Introduction
Anterior spinal artery syndrome, also known as anterior cord syndrome or ventral cord syndrome, is a neurologic condition that results in motor paralysis, pain, temperature sensory loss, and occasionally autonomic dysfunction due to occlusion of the anterior spinal artery. This artery supplies the anterior two-thirds of the spinal cord and runs along the entire length of the anterior surface of the spinal cord. Occlusion of the anterior spinal artery leads to reduced blood flow and eventual infarction of the spinal cord, resulting in bilateral lower extremity paresis or paraplegia with loss of pain and temperature sensation. Generally, the patient presentation varies depending on the portion of the spinal cord affected and the time to treatment. In addition to motor deficits and loss of pain and temperature sensation, clinical findings include back pain or autonomic dysfunction, such as hypotension, neurogenic bowel or bladder, and sexual dysfunction. The severity of motor dysfunction can range from paraplegia to quadriplegia, depending on the rostral level of the involved spinal cord. This clinical manifestation is known as anterior spinal artery syndrome. Typically, proprioception, vibratory sense, and fine touch are preserved. The neurological deficits manifest below the level of insult due to the anatomical distribution of the spinal cord tracts.
The anterior spinal artery forms from the bilateral vertebral arteries at the foramen magnum and runs as an uninterrupted artery within the anterior median sulcus of the spinal cord to the conus medullaris. Radicular arteries enter the spinal canal through the intervertebral foramen and primarily supply the nerve roots; however, some anastomoses contribute to the anterior spinal artery. The largest of these radicular arteries is the artery of Adamkiewicz, also known as arteria radicularis magna, which most commonly arises from a left intercostal artery between segments T9 to T12 but can vary anatomically. The anterior spinal artery branches into small sulcal and penetrating arteries that enter the body of the spinal cord.[1] The anterior spinal artery supplies blood to the spinal cord's bilateral anterior and lateral horns and the bilateral spinothalamic and corticospinal tracts. The anterior horns and corticospinal tracts control the somatic motor system from the neck to the feet. The lateral horns, spanning levels T1 to L2 of the spinal cord, comprise the neuronal cell bodies of the sympathetic nervous system. The spinothalamic tracts relay pain, temperature, and sensory information. The anterior spinal artery, with a few radicular artery contributions, is the sole source of blood supply to these areas of the spinal cord through sulcal and penetrating arteries. Ischemia of the anterior spinal artery causes symptoms consistent with the dysfunction of these tracts. As the anterior spinal artery is formed from the more cephalad vertebral arteries and runs caudally, the more rostral the location of ischemia, the more widespread or severe the symptoms. As the lateral horns are located between the T1 and L2 of the spinal cord, autonomic symptoms are not always present if ischemia does not involve this region.[2] Proprioception, vibratory sense, two-point discrimination, and fine touch are not affected in anterior cord syndrome and are carried by the dorsal column of the spinal cord, which is perfused by 2 posterior spinal arteries running in the posterior lateral sulci.[3]
Anterior spinal artery syndrome is a rare cause of spinal cord acute ischemic myelopathy. However, it is the most common cause of spinal cord infarcts.[4] The syndrome is caused by any etiology that decreases or affects blood flow to the anterior spinal artery, with aortic surgery and atherosclerotic disease being the most common causes. Diagnosis of anterior spinal artery syndrome is based on characteristic clinical features and confirmatory neuroimaging studies.[5] The prognosis is generally poor, as no acute management is available. Management focuses on treating the underlying cause and preventing complications. Current treatment follows the same guidelines for cerebral ischemia, atherosclerotic vascular disease, and acute traumatic spinal cord injury.[6]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Anterior spinal artery syndrome occurs from direct or indirect damage to the ventral spinal cord. A direct injury arises when the spinal cord is mechanically crushed or compressed, as in the case of an enlarging hematoma. Indirect injuries are the primary cause of anterior spinal artery syndrome caused by ischemia secondary to occlusion or hypoperfusion of the anterior spinal artery or its major branches, such as the radiculomedullary arteries.[7] The artery of Adamkiewicz, also known as arteria radicularis magna, is the most common location for occlusion.[8] The occlusion or hypoperfusion can lead to ischemia within the vascular distribution of the anterior spinal artery.[5]
The integrity of the aorta is critical for the proper perfusion of the ventral aspect of the spinal cord, and knowledge of vascular anatomy is required. The aorta gives rise to subclavian arteries, which in turn give rise to the vertebral arteries. The vertebral arteries give rise to the anterior spinal artery, which supplies the anterior two-thirds of the spinal cord. Therefore, any condition compromising the aorta, such as aortic dissection, may cause indirect injuries to the spinal cord.
Some risk factors associated with anterior spinal artery syndrome include smoking, hypertension, diabetes mellitus, dyslipidemia, and family or patient history of ischemic cerebrovascular accidents.[6][9] Anterior spinal artery syndrome can also result from iatrogenic damage to the aorta following surgical complications during attempted vascular repairs or with prolonged clamping of the aorta. Other less common causes of infarcts to the ventral cord are intrinsic arterial occlusion due to emboli or atheroma of the anterior spinal artery or the radicular branches.[1]
The anterior aspect of the spinal cord is particularly susceptible to ischemia because the supplying vessels are end arteries and typically have no anastomoses. Anterior spinal artery infarction may also occur due to impairment of perfusion through the arteria radicularis magna, the major contributor to the caudal two-thirds of the anterior spinal artery in the lower thoracic and upper lumbar region, T5 to L2, of the spinal cord. Other causes of disruption of blood flow in the anterior spinal artery are aneurysms, trauma, or syphilitic arteries.[10] Furthermore, hypotensive states, venous infarctions, multiple sclerosis, and cord impingement by fracture fragments are less common causes of vascular damage to the anterior cord.
Occlusive and Hypoperfusion Etiologies of Anterior Spinal Artery Syndrome
Occlusive and hypoperfusion etiologies of anterior spinal artery syndrome refer to the different mechanisms that can lead to reduced blood flow to the anterior spinal artery, resulting in spinal cord ischemia.
- Aortic surgery: Iatrogenic injury is the most common cause of anterior spinal artery syndrome. Factors that may increase the risk for complications are hypotension, prolonged cross-clamping of the aorta, increased spinal cord pressure, and occlusion of radiculomedullary arteries. These factors may decrease blood flow through the anterior spinal artery, leading to ischemia. Other complications may involve direct surgical injury to major radiculomedullary arteries.[2][6][11]
- Atherosclerotic disease: This disease is a common cause of anterior spinal artery syndrome due to the development of a thrombus or emboli that occludes the anterior spinal artery.[12]
- Thoracoabdominal aortic dissection: Dissection of the aorta frequently causes occlusion of radiculomedullary arteries.[9]
- Cardiac arrest: Cardiac arrest can cause decreased or absent blood flow to the anterior spinal artery.
- Cardiac emboli: This etiology is an infrequent cause of anterior spinal artery syndrome. The category includes infectious endocarditis, which may lead to septic emboli and occlusion of an anterior spinal artery or major radiculomedullary arteries.[13]
- Vasculitis: Vascular inflammation, such as polyarteritis nodosa, may rarely cause anterior spinal artery syndrome.
- Degenerative spine disease: Although a rare cause of anterior spinal artery syndrome, the literature describes patients with cervical spondylosis or thoracic disc herniation who present with anterior spinal artery syndrome. A large disc herniation can compress the anterior spinal artery and cause spinal cord infarction.[5][14]
- Vertebral fracture: This may cause anterior spinal artery syndrome through anterior cord impingement by a fracture fragment.[4]
- Fibrocartilaginous embolic myelopathy: This condition is sometimes known as idiopathic transverse myelitis. Migration of nucleus pulposus material into vessels supplying the anterior spinal artery may result in embolic infarction of the spinal cord and anterior spinal artery syndrome.[15][16][17]
- Hypotensive shock: Hypotension may lead to anterior spinal artery syndrome.
- Luetic aortitis: This condition was common during the syphilitic era but is rarely observed today.[6]
- Minimally invasive spine surgery: Surgical procedures, such as percutaneous balloon kyphoplasty, may cause anterior spinal artery syndrome.[18][19]
- Arteriovenous malformations: Spinal cord arteriovenous malformations may cause anterior spinal artery syndrome.[20]
- Digital subtraction angiography: This diagnostic and embolization procedure is a rare cause of anterior spinal artery syndrome, with an overall incidence of 1.4% to 6.5%.[7]
- Sickle cell disease: Due to thrombosis, sickle cell disease may lead to anterior spinal artery syndrome, among other complications.[21]
Epidemiology
Anterior spinal artery syndrome is a rare condition; statistical studies evaluating the incidence and prevalence of this condition are scarce.[6] Approximately 5% to 8% of the cases of acute spinal myelopathies are due to vascular causes such as spinal cord infarctions.[4][15] Spinal cord infarction accounts for 1.2% of all strokes and has an incidence of 3.1 per 100,000 persons in the United States.[28] Studies with small groups have described spinal cord infarction in patients with an average age between 50 and 70.[29] Anterior spinal artery syndrome is the most common of all spinal cord infarction syndromes described in some studies in up to 87.2% of the cases.[12][30] No statistical evidence is available for a sex preference in anterior spinal artery syndrome; however, a few studies have reported that older women with anterior spinal artery syndrome have the worst prognosis.[9]Given the lack of available data and the rarity of this syndrome, further studies are required.
Pathophysiology
The underlying pathophysiology of anterior spinal artery syndrome is better understood by having familiarity with the anatomy. The anterior spinal artery supplies the anterior two-thirds of the medulla and spinal cord and is formed at the level of the foramen magnum by 2 arteries branching from their respective vertebral arteries. This artery is uninterrupted within the anterior median sulcus up to the conus medullaris. Although the anterior spinal artery is a continuous artery, multiple anastomotic networks are supplied by the anterior medullar or radiculomedullary arteries. The anterior spinal artery varies in caliber along the course, with the smallest diameter at the thoracic level, making this segment most vulnerable to ischemia. The largest diameter of this artery is at the lumbar level.
The anterior spinal artery provides small sulcal and penetrating arteries that enter the spinal cord's body at each level to supply blood to the spinal cord. Each spinal cord level contains a radicular artery that enters through the intervertebral foramen, primarily supplying the nerve root and dura. At some levels, they are enlarged and include embryonic connections with the anterior spinal artery, making them radiculomedullary arteries. The anterior spinal artery connections from the radiculomedullary arteries can be variable in the cervical regions. Typically, the cut-off is above the aortic arch, from either the vertebral or subclavian arteries. At the thoracic and lumbar regions, the intercostal arteries give rise to the radiculomedullary arteries, supplying the anterior spinal artery and the spinal cord at and below their entry-level.
On average, the spinal cord has 7 to 8 radiculomedullary arteries. The largest radiculomedullary artery is the great anterior radiculomedullary artery or artery of Adamkiewicz, also known as arteria radicularis magna. The arteria radicularis magna is the largest and most common radiculomedullary artery occluded, resulting in the development of anterior spinal artery syndrome. This artery originates from the intercostal artery, most commonly from the left side in 70% to 80% of patients; however, the origin may vary. The most common site of origin is the T9 to T12 vertebra in 75% of patients, followed by the T5 to T8 vertebra in 15% of patients and the L1 to L2 vertebra in 10%. Digital subtraction angiography describes the arteria radicularis magna as a straight vessel with a hairpin turn and subsequent communication with the anterior spinal artery coursing along the spine's midline.[7]
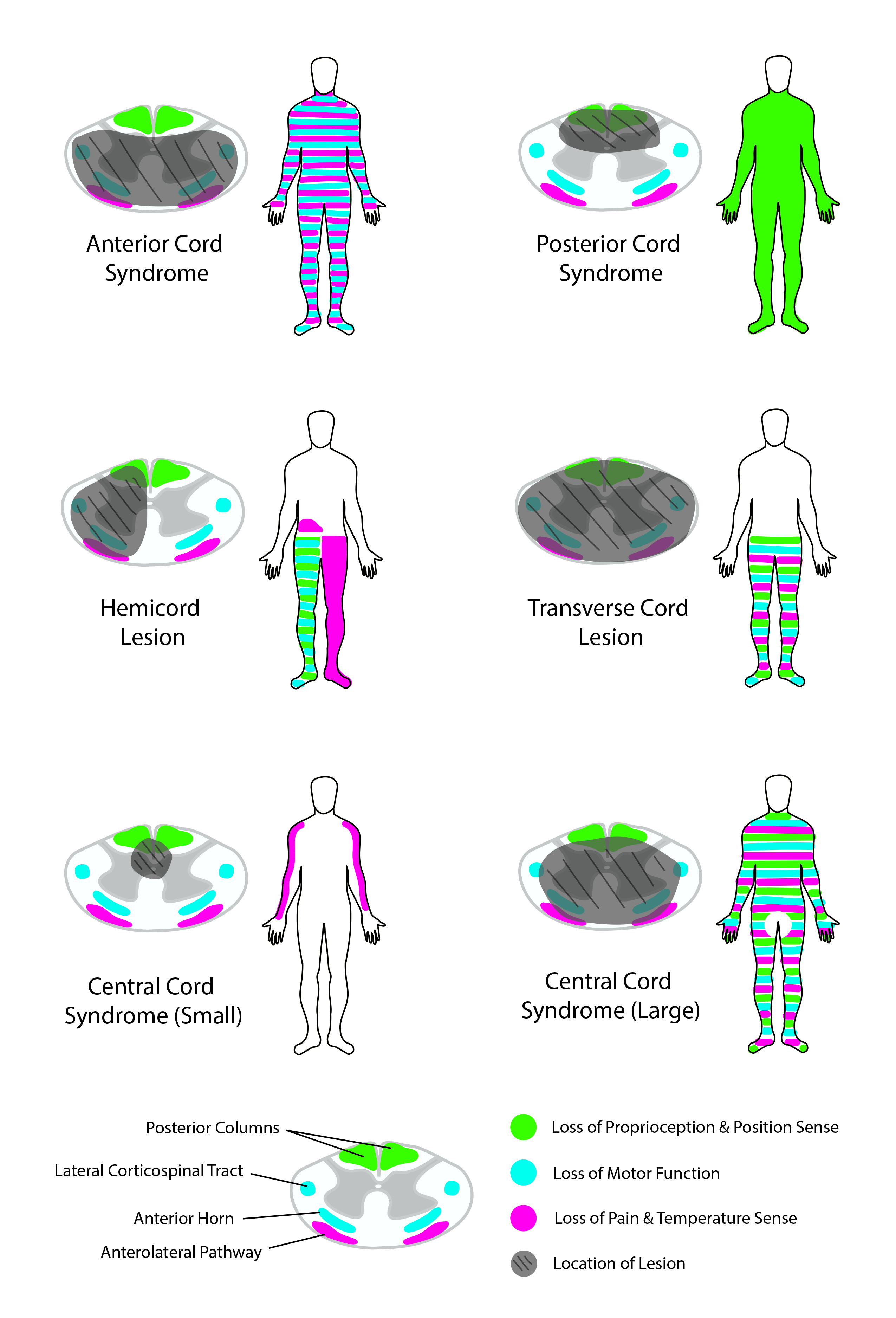
Understanding the clinical presentation of anterior spinal artery syndrome requires knowledge of the anatomy of the spinal cord to explain the pathophysiology behind this condition. The spinal cord is subdivided into 3 portions—the posterior part, anterolateral portion, and anteromedial portion. The posterior part of the cord contains the fasciculus gracilis and cuneatus, which are responsible for proprioception, vibration sense, and fine touch. This area is supplied by the 2 posterior spinal arteries that run in the spinal cord's posterior lateral sulcus, which explains why these modalities are preserved in anterior spinal artery syndrome. The anterolateral portion of the spinal cord contains the spinothalamic and spinocerebellar tracts responsible for transmitting pain and temperature sensation from the extremities to the brain. These portions are watershed areas in the spine, and the level of dysfunction depends on the level of anterior spinal artery syndrome ischemic insult. The sensory deficits may not be directly related to the exact injury level because spinothalamic tracts slope 2 or 3 levels upwards before crossing at the anterior commissure of the spinal cord. The anteromedial portion contains the corticospinal and corticobulbar tracts responsible for transmitting motor function from the brain to the extremities.[2] The lateral horns exist from the T1 level to the L2 level. They contain the cell bodies of the sympathetic nervous system. The anterior spinal artery is responsible for the vascular supply to these areas. If an infarction occurs, the sympathetic network is affected, and patients may present with neurogenic bladder and impaired bowel and sexual function.[9]
When anterior spinal artery blood flow is reduced or absent, several events occur at the cellular level. The disruption of blood flow initiates a signaling cascade that results in neuronal destruction. The cascade begins with the ischemic insult when blood flow and oxygen to the spinal cord decrease. Subsequently, astrocytes and microglia are promptly activated, leading to tissue edema, inflammation, disruption of the blood-brain barrier, and the influx of neutrophils. The disruption of blood flow leads to ionic pump failure and anoxic depolarization, which leads to an increased concentration of intracellular calcium and glutamate. The ionic pump failure and anoxic depolarization, in part, produce increased activation of the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors and N-methyl-D-aspartate receptor, alteration in the metabolic and mitochondrial activity, which ultimately causes cell death. Furthermore, the increased intracellular calcium produces free radicals, elevates nitric oxide production, and causes cytotoxic edema, further contributing to cell death. A later reperfusion injury occurs during the subacute stage, where cell death occurs secondary to the inflammatory oxidative response with the activation of the apoptotic cascade.[4]
History and Physical
Clinical Features
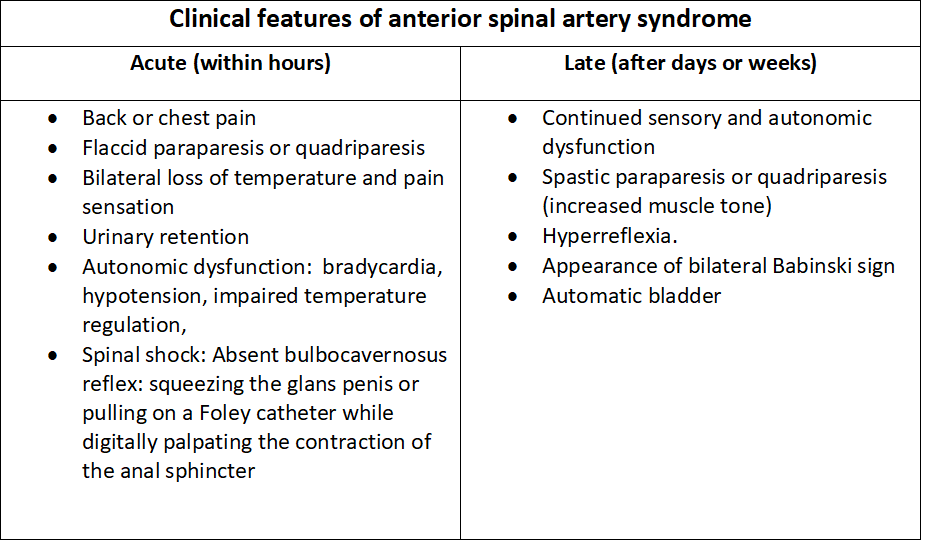
Due to the ischemic etiology of anterior spinal artery syndrome, patients commonly have an acute onset of motor dysfunction and loss of pain and temperature sensation below the infarction level following the insult (see Image. Clinical Features of Anterior Spinal Artery Syndrome). Typically, the first presenting symptom is acute back pain, whose location most of the time corresponds with the level of the spinal cord injury. Below this level, bilateral loss or dysfunction of motor function and sensation occurs; however, proprioception, vibratory sense, fine touch, and two-point discrimination are preserved.[2][5][6] These symptoms are almost always bilateral because both halves of the anterior spinal cord receive vascular supply from a midline anterior spinal artery. Motor dysfunction severity varies from paraplegia to quadriplegia, depending on the infarction location (see Image. Corticospinal Tract Lesion Locations).
Often, the location of infarction includes the lateral horns within levels T1 to L2 of the spinal cord, causing autonomic dysfunction, which most commonly results in neurogenic bladder and bowel, hypotension, bradycardia, and sexual dysfunction.[4] If the injury is at a high cervical level, dysfunction of the phrenic nerve may lead to respiratory failure.[31][32] Rare cases of anterior cord syndrome with unilateral symptomatology have been reported; this may be due to occlusion of unilateral sulcal arteries or collateralization from a posterior spinal artery.[9][33] As anterior spinal artery syndrome has no acute treatment, late signs and symptoms, including spasticity, hyperreflexia, neurogenic bladder, and sexual dysfunction, are typically present. The following clinical features are findings observed in patients with acute- and late-onset syndromes.
Acute clinical findings: Acute-onset symptoms appear immediately after the ischemic insult.
- Acute back pain at the level of injury
- Bilateral flaccid paraparesis or quadriparesis or plegia
- Loss of pain and temperature
- Preservation of proprioception, vibratory sense, fine touch, and two-point discrimination
- Autonomic dysfunction with hypotension, bradycardia, and impaired temperature regulation
- Respiratory failure
- Spinal shock
Late clinical findings: Late-onset symptoms develop gradually over days to weeks after the initial injury.
- Continued or permanent motor and sensory dysfunction
- Bilateral spastic paraparesis or quadriparesis or plegia
- Hyperreflexia
- Bilateral Babinski signs
- Neurogenic bladder and bowel
- Sexual dysfunction
Evaluation
Characteristic clinical features and confirmatory neuroimaging diagnostic studies primarily diagnose anterior spinal artery syndrome.
Diagnostic Imaging Studies
The gold-standard diagnostic study for anterior spinal artery syndrome is magnetic resonance imaging (MRI) of the spine. Although results can be negative in the first 24 hours, MRIs are indicated to confirm the diagnosis. The hallmark finding is a hyperintensity at anterior horns in the T2-weighted image. Characteristic MRI features of acute spinal cord ischemia are a thin pencil-like hyperintense region that extends vertically, involving multiple spinal levels in the sagittal view. Axial MRI studies show a central T2-hyperintense signal on either side of the median fissure because of relative sparing of the peripheral and posterior cords due to collateral vessels from the vascular pial plexus and posterior spinal arteries. Occasionally, a central T2-hyperintense signal resembling snake or owl's eyes appears in the anterior spinal cord on either side of the median fissure. Although this finding is considered specific to spinal cord infarction, the association with compression myelopathy and various infectious or inflammatory spinal conditions is common.[34] Other findings that suggest a spinal cord infarct are a T1-weighted hypointensity at the area of injury, spinal cord expansion at the injury site due to early signs of inflammation or edema where diffusion-weighted images may help distinguish between ischemia and inflammation, and signs of vertebral body infarction that may not always present. Still, when vertebral body infarction signs are present, the specificity for cord infarction is increased.[6] In addition, MRI studies identify possible etiologies behind anterior spinal artery syndrome, such as herniated discs or tumors.
MRI angiography or computed tomography (CT) angiography are appropriate adjunctive modalities for further delineating vascular pathology. MRI angiography may help further delineate and visualize spinal vascular pathology. CT angiography helps identify the anterior spinal artery and provides adequate visualization of bone anatomy. However, CT angiography also exposes the patient to radiation doses and nephrotoxic agents. In addition, this modality may help rule out an aortic or vertebral artery dissection.[4] Digital subtraction angiography can also be used, although it is not preferred. However, it delineates the vascular anatomy, which is necessary if an interventional procedure is required.[35] Digital subtraction angiography is an ideal imaging technique for diagnosing spinal vascular lesions and identifying aortic or vertebral dissections.[5]
Additional Diagnostic Studies
Laboratory studies are performed to screen for conditions that cause ischemic damage to vessels, such as diabetes mellitus, atherosclerosis, substance abuse, and hypercholesterolemia. These tests include a complete blood cell count, erythrocyte sedimentation rate, electrolytes, glucose, creatinine, cholesterol levels, liver enzymes, protein electrophoresis, and coagulation tests. Serological tests, including borreliosis, Lyme disease, syphilis, HIV, human T-cell lymphotropic virus, and herpes virus, are necessary when considering infectious etiologies of infarction. Screening for antinuclear and antineutrophil cytoplasmic antibodies can be considered in patients with potential causes of spinal cord infarction.
Current treatment is primarily supportive and follows the same guidelines as those for cerebral ischemia, atherosclerotic vascular disease, and acute traumatic spinal cord injury. If the underlying cause is treatable, the promptness of treatment is the most important factor for prognosis. No effective treatment is available to stop or revert anterior spinal artery syndrome. The primary focus is to address the underlying cause, including fever and glycemic control, management of anticoagulation, antiplatelet, and thromboprophylaxis therapy.[36] Protecting airways and maintaining blood pressure support in an acute setting is paramount. If the patient is hypotensive, then blood pressure must be raised using phenylephrine, norepinephrine, and high-dose dopamine. For blood pressure reduction, preferred medications include labetalol, esmolol, and nicardipine.[30]
The use of high-dose corticosteroids for patients with anterior spinal artery syndrome is ineffective, with few reports showing mild improvement and most patients showing no improvement.[2][5] In cases where atherosclerotic or embolic disease is the underlying cause, patients treated with intravenous thrombolysis within 4.5 hours of symptom onset have reported excellent improvement without complications.[18] In theory, spinal cord strokes can be treated similarly to cerebral strokes; however, due to the need for further studies, this approach is not currently considered the standard of care. Supportive treatment focuses on addressing acute and late symptoms or complications, including:
- Neurogenic hypotension is treated with intravenous fluids and vasopressors
- Manage phrenic nerve impairment and dysfunction of the diaphragm in high cervical lesions with mechanical ventilation
- Neurogenic urinary retention can be managed with bladder catheterization
- Deep venous thrombosis prevention prophylaxis
- Pressure sore prevention
- Gastrointestinal stress ulcer prevention
- Physical, occupational, and psychological therapies
The most common cause of anterior spinal artery syndrome is aortic surgery; therefore, treatment should focus on preventing iatrogenic anterior spinal artery syndrome. A protocol to prevent anterior spinal artery syndrome during aortic surgery has been established. This protocol involves increasing blood flow to the anterior spinal artery by increasing mean arterial pressure and placing a lumbar drain to remove cerebrospinal fluid, thereby decreasing pressure on the spinal cord.[4][9] Furthermore, excluding an arteriovenous malformation or aortic dissection as the underlying cause before thrombolysis is essential due to a thrombolytic contraindication.[6]
Treatment / Management
Current treatment is primarily supportive and follows the same guidelines as those for cerebral ischemia, atherosclerotic vascular disease, and acute traumatic spinal cord injury. If the underlying cause is treatable, the promptness of treatment is the most important factor for prognosis. No effective treatment is available to stop or revert anterior spinal artery syndrome. The primary focus is to address the underlying cause, including fever and glycemic control, management of anticoagulation, antiplatelet, and thromboprophylaxis therapy.[36] Protecting airways and maintaining blood pressure support in an acute setting is paramount. If the patient is hypotensive, then blood pressure must be raised using phenylephrine, norepinephrine, and high-dose dopamine. For blood pressure reduction, preferred medications include labetalol, esmolol, and nicardipine.[30] (B3)
The use of high-dose corticosteroids for patients with anterior spinal artery syndrome is ineffective, with few reports showing mild improvement and most of them showing no improvement.[2][5][5] In cases where atherosclerotic or embolic disease is the underlying cause, patients treated with intravenous thrombolysis within 4.5 hours of symptom onset have reported excellent improvement without complications.[18] In theory, spinal cord strokes can be treated similarly to cerebral strokes; however, due to the need for further studies, this approach is not currently considered the standard of care. Supportive treatment focuses on addressing acute and late symptoms or complications, including:(B3)
- Neurogenic hypotension is treated with intravenous fluids and vasopressors
- Phrenic nerve impairment and dysfunction of the diaphragm in high cervical lesions are typically managed with mechanical ventilation
- Neurogenic urinary retention can be managed with bladder catheterization
- Deep venous thrombosis prevention prophylaxis
- Pressure sore prevention
- Gastrointestinal stress ulcer prevention
- Physical, occupational, and psychological therapy
The most common cause of anterior spinal artery syndrome is aortic surgery; therefore, treatment should focus on preventing iatrogenic anterior spinal artery syndrome. A protocol to prevent anterior spinal artery syndrome during aortic surgery has been established. This protocol involves increasing blood flow to the anterior spinal artery by increasing mean arterial pressure and placing a lumbar drain to remove cerebrospinal fluid, thereby decreasing pressure at the spinal cord.[4][9] Furthermore, excluding an arteriovenous malformation or aortic dissection as the underlying cause before thrombolysis is essential due to a thrombolytic contraindication.[6](B3)
Differential Diagnosis
Differential diagnoses that should be considered when evaluating a patient with suspected anterior spinal artery syndrome include:
- Transverse myelitis, which has a slower onset
- Multiple sclerosis has similar MRI findings; however, patients show cranial and ocular manifestations
- On imaging, venous congestive myelopathy may demonstrate prominent enlarged pial veins with central, peripheral white matter changes
- Spinal cord neoplasms have a slower onset
- Spinal cord arteriovenous malformations
- Disk herniation
- Epidural hematoma
- Epidural abscess
- Spinal cord compressive pathologies
- Central cord syndrome
- Dorsal cord syndrome
- Brown-Séquard syndrome
- Guillain-Barré syndrome
- Conus medullaris syndrome
- Cauda equina syndrome (see Image. Spinal Cord Syndromes)
Pertinent Studies and Ongoing Trials
Current preclinical research is exploring promising pharmacological agents as a possible treatment for ischemic spinal cord injuries. Neuroprotective therapies limit secondary injury after the initial damage by modulating inflammation, reducing cell death, and guarding against excitotoxicity. Currently, no proven pharmacological therapies for human spinal cord injury provide neuroprotection. Studies use propofol, thymoquinone, minocycline, erythropoietin, and curcumin as possible neuroprotective pharmacologic agents. Antioxidants protect against the oxidative damage caused by secondary neurodegeneration after acute spinal cord injury. These antioxidants also stabilize mitochondrial bioenergetics, protect neurons, and achieve modest neurological recovery.
Emerging research is focusing on measures that could help prevent ischemic spinal cord injury during vascular surgery. Hypothermia preserves tissue integrity during periods of interrupted blood flow; animal studies suggest that hypothermia suppresses excitatory synaptic transmission and reduces injury to neurons. Hyperbaric oxygen increases tissue oxygenation and metabolism in penumbral tissue. Ischemic preconditioning is a way to prepare tissue for the ischemic state to render resistance to a hypoxic insult. Numerous preclinical studies have examined the molecular pathways and potential benefits of pre- and post-ischemic preconditioning with encouraging results. Other studies explore methods to counteract apoptosis, which leads to neuronal cell death. Many of the current treatments for spinal cord infarcts show only modest improvements in the functional outcome if used as monotherapy, which is probably attributable to the development of permanent neurological deficits. Thus, future clinical trials should consider a combined and multimodal treatment approach.[36]
Prognosis
The long-term prognosis of anterior spinal artery syndrome is only briefly described in the literature. In general, the prognosis is poor, but a study mentioned that spinal strokes have a better prognosis compared to cerebral strokes, which also involve cognitive damage.[6] The primary etiology behind anterior spinal artery syndrome is important for prognosis. If treated rapidly and adequately, less common etiologies, such as disc herniation, can completely alleviate symptoms.[5] Spinal cord infarction has an overall mortality rate of 9% to 23% and typically occurs shortly after the injury. A review of patients with spinal cord infarction reported that 24% did not improve, and only 20% improved significantly.[37] Two other studies indicated that only 1% to 5% of patients recovered completely.[32][38]
Most survivors experience varying degrees of functional motor and sensory dysfunction. Patients with more severe presentation and without improvement in the first 24 hours after insult result in a poorer prognosis.[4] In less severe cases, favorable outcomes with improvement in neurological deficits are possible, with a small portion of patients regaining the ability to walk. In others, functional improvement can develop slowly over the years.[9] Multiple factors may affect the prognoses.[39][40][41]
- Etiology: Conditions such as aortic dissection or rupture and high cervical lesions carry a greater risk of death
- Symptom severity at initial presentation: Severe initial presenting symptoms have worse outcomes
- Improvement in the first 24 hours: No improvement within the first 24 hours has the worst outcomes
- Female sex: Correlates with poorer outcomes
- Older adults: Associated with poorer outcomes
Complications
Patients with anterior spinal artery syndrome develop multiple complications associated with the condition. These complications arise due to the highly incapacitating injury to the spinal cord. The complications and presenting symptoms depend on the level of the spinal cord where the insults occur. The majority of life-threatening complications in anterior spinal artery syndrome present shortly after the ischemic insult or during the hospitalization period, including:
- Hypotension: Involvement of lateral horns T1 to L2 may lead to shock, which can be fatal if not treated or corrected
- Respiratory failure: High cervical-level injury involving the phrenic nerve (C3 to C5) may lead to prolonged mechanical ventilation
- Bradycardia: This can lead to cardiac arrest, for which patients may require atropine or permanent electrical pacing
- Infections such as urinary tract infections, pneumonia, and bacteremia
- Electrolyte imbalances
- Renal failure
- Depression [2]
The prognosis for recovery from anterior spinal artery syndrome is dismal, with poor outcomes. Many complications develop as most patients do not recover their motor and sensory dysfunction and experience prolonged immobilization, including:
- Permanent paraparesis or quadriparesis or plegia
- Deep venous thrombosis
- Pulmonary embolism
- Pressure ulcers with a 50% lifetime risk
- Neurogenic bowel or bladder
- Sexual dysfunction
- Neuropathic pain
- Gastrointestinal stress ulcers
- Gastrointestinal dysmotility
- Chronic pain in 79% of the patients
- Increased risk of osteoporosis due to prolonged immobilization
- Spasticity
- Psychological depression and other conditions
- Economic issues
- Dependency on others in activities of daily living [4][6][9][41][42][43]
Postoperative and Rehabilitation Care
The successful long-term management of anterior spinal artery syndrome depends on adequate and comprehensive rehabilitation before and after discharge. Rehabilitation focuses on improving the quality of life, independence, and prevention of secondary complications. Although important, the rehabilitation focuses on improving motor function and includes physical, occupational, vocational, and neuropsychological therapies.[2][4]
As postoperative treatments for anterior spinal artery syndrome are often ineffectual, measures to prevent perioperative cord ischemia should include avoiding anemia, systemic hypotension, and hyperextension or hyperflexion during positioning. Efforts to minimize any surgical accidents resulting in vascular dissection are imperative.[44] During procedures that involve interventions of the thoracoabdominal aorta, early detection of spinal cord ischemia can be achieved by neuromonitoring with evoked potentials in the anesthetized patient and with the neurological examination in the awake patient.[45]
Tetraplegia and paraplegia are among the most devastating consequences of ventral cord syndrome. These movement impediments have a significant medical, social, and financial cost to the individual and society. As no current treatment can reverse anterior spinal artery syndrome, patients must receive rehabilitation focused on improving independence and quality of life while preventing secondary complications. Patients are candidates for several forms of rehabilitation, including but not limited to physical, occupational, vocational, and neuropsychological therapies.
Consultations
To effectively manage anterior spinal artery syndrome, a multidisciplinary approach involving various specialists such as neurologists, neuroradiologists, neurointensivists, pneumologists, urologists, psychologists, psychiatrists, social workers, and specialists in physical medicine and rehabilitation is crucial.
Deterrence and Patient Education
Although anterior spinal artery syndrome is unpreventable and typically nonreversible, the most common underlying causes, such as aortic aneurysm or dissection and atherosclerotic disease, that lead to the development of anterior spinal artery syndrome are preventable. The patient's education should focus on primary disease prevention, especially on preventing major risk factors, such as hypertension, dyslipidemia, diabetes mellitus, and smoking, including healthy nutrition, exercise, and smoking cessation. If a patient develops morbidities that are risk factors, starting medical management is crucial. Strict management of risk factors helps prevent recurrence or worsening functionality.[9]
During hospitalization, patients require guidance on adapting to life after being diagnosed with anterior spinal artery syndrome. Follow-up visits also provide an opportunity for support and patient education. The journey of adjustment to disability is an evolving, dynamic, general process from which the patient progresses to an optimal state of person-environment congruence. The clinical team should focus on instructing and coaching the patient. Patients must receive training in developing effective coping strategies.
Results from a study by Kennedy et al showed that 45 patients who participated in a coping training program had lower scores for depression and anxiety compared to a control group that received the standard of care.[46] Hospital discharge provides clinicians an excellent opportunity to educate patients, family, friends, and caretakers. The medical team must assess the options and equipment available to facilitate the patient's life and integration into society. Discharge planning must consider preparing for emergencies and complications, establishing follow-up appointments and support, providing the patient with appropriate medical equipment, and conducting a home assessment.
Psychological resources protect and promote the inner health potentials of a person, which represents a means to deal with challenging situations. These resources may include a person's abilities, skills, knowledge, experience, talents, strengths, and behavioral patterns. Clinicians must help patients strengthen their psychological resources to support a successful adjustment.[47] The patient must obtain counsel on the importance of sports participation and exercise to improve health. Before engaging in physical activity, an appropriate screening should determine the exercise prescription, identify precautions, and provide necessary modifications to physical activity. Education also centers around other aspects of patient life, such as employment and social relationships. The medical team needs to assess and provide options and modifications for transportation.
Pearls and Other Issues
Key facts to bear in mind when managing anterior spinal artery syndrome include:
- The syndrome is caused by ischemia at the anterior two-thirds of the spinal cord due to the anterior spinal artery's reduced blood flow.
- The most common etiology is aortic surgery.
- Patient education about preventable risk factors is essential for preventable conditions such as aortic aneurysm or dissection.
- The syndrome presents as acute back pain at the level of injury.
- The patient shows bilateral motor dysfunction and loss of pain and temperature, with preservation of proprioception and vibratory sense.
- Patients may have autonomic dysfunction.
- MRI is the gold standard for diagnosis.
- No acute treatment is available; management focuses on treating presenting symptoms and complications.
- Most patients remain functionally incapacitated.
- Physical and emotional support is essential.
Enhancing Healthcare Team Outcomes
Although rare, anterior spinal artery syndrome is a severe, incapacitating syndrome that typically affects multiple organs and produces poor outcomes. A team approach to managing this condition after the arrival in the emergency room results in the best possible outcome. The emergency room clinician must rapidly recognize the problem and obtain imaging that confirms the diagnosis, which leads to early treatment. Neurologists play a vital role in evaluating and treating spinal cord injury, as do vascular surgeons if an aortic etiology is suspected, as do neurosurgeons and orthopedic spine surgeons if a structural etiology is suspected. Nurses can identify and prevent complications such as pressure sores, urinary retention, and deep venous thromboses. Pharmacists can help manage multiple medications for patients with spinal cord infarction, preventing complications.
The interprofessional team is necessary for managing anterior spinal artery syndrome. Nurses are vital members whose role is crucial in identifying and preventing complications, such as pressure ulcers and urinary retention. Pharmacists play a crucial role as patients with anterior spinal artery syndrome often require multiple medications, leading to polypharmacy, especially during the acute period. The physical and occupational therapist's role is essential in determining a patient's prognosis, especially in the long term. Physical and occupational therapies also play a crucial prognostic role in helping patients with rehabilitation and improving their functional status. Patients with spinal cord injury have experienced improvements in lifespan and functional independence due to advancements in multidisciplinary patient care.[48] In addition, social workers treat mental, social, economic, and physical disabilities. In patients with spinal cord infarction, functional independence and overall lifespan have improved due to improvements in treatment strategies.[9]
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Yogendranathan N, Herath HMMTB, Jayamali WD, Matthias AT, Pallewatte A, Kulatunga A. A case of anterior spinal cord syndrome in a patient with unruptured thoracic aortic aneurysm with a mural thrombus. BMC cardiovascular disorders. 2018 Mar 5:18(1):48. doi: 10.1186/s12872-018-0786-4. Epub 2018 Mar 5 [PubMed PMID: 29506472]
Level 3 (low-level) evidenceKlakeel M, Thompson J, Srinivasan R, McDonald F. Anterior spinal cord syndrome of unknown etiology. Proceedings (Baylor University. Medical Center). 2015 Jan:28(1):85-7 [PubMed PMID: 25552812]
Level 3 (low-level) evidenceMcKinley W, Hills A, Sima A. Posterior cord syndrome: Demographics and rehabilitation outcomes. The journal of spinal cord medicine. 2021 Mar:44(2):241-246. doi: 10.1080/10790268.2019.1585135. Epub 2019 Apr 2 [PubMed PMID: 30939076]
Santana JA, Dalal K. Ventral Cord Syndrome. StatPearls. 2024 Jan:(): [PubMed PMID: 31082055]
Peng T, Zhang ZF. Anterior Spinal Artery Syndrome in a Patient with Cervical Spondylosis Demonstrated by CT Angiography. Orthopaedic surgery. 2019 Dec:11(6):1220-1223. doi: 10.1111/os.12555. Epub 2019 Nov 3 [PubMed PMID: 31680448]
Müller KI, Steffensen LH, Johnsen SH. Thrombolysis in anterior spinal artery syndrome. BMJ case reports. 2012 Sep 7:2012():. doi: 10.1136/bcr-2012-006862. Epub 2012 Sep 7 [PubMed PMID: 22962400]
Level 3 (low-level) evidencePadgett M, Abi-Jaoudeh N, Benn BS, Rahimian R, Nelson K. Anterior Cord Syndrome after Embolization for Malignant Hemoptysis. Seminars in interventional radiology. 2019 Jun:36(2):111-116. doi: 10.1055/s-0039-1688424. Epub 2019 May 22 [PubMed PMID: 31123382]
Perez Perez VH, Hernesniemi J, Small JE. Anatomy of the Great Posterior Radiculomedullary Artery. AJNR. American journal of neuroradiology. 2019 Dec:40(12):2010-2015. doi: 10.3174/ajnr.A6304. Epub 2019 Nov 21 [PubMed PMID: 31753838]
Pearl NA, Weisbrod LJ, Dubensky L. Anterior Cord Syndrome (Archived). StatPearls. 2024 Jan:(): [PubMed PMID: 32644543]
Ng KS, Abdul Halim S. Anterior spinal cord syndrome as a rare complication of acute bacterial meningitis in an adult. BMJ case reports. 2018 Oct 24:2018():. pii: bcr-2018-226082. doi: 10.1136/bcr-2018-226082. Epub 2018 Oct 24 [PubMed PMID: 30361450]
Level 3 (low-level) evidenceWang MX, Smith G, Albayram M. Spinal cord watershed infarction: Novel findings on magnetic resonance imaging. Clinical imaging. 2019 May-Jun:55():71-75. doi: 10.1016/j.clinimag.2019.01.023. Epub 2019 Jan 31 [PubMed PMID: 30763904]
Pikija S, Mutzenbach JS, Kunz AB, Nardone R, Leis S, Deak I, McCoy MR, Trinka E, Sellner J. Delayed Hospital Presentation and Neuroimaging in Non-surgical Spinal Cord Infarction. Frontiers in neurology. 2017:8():143. doi: 10.3389/fneur.2017.00143. Epub 2017 Apr 12 [PubMed PMID: 28446898]
Mahtabfar A, Eshraghi H, D'Souza M, Berrigan W, Casey K. A Case of Anterior Spinal Artery Syndrome Caused by Streptococcus mitis Endocarditis. Case reports in medicine. 2018:2018():9658120. doi: 10.1155/2018/9658120. Epub 2018 Jan 29 [PubMed PMID: 29560019]
Level 3 (low-level) evidenceGhias F, Zaidi MH, Oak K, Lai YJ. Poster 431 Anterior Spinal Artery Syndrome Caused by Traumatic Disk Herniation: A Case Report. PM & R : the journal of injury, function, and rehabilitation. 2016 Sep:8(9S):S301-S302. doi: 10.1016/j.pmrj.2016.07.356. Epub 2016 Sep 24 [PubMed PMID: 27673181]
Level 3 (low-level) evidenceGupta M, Chhabra HS. Nucleus Polposus Embolism Causing Anterior Spinal Artery Occlusion: A Rare but Possible Cause of Fibrocartilaginous Embolic Myelopathy. International journal of spine surgery. 2020 Jun:14(3):391-396. doi: 10.14444/7051. Epub 2020 Jun 30 [PubMed PMID: 32699762]
NAIMAN JL, DONOHUE WL, PRICHARD JS. Fatal nucleus pulposus embolism of spinal cord after trauma. Neurology. 1961 Jan:11():83-7 [PubMed PMID: 13727534]
AbdelRazek MA, Mowla A, Farooq S, Silvestri N, Sawyer R, Wolfe G. Fibrocartilaginous embolism: a comprehensive review of an under-studied cause of spinal cord infarction and proposed diagnostic criteria. The journal of spinal cord medicine. 2016:39(2):146-54. doi: 10.1080/10790268.2015.1116726. Epub 2015 Dec 17 [PubMed PMID: 26833287]
Bredow J, Oppermann J, Keller K, Beyer F, Boese CK, Zarghooni K, Sobottke R, Eysel P, Siewe J. Anterior spinal artery syndrome: reversible paraplegia after minimally invasive spine surgery. Case reports in orthopedics. 2014:2014():205732. doi: 10.1155/2014/205732. Epub 2014 Aug 20 [PubMed PMID: 25210639]
Level 3 (low-level) evidenceTsai YD, Liliang PC, Chen HJ, Lu K, Liang CL, Wang KW. Anterior spinal artery syndrome following vertebroplasty: a case report. Spine. 2010 Feb 15:35(4):E134-6. doi: 10.1097/BRS.0b013e3181b52221. Epub [PubMed PMID: 20110843]
Level 3 (low-level) evidenceLevin KH, Daube JR. Spinal cord infarction: another cause of "lumbosacral polyradiculopathy". Neurology. 1984 Mar:34(3):389-90 [PubMed PMID: 6322047]
Level 3 (low-level) evidenceEdwards A, Clay EL, Jewells V, Adams S, Crawford RD, Redding-Lallinger R. A 19-year-old man with sickle cell disease presenting with spinal infarction: a case report. Journal of medical case reports. 2013 Aug 23:7():210. doi: 10.1186/1752-1947-7-210. Epub 2013 Aug 23 [PubMed PMID: 23972124]
Level 3 (low-level) evidenceKhoueiry M, Moussa H, Sawaya R. Spinal cord infarction in a young adult: What is the culprit? The journal of spinal cord medicine. 2021 Nov:44(6):1015-1018. doi: 10.1080/10790268.2019.1671077. Epub 2019 Oct 9 [PubMed PMID: 31596188]
Lin WP, Kuan TS, Lin CI, Hsu LC, Lin YC. Spinal cord infarction during physical exertion due to polycythemia vera and aortoiliac occlusive disease: A case report. Medicine. 2018 Aug:97(35):e12181. doi: 10.1097/MD.0000000000012181. Epub [PubMed PMID: 30170466]
Level 3 (low-level) evidenceFarrell CM, Cucu DF. Cocaine-Related Acute Spinal Cord Infarction. Rhode Island medical journal (2013). 2018 Feb 2:101(1):28-29 [PubMed PMID: 29393308]
Mody CK, Miller BL, McIntyre HB, Cobb SK, Goldberg MA. Neurologic complications of cocaine abuse. Neurology. 1988 Aug:38(8):1189-93 [PubMed PMID: 3399066]
Level 3 (low-level) evidenceSchreiber AL, Formal CS. Spinal cord infarction secondary to cocaine use. American journal of physical medicine & rehabilitation. 2007 Feb:86(2):158-60 [PubMed PMID: 17167348]
Level 3 (low-level) evidenceJumma O, Koulaouzidis A, Ferguson IT. Cocaine-induced spinal cord infarction. Postgraduate medical journal. 2008 Jul:84(993):391. doi: 10.1136/pgmj.2007.062224. Epub [PubMed PMID: 18716023]
Level 3 (low-level) evidenceQureshi AI, Afzal MR, Suri MFK. A Population-Based Study of the Incidence of Acute Spinal Cord Infarction. Journal of vascular and interventional neurology. 2017 Jun:9(4):44-48 [PubMed PMID: 28702119]
Rubin MN, Rabinstein AA. Vascular diseases of the spinal cord. Neurologic clinics. 2013 Feb:31(1):153-81. doi: 10.1016/j.ncl.2012.09.004. Epub [PubMed PMID: 23186899]
Schneider GS. Anterior spinal cord syndrome after initiation of treatment with atenolol. The Journal of emergency medicine. 2010 Jun:38(5):e49-52. doi: 10.1016/j.jemermed.2007.08.061. Epub 2008 Jul 2 [PubMed PMID: 18597977]
Level 3 (low-level) evidenceWeidauer S, Nichtweiss M, Lanfermann H, Zanella FE. Spinal cord infarction: MR imaging and clinical features in 16 cases. Neuroradiology. 2002 Oct:44(10):851-7 [PubMed PMID: 12389137]
Level 3 (low-level) evidenceFoo D, Rossier AB. Anterior spinal artery syndrome and its natural history. Paraplegia. 1983 Feb:21(1):1-10 [PubMed PMID: 6835686]
Level 3 (low-level) evidenceNovy J, Carruzzo A, Maeder P, Bogousslavsky J. Spinal cord ischemia: clinical and imaging patterns, pathogenesis, and outcomes in 27 patients. Archives of neurology. 2006 Aug:63(8):1113-20 [PubMed PMID: 16908737]
Level 2 (mid-level) evidenceKunam VK, Velayudhan V, Chaudhry ZA, Bobinski M, Smoker WRK, Reede DL. Incomplete Cord Syndromes: Clinical and Imaging Review. Radiographics : a review publication of the Radiological Society of North America, Inc. 2018 Jul-Aug:38(4):1201-1222. doi: 10.1148/rg.2018170178. Epub [PubMed PMID: 29995620]
Oyinkan Marquis B, Capone PM. Myelopathy. Handbook of clinical neurology. 2016:136():1015-26. doi: 10.1016/B978-0-444-53486-6.00052-1. Epub [PubMed PMID: 27430455]
Nardone R, Pikija S, Mutzenbach JS, Seidl M, Leis S, Trinka E, Sellner J. Current and emerging treatment options for spinal cord ischemia. Drug discovery today. 2016 Oct:21(10):1632-1641. doi: 10.1016/j.drudis.2016.06.015. Epub 2016 Jun 17 [PubMed PMID: 27326910]
Cheshire WP, Santos CC, Massey EW, Howard JF Jr. Spinal cord infarction: etiology and outcome. Neurology. 1996 Aug:47(2):321-30 [PubMed PMID: 8757000]
Kumral E, Polat F, Güllüoglu H, Uzunköprü C, Tuncel R, Alpaydın S. Spinal ischaemic stroke: clinical and radiological findings and short-term outcome. European journal of neurology. 2011 Feb:18(2):232-239. doi: 10.1111/j.1468-1331.2010.02994.x. Epub [PubMed PMID: 20402756]
Nedeltchev K, Loher TJ, Stepper F, Arnold M, Schroth G, Mattle HP, Sturzenegger M. Long-term outcome of acute spinal cord ischemia syndrome. Stroke. 2004 Feb:35(2):560-5 [PubMed PMID: 14726546]
Salvador de la Barrera S, Barca-Buyo A, Montoto-Marqués A, Ferreiro-Velasco ME, Cidoncha-Dans M, Rodriguez-Sotillo A. Spinal cord infarction: prognosis and recovery in a series of 36 patients. Spinal cord. 2001 Oct:39(10):520-5 [PubMed PMID: 11641795]
Level 2 (mid-level) evidenceRobertson CE, Brown RD Jr, Wijdicks EF, Rabinstein AA. Recovery after spinal cord infarcts: long-term outcome in 115 patients. Neurology. 2012 Jan 10:78(2):114-21. doi: 10.1212/WNL.0b013e31823efc93. Epub 2011 Dec 28 [PubMed PMID: 22205760]
Hanson SR, Romi F, Rekand T, Naess H. Long-term outcome after spinal cord infarctions. Acta neurologica Scandinavica. 2015 Apr:131(4):253-7. doi: 10.1111/ane.12343. Epub 2014 Oct 27 [PubMed PMID: 25346212]
Romi F, Naess H. Spinal Cord Infarction in Clinical Neurology: A Review of Characteristics and Long-Term Prognosis in Comparison to Cerebral Infarction. European neurology. 2016:76(3-4):95-98 [PubMed PMID: 27487411]
Erhan B, Kemerdere R, Kizilkilic O, Gunduz B, Hanci M. Acute tetraplegia following laryngotracheal reconstruction surgery. Surgical neurology international. 2018:9():11. doi: 10.4103/sni.sni_405_17. Epub 2018 Jan 16 [PubMed PMID: 29416908]
Augoustides JG, Stone ME, Drenger B. Novel approaches to spinal cord protection during thoracoabdominal aortic interventions. Current opinion in anaesthesiology. 2014 Feb:27(1):98-105. doi: 10.1097/ACO.0000000000000033. Epub [PubMed PMID: 24322209]
Level 3 (low-level) evidenceKennedy P, Duff J, Evans M, Beedie A. Coping effectiveness training reduces depression and anxiety following traumatic spinal cord injuries. The British journal of clinical psychology. 2003 Mar:42(Pt 1):41-52 [PubMed PMID: 12675978]
Level 1 (high-level) evidencePeter C, Müller R, Cieza A, Geyh S. Psychological resources in spinal cord injury: a systematic literature review. Spinal cord. 2012 Mar:50(3):188-201. doi: 10.1038/sc.2011.125. Epub 2011 Nov 29 [PubMed PMID: 22124343]
Level 2 (mid-level) evidenceRagnarsson KT. Medical rehabilitation of people with spinal cord injury during 40 years of academic physiatric practice. American journal of physical medicine & rehabilitation. 2012 Mar:91(3):231-42. doi: 10.1097/PHM.0b013e3182489f5e. Epub [PubMed PMID: 22317933]