026. Epub [PubMed PMID: 24428282]
Zhou WY, Li YY, He XQ, Wang YB. Absent Pulmonary Valve Syndrome in a Fetus: A Case Report and Literature Review. Fetal and pediatric pathology. 2019 Feb:38(1):57-62. doi: 10.1080/15513815.2018.1529066. Epub 2019 Jan 20 [PubMed PMID: 30661433]
Level 3 (low-level) evidence