Introduction
Ketogenesis is a metabolic pathway that produces ketone bodies, which provide an alternative form of energy for the body (see Image. Ketogenesis). The body is constantly producing small amounts of ketone bodies that can make 22 ATP each in normal circumstances, and it is regulated mainly by insulin. In a state of ketosis, ketone body production is increased when there are decreased carbohydrates or increased fatty acids. However, ketoacidosis can occur if too many ketone bodies accumulate, such as in cases of uncontrolled diabetes.[1][2][3]
Molecular Level
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Molecular Level
Ketogenesis produces acetone, acetoacetate, and beta-hydroxybutyrate molecules by breaking down fatty acids. These ketones are water-soluble lipid molecules made up of two R-groups attached to a carbonyl group (C = O). Because they are water-soluble, they do not require lipoproteins for transport. Of the three, acetoacetate and beta-hydroxybutyrate are acidic, having pKa values of 3.6 and 4.7, respectively.[4][5]
Function
In healthy humans, the body is continually making a small number of ketones to be used by the body for energy. In times of fasting, even overnight while sleeping, the level of ketone bodies in the blood increases. The normal pathways to create energy involve either stored carbohydrate or non-carbohydrate substances. When ample carbohydrate stores are available, the main pathway used is glycogenolysis. This involves the breakdown of glycogen stores in muscle and liver. Gluconeogenesis, the production of glucose from non-carbohydrate sources such as lactate, is often utilized as well, especially in situations involving exercise.
When carbohydrate stores are significantly decreased or fatty acid concentration increases, there is an upregulation of the ketogenic pathway and an increased production of ketone bodies. This can be seen in conditions such as type 1 diabetes, alcoholism, and starvation. Most organs and tissues can use ketone bodies as an alternative source of energy. The brain uses them as a major source of energy during periods where glucose is not readily available. This is because, unlike other organs in the body, the brain has an absolute minimum glucose requirement. The heart typically uses fatty acids as its source of energy but also can use ketones. Although it is the primary site that produces ketone bodies, the liver does not use ketone bodies because it lacks the necessary enzyme beta ketoacyl-CoA transferase.
Mechanism
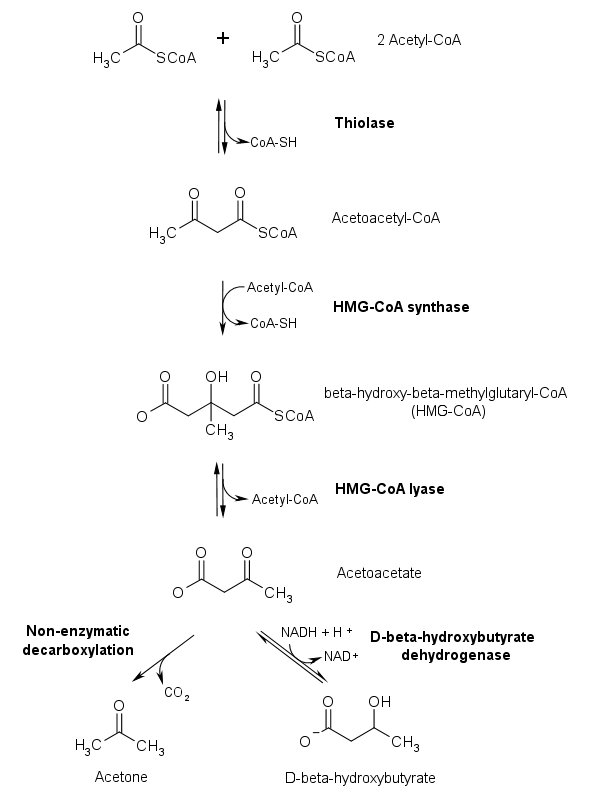
Ketogenesis occurs primarily in the mitochondria of liver cells. Fatty acids are brought into the mitochondria via carnitine palmitoyltransferase (CPT-1) and then broken down into acetyl CoA via beta-oxidation. Two acetyl-CoA molecules are converted into acetoacetyl-CoA via the enzyme thiolase; this is also known as acetyl coenzyme A acetyltransferase (ACAT). Afterward, acetoacetyl-CoA is converted to HMG-CoA via the enzyme HMG-CoA synthase. HMG-CoA lyase then converts HMG-CoA to acetoacetate. Acetoacetate can be converted to either acetone through non-enzymatic decarboxylation or to beta-hydroxybutyrate via beta-hydroxybutyrate dehydrogenase.
Acetoacetate and beta-hydroxybutyrate are the two ketone bodies used by the body for energy. Once they reach extrahepatic tissues, beta-hydroxybutyrate is converted to acetoacetate via the enzyme beta-hydroxybutyrate dehydrogenase, and acetoacetate is converted back to acetyl-CoA via the enzyme beta-ketoacyl-CoA transferase. Acetyl-CoA goes through the citric acid cycle, and after oxidative phosphorylation, produces 22 ATP per molecule. Acetone does not convert back to acetyl-CoA, so it is either excreted through urine or exhaled.
Regulation of Ketogenesis
Ketogenesis can be upregulated by hormones such as glucagon, cortisol, thyroid hormones, and catecholamines by causing a more significant breakdown of free fatty acids, thus increasing the amount available to be used in the ketogenic pathway. However, insulin is the primary hormonal regulator of this process.
Insulin regulates many key enzymes in the ketogenic pathway, and a state of low insulin triggers the process. A low insulin state leads to:
- Increased free fatty acids (FFAs)
- Due to decreased inhibition of hormone-sensitive lipase
- Increased uptake of FFAs into the mitochondria
- Due to decreased activation of acetyl-CoA carboxylase, decreasing malonyl CoA, which disinhibits Carnitine Palmitoyltransferase 1 (CPT1)
- Increased production of ketone bodies
- Due to increased HMG-CoA activity
Testing
Ketone bodies produced during ketogenesis can be measured with a urinalysis. Results range from 0 (not detected) to +4 (high amount detected). Acetone produced from ketogenesis can be directly measured in blood serum, and a normal level is below 0.6 mmol/L.
Clinical Significance
An overproduction of ketone bodies through increased ketogenesis can pose a problem due to their acidic nature.[6][7][8][9]
Diabetic ketoacidosis (DKA) is an example involving the overproduction of ketone bodies. It occurs when there is a lack of or resistance to insulin. This usually occurs in people with type I diabetes, although it can happen to people with advanced type II diabetes as well. In most cases of type II diabetes, enough insulin production continues to prevent excessive ketogenesis.
Due to the lack of glucose brought in by insulin, cells start to produce glucose via gluconeogenesis. This process, along with existing glucose that cannot be brought in with insulin, significantly elevates serum glucose levels. The threshold for DKA is a glucose level of 250. However, it is typically greater than this amount.
Once carbohydrate stores become depleted and gluconeogenesis cannot occur anymore, ketogenesis is substantially increased, and greater amounts of ketone bodies are produced. Due to the acidic nature of beta-hydroxybutyrate and acetoacetate, this causes an anion gap metabolic acidosis.
On presentation, patients are usually very dehydrated from being hyperglycemic. The high glucose levels lead to osmotic diuresis, involving greater osmole concentrations (in this case, the osmole being glucose) that cause an increased osmotic pressure, which leads to reduced water reabsorption in the kidneys. Along with being dehydrated, patients typically present with confusion, nausea, vomiting, and abdominal pain. Because of the acidosis, patients often breathe very deeply and rapidly to eliminate carbon dioxide and cause respiratory alkalosis. This process is known as Kussmaul breathing, and, over time, a patient can experience respiratory distress due to the prolonged exertion of respiratory muscles. Cerebral edema can occur in severe cases of DKA. Because of the acetone produced by ketogenesis, patients can have breath that smells fruity or like nail polish remover.
The main goal of treating DKA is to resolve metabolic acidosis, which involves giving glucose and insulin to lower blood glucose levels and downregulate the ketogenic pathway and decrease the number of ketone bodies produced.
Ketoacidosis also can occur with severe alcoholism and prolonged starvation.
Media
(Click Image to Enlarge)
References
Green A, Bishop RE. Ketoacidosis - Where Do the Protons Come From? Trends in biochemical sciences. 2019 Jun:44(6):484-489. doi: 10.1016/j.tibs.2019.01.005. Epub 2019 Feb 8 [PubMed PMID: 30744927]
d'Avignon DA, Puchalska P, Ercal B, Chang Y, Martin SE, Graham MJ, Patti GJ, Han X, Crawford PA. Hepatic ketogenic insufficiency reprograms hepatic glycogen metabolism and the lipidome. JCI insight. 2018 Jun 21:3(12):. pii: 99762. doi: 10.1172/jci.insight.99762. Epub 2018 Jun 21 [PubMed PMID: 29925686]
Adeva-Andany MM, Funcasta-Calderón R, Fernández-Fernández C, Castro-Quintela E, Carneiro-Freire N. Metabolic effects of glucagon in humans. Journal of clinical & translational endocrinology. 2019 Mar:15():45-53. doi: 10.1016/j.jcte.2018.12.005. Epub 2018 Dec 20 [PubMed PMID: 30619718]
Ghimire P,Dhamoon AS, Ketoacidosis 2019 Jan; [PubMed PMID: 30521269]
Westman EC, Tondt J, Maguire E, Yancy WS Jr. Implementing a low-carbohydrate, ketogenic diet to manage type 2 diabetes mellitus. Expert review of endocrinology & metabolism. 2018 Sep:13(5):263-272. doi: 10.1080/17446651.2018.1523713. Epub [PubMed PMID: 30289048]
Nakagawa Y, Shimano H. CREBH Regulates Systemic Glucose and Lipid Metabolism. International journal of molecular sciences. 2018 May 8:19(5):. doi: 10.3390/ijms19051396. Epub 2018 May 8 [PubMed PMID: 29738435]
Youngson NA, Morris MJ, Ballard JWO. The mechanisms mediating the antiepileptic effects of the ketogenic diet, and potential opportunities for improvement with metabolism-altering drugs. Seizure. 2017 Nov:52():15-19. doi: 10.1016/j.seizure.2017.09.005. Epub 2017 Sep 13 [PubMed PMID: 28941398]
Cardoso L,Vicente N,Rodrigues D,Gomes L,Carrilho F, Controversies in the management of hyperglycaemic emergencies in adults with diabetes. Metabolism: clinical and experimental. 2017 Mar; [PubMed PMID: 28183452]
Qiu H, Novikov A, Vallon V. Ketosis and diabetic ketoacidosis in response to SGLT2 inhibitors: Basic mechanisms and therapeutic perspectives. Diabetes/metabolism research and reviews. 2017 Jul:33(5):. doi: 10.1002/dmrr.2886. Epub 2017 Feb 23 [PubMed PMID: 28099783]
Level 2 (mid-level) evidence